Allotropes of Carbon
Dr. Davinder Bhachu & MyEdSpace Chemistry
Teacher


Contents
Allotropes of Carbon
Carbon is unusual because it can catenate — it can bond to itself repeatedly, forming chains, rings, layers and 3-D networks. Each carbon atom can form four covalent bonds, and this flexibility allows carbon to exist in several different structural forms called allotropes.
Even though allotropes are made of the same element, differences in their bonding and structure give them very different properties and uses. The four you need to know are:
Diamond
Graphite
Graphene
Fullerenes (including nanotubes)
Diamond
Structure:
Giant covalent structure
Each carbon atom forms 4 strong covalent bonds in a 3-D tetrahedral network.
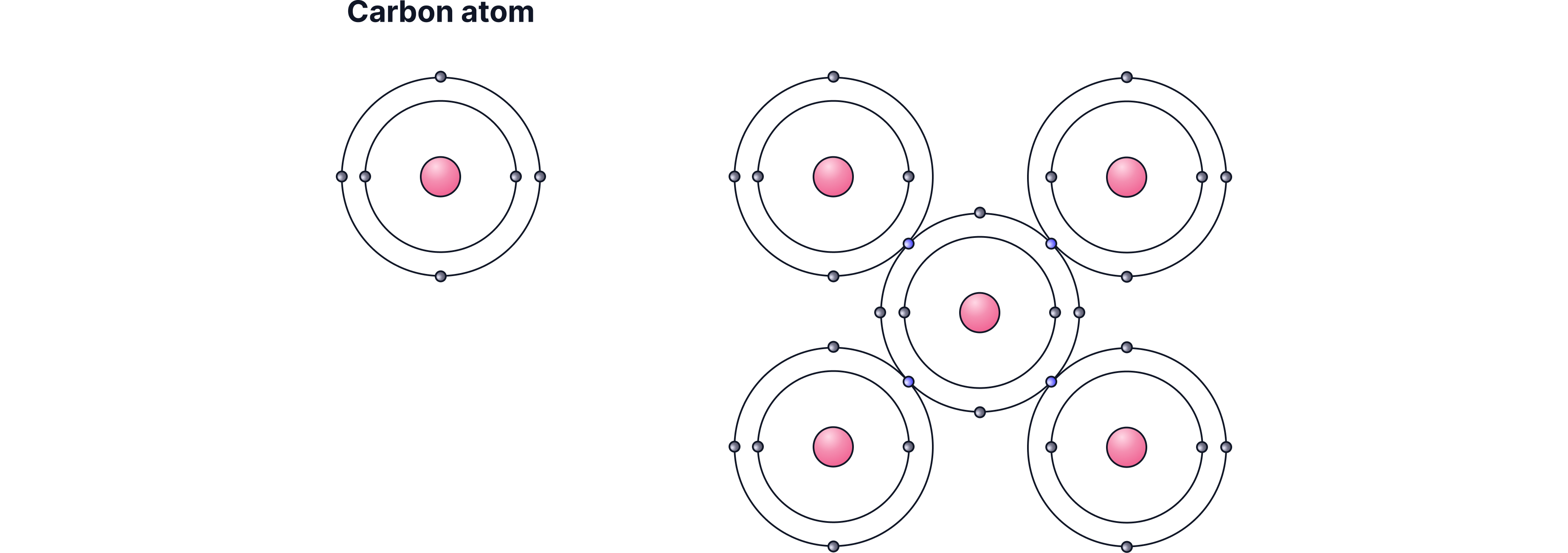
This creates a rigid giant covalent structure.
Properties Explained by Structure:
Very hard - every atom is strongly bonded in all directions.
Very high melting point - many strong covalent bonds must be overcome.
Does not conduct electricity - no free electrons or ions.
Insoluble - covalent network is too strong to break.
Uses:
Cutting tools, drill tips, jewellery.
Diamond is hard because each carbon atom is bonded to four other carbons, forming a rigid structure.
Graphite
Structure:
Giant covalent structure
Each carbon atom forms 3 covalent bonds, creating flat hexagon layers.
The fourth electron becomes delocalised, able to move within the layer but cannot move perpendicular to the layer.
The layers themselves are held together by weak forces, so they can slide and rub off easily.
