Bonding and properties in metals and alloys
Dr. Davinder Bhachu
Teacher

Metallic Bonding
Metallic bonding is the force that holds metal atoms together in a solid structure. Unlike ionic or covalent bonding, metallic bonding involves electrons that are free to move throughout the entire metal structure.
The Structure of Metals
Metals consist of a giant structure where atoms are arranged in a regular, repeating pattern (known as a lattice). The atoms are said to have regular arrangements because they have lots of atoms - exactly how many depends on how big the piece of metal is.

Key Features of Metal Structure:
Atoms are arranged in a regular way (often in rows or layers)
The structure is described as a "giant metallic structure" or "giant metallic lattice"
Metals contain huge numbers of atoms all bonded together
Bonding in Metals
In metals, the electrons in the outer shells of the atoms are delocalised. This means that they aren't associated with a particular atom or bond, so they're free to move through the whole structure.
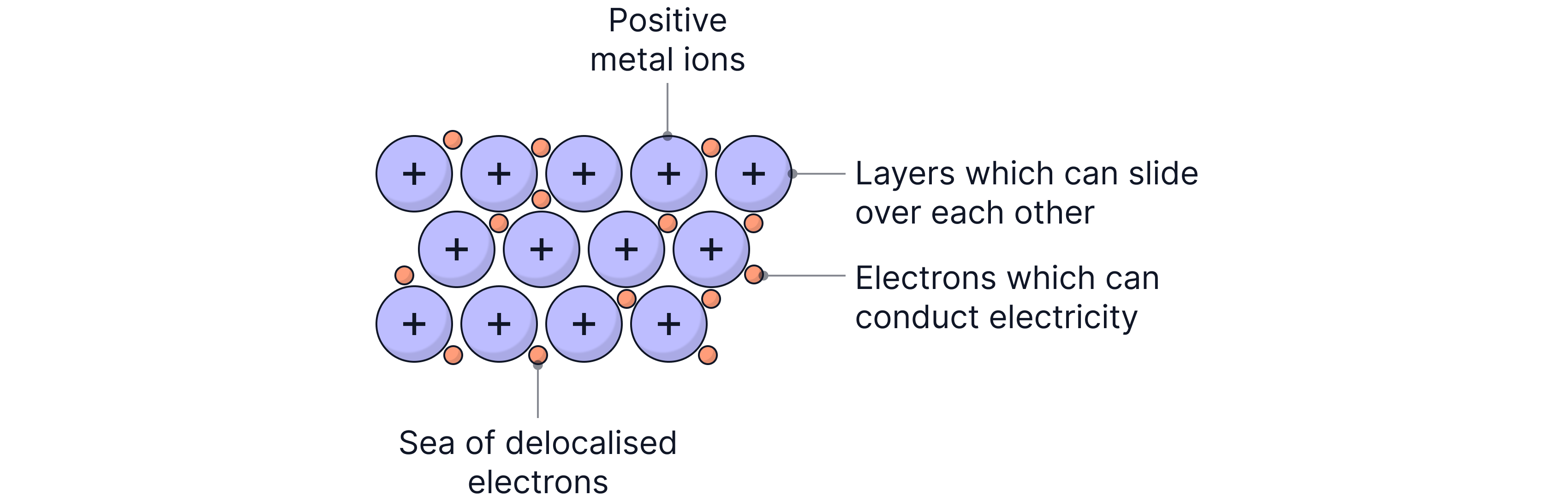
What Happens to the Atoms?
Metal atoms lose their outer shell electrons
They become positively charged metal ions
These ions remain fixed in position in the lattice
What Happens to the Electrons?
The outer electrons become delocalised (or "free")
They form a "sea of electrons" that can move throughout the structure
These mobile electrons are sometimes called "free electrons"
Think of a ping pong balls in a box arranged in fixed positions that represent the positive ions, surrounded by jelly which represents the sea of delocalised electrons.
The Metallic Bond is defined as the electrostatic force of attraction between:
The positive metal ions, and
The delocalised (negative) electrons
These forces hold the metal structure together.
Properties of Metals
1. High Melting and Boiling Points
Metals have very high melting and boiling points because:
The electrostatic forces between the metal ions and delocalised electrons are very strong
A lot of energy is needed to break these forces
Most compounds with metallic bonds have very high melting and boiling points, so they're generally solid at room temperature
Therefore, metals are solid at room temperature (except mercury).