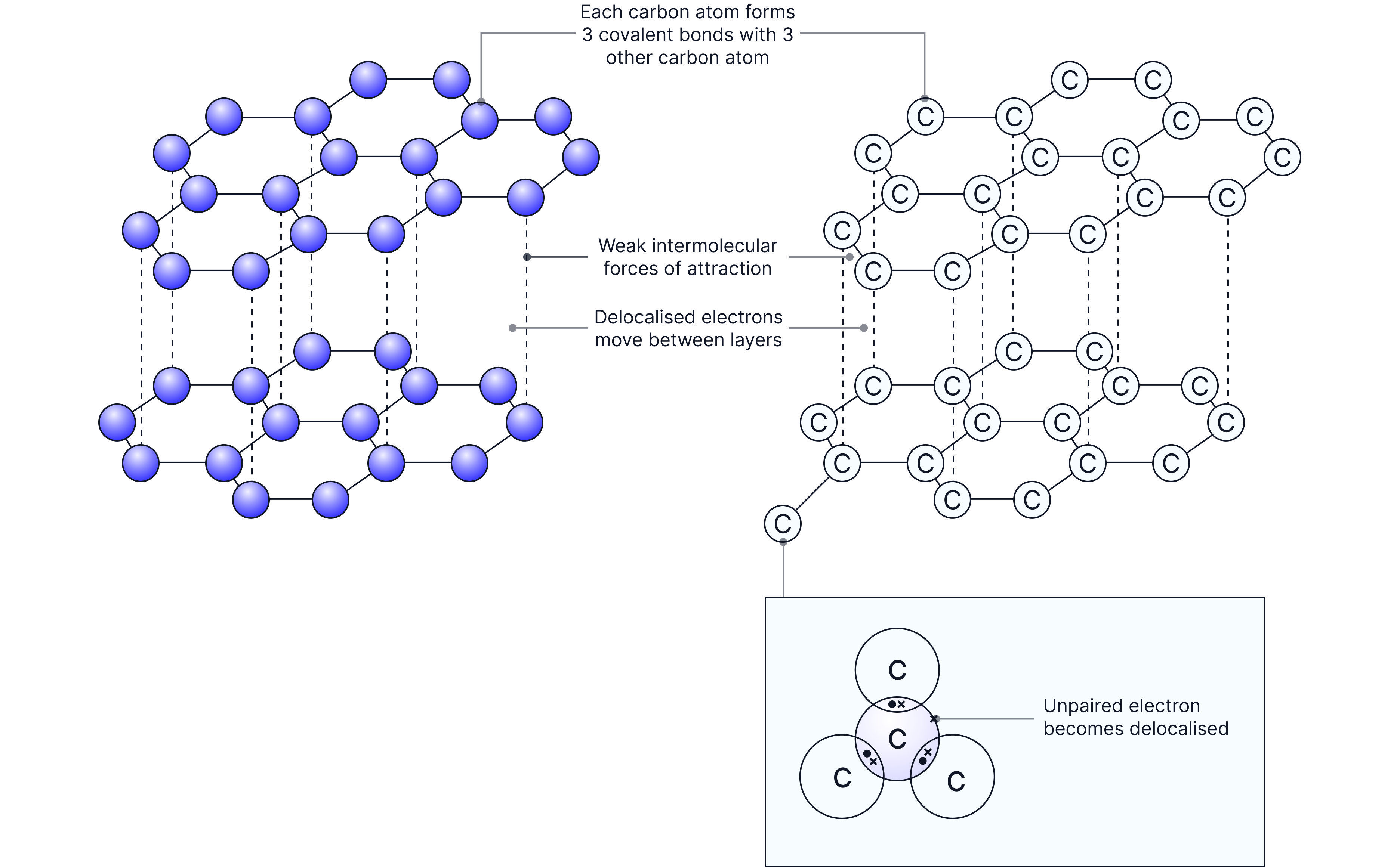
Properties Explained by Structure:
Slippery and soft — layers slide easily.
Conducts electricity and heat — delocalised electrons move through the structure.
Very high melting point — covalent bonds within layers are still strong.
Uses:
Lubricants, electrodes, pencil “lead”.
💬 Misconception warning:
Graphite is soft not because covalent bonds are weak, but because the forces between the layers are weak.
Graphene
Structure:
Giant covalent structure
A single layer of graphite, just one atom thick.