2. Electrical and Thermal Conductivity
Electrical Conductivity: Metals conduct electricity well because:
They have delocalised electrons that are free to move through the whole structure
These electrons can carry electric charge through the metal
The electrons act as charge carriers
Thermal Conductivity: Metals are good conductors of heat because:
The delocalised electrons are free to move
They can transfer thermal energy quickly through the structure
The electrons carry the thermal energy through the metal
Science in Action: The high thermal conductivity of metals makes them ideal for cooking pots and pans. Steel and aluminium cookware needs to be durable, have a very high melting point and be unreactive so that the pan does not react with the food being cooked. The metal chosen for pans also needs to conduct heat well so that the heat transfers efficiently from the hob to the food being cooked.
3. Malleability
Metals are malleable - they can be bent, shaped, hammered or rolled into different forms without breaking.
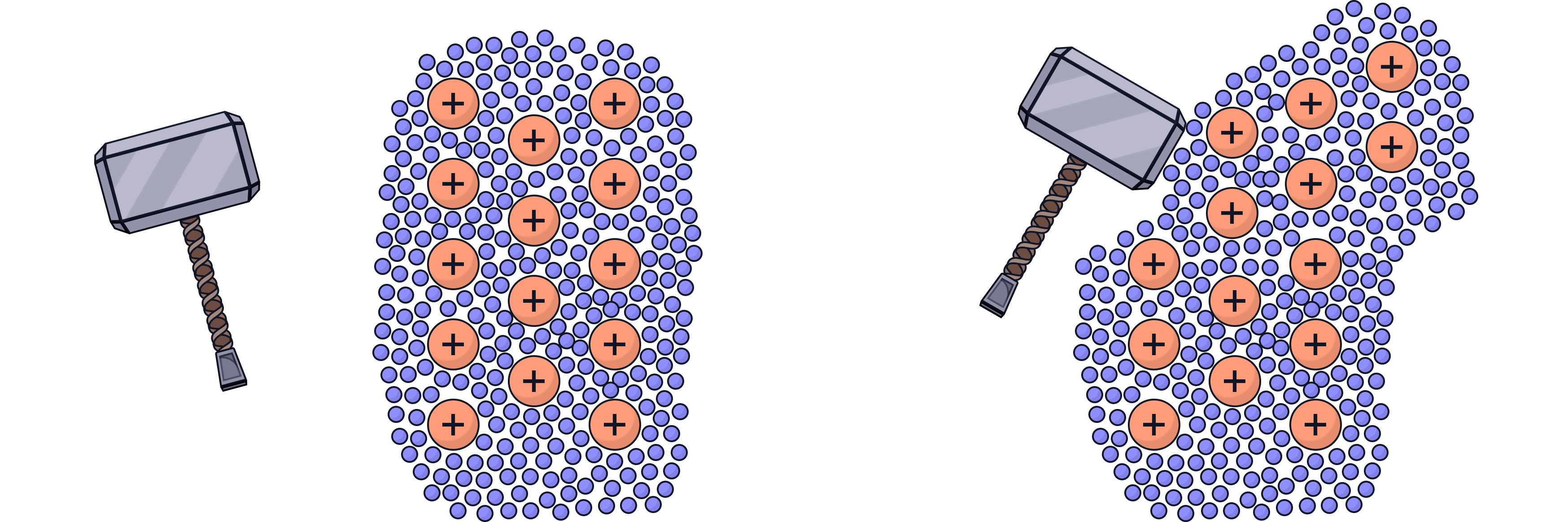
Why are metals malleable?
Metals consist of atoms held together in a regular structure
The atoms form layers that are able to slide over each other
When force is applied, the layers can move while the metallic bonding remains intact
The delocalised electrons continue to hold the structure together even after the layers have moved
This means metals can be bent and shaped without shattering, and they can also be hammered or rolled into different shapes.
Alloys
Pure metals often aren't quite suitable for certain jobs - they're often too soft when they're pure. To overcome this, metals are mixed with other metals (or sometimes other elements) to make them harder. These mixtures are called alloys.
Definition: An alloy is a mixture of two or more metals, or a metal and another element. Alloys are harder than pure metals.