Atoms arranged in a hexagonal sheet with delocalised electrons.
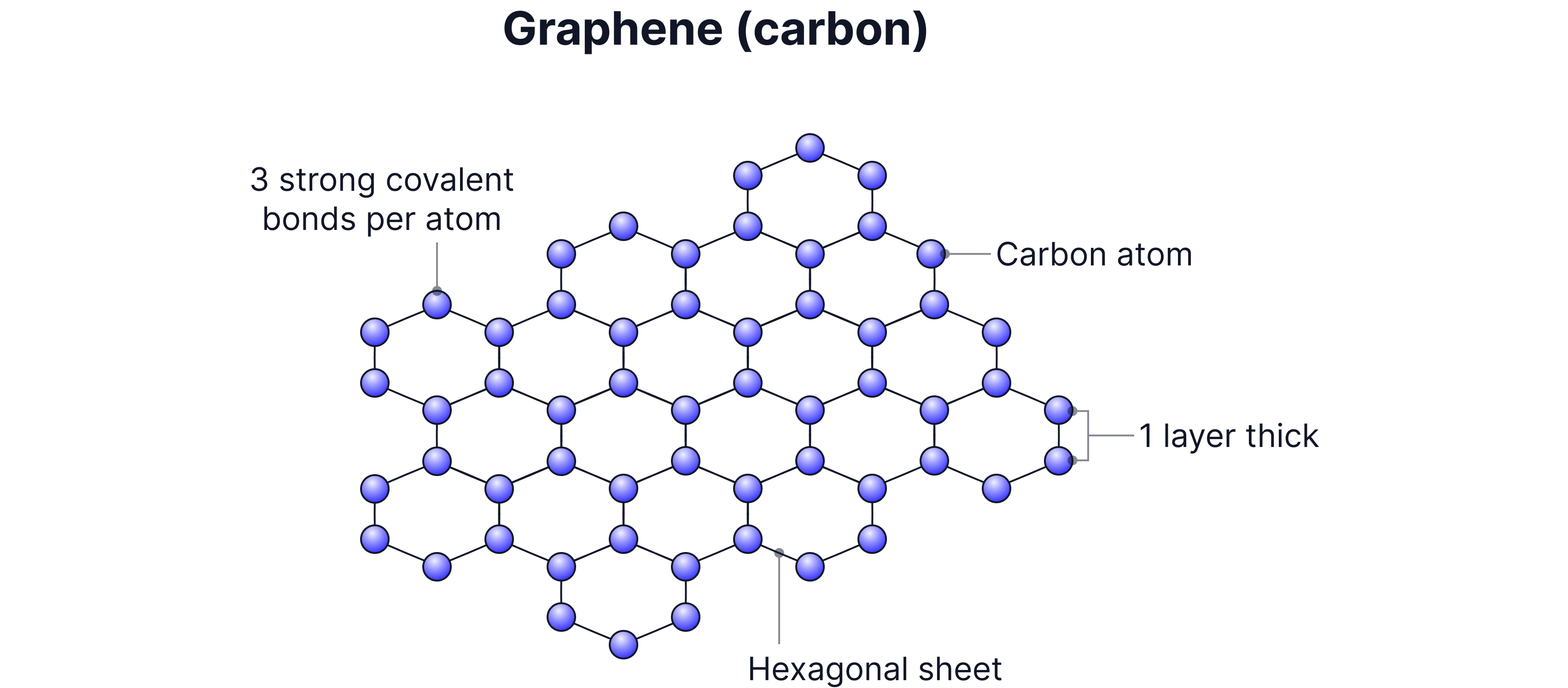
Properties:
Extremely strong (due to covalent network)
Flexible
Very light (one layer only)
Excellent conductor of electricity and heat
Uses:
Flexible electronics, composite materials, high-strength lightweight structures.
Fullerenes and Nanotubes
Fullerenes are simple molecular forms of carbon. Unlike diamond, graphite and graphene (which have giant covalent structures), fullerenes exist as individual molecules made of carbon atoms arranged in hollow shapes such as balls, cages or tubes.
The carbon atoms are joined by strong covalent bonds, but only within each molecule — the molecules are held to each other by weak intermolecular forces.
This means fullerenes typically have lower melting points than diamond or graphite and they are often soft or slippery.