Examples of Alloys:
Alloy | Composition | Uses |
Brass | Copper + zinc | Pipes, musical instruments |
Bronze | Copper + tin | Sculptures, electrical connectors |
Steel | Iron + carbon | Construction, tools, machinery |
Gold alloys | Gold + copper/silver | Jewellery (makes gold harder) |
Why Are Alloys Harder Than Pure Metals?
Different elements have different sized atoms. When another element is mixed with a pure metal, the new element atoms will distort (disrupt) the layers of metal atoms.
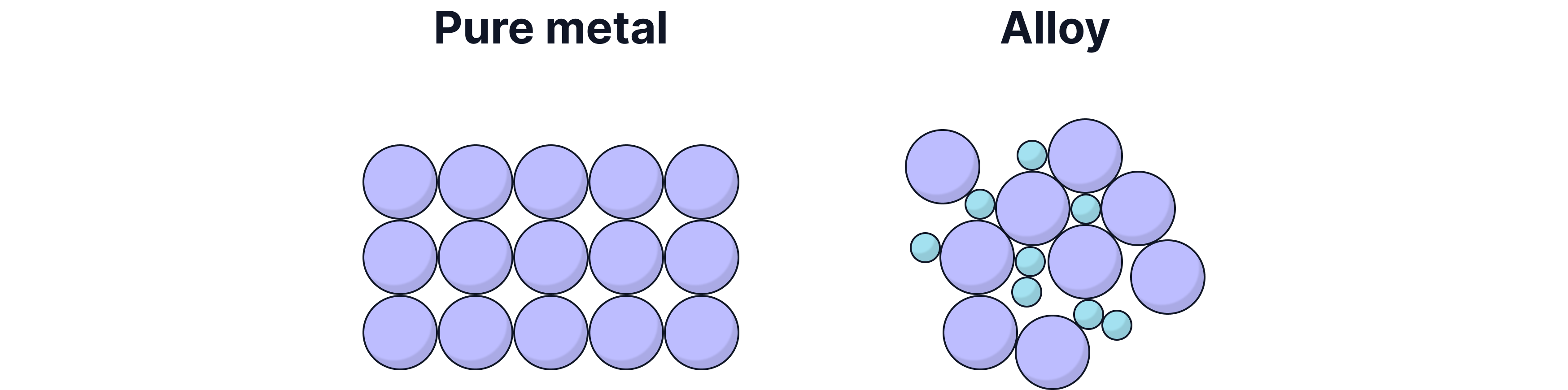
This distortion makes it more difficult for the layers to slide over each other, which is why alloys are harder than pure metals.
Comparison Table: Metallic vs Giant Covalent Structures
Property | Metallic Structures | Giant Covalent Structures |
Bonding | Metallic bonding (electrostatic attraction between metal ions and delocalised electrons) | Strong covalent bonds throughout structure |
Melting/Boiling Points | High (strong metallic bonds need lots of energy to break) | Very high (many strong covalent bonds throughout lattice) |
Electrical Conductivity | Yes (delocalised electrons can move and carry charge) | No (no free electrons or ions) |
Malleability | Yes (layers can slide over each other) | No (rigid 3-D network) |
Solubility in water | Insoluble (but some react with water) | Insoluble |
Practice Questions
Recall
Q1. Describe the structure of a metal.
Metals have a giant structure in which atoms are arranged in a regular pattern (layers). The outer electrons from the metal atoms are delocalised and free to move throughout the structure.
Q2. What type of forces hold the particles in a metal together?
Metallic bonding — the electrostatic forces of attraction between the positive metal ions and the delocalised (negative) electrons.
Apply
Q3. Explain why metals can be easily bent.
Metals consist of layers of atoms that are able to slide over each other. The metallic bonding holds the structure together even when the layers move, so metals can be bent and shaped without breaking.
Q4. Metals are good conductors of heat. Explain why.
Metals have delocalised electrons that are free to move through the whole structure. These electrons can transfer thermal energy quickly through the metal.
Q5. Why are alloys harder than pure metals?
Alloys contain atoms of different sizes. The different-sized atoms distort/disrupt the layers of metal atoms, making it more difficult for the layers to slide over each other. This makes alloys harder than pure metals.