Buckminsterfullerene (C₆₀)
Structure:
A simple molecular substance
A spherical molecule made of 20 hexagons and 12 pentagons
Each carbon forms 3 covalent bonds
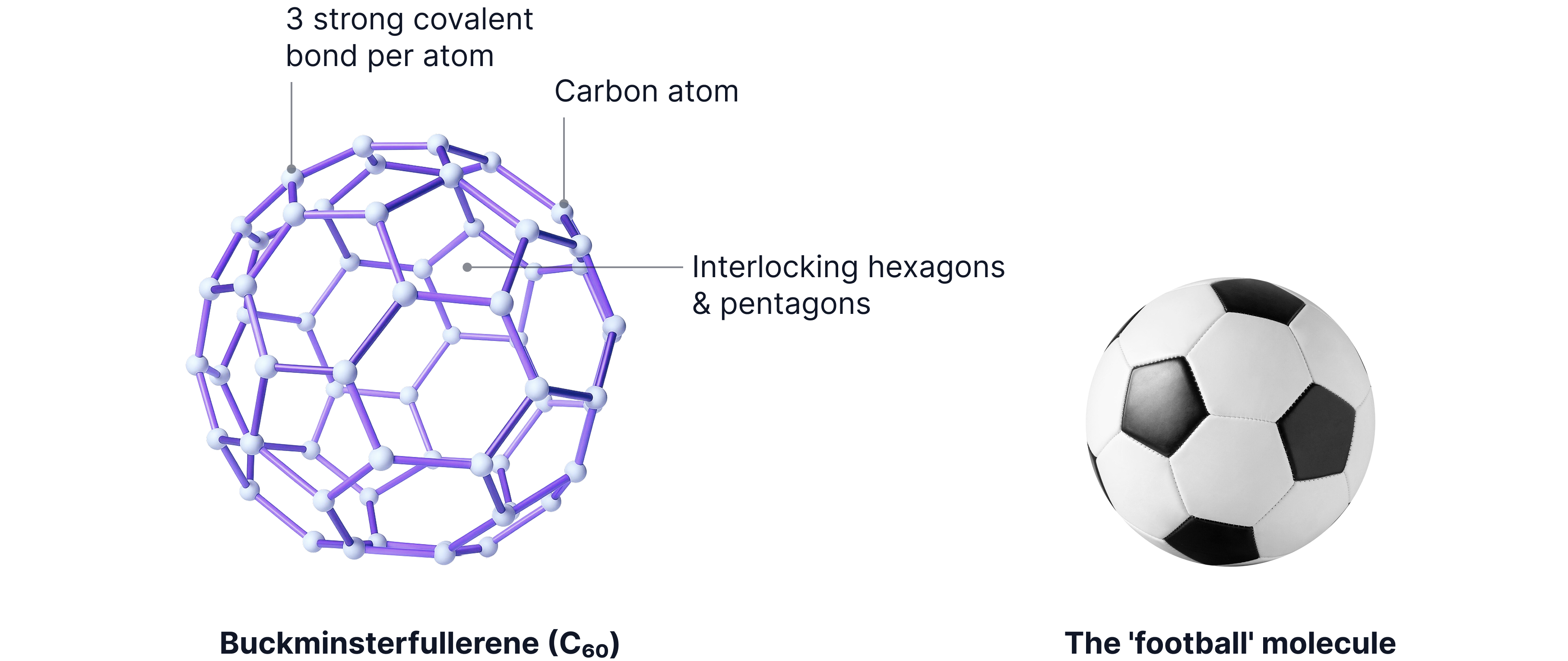
Properties (explained by structure):
Low melting point (weak intermolecular forces between C₆₀ molecules)
Soft (molecules can roll over each other)
Does not conduct electricity well (electrons are not fully delocalised across molecules)
Uses:
Drug delivery (hollow cage can trap molecules), which can then be used to deliver drugs at specific sites in a controlled way
Lubricants (ball shape reduces friction)
Catalysts They have a large surface area, making them suitable as catalyst supports (where catalysts could be attached to fullerenes)
Carbon Nanotubes
Structure:
A form of fullerene shaped into a long cylinder
Still a simple molecular structure, but with very long tubular molecules
Each carbon atom forms 3 covalent bonds in hexagonal rings

Properties (explained by structure):
Very strong (covalent bonding within each tube gives high tensile strength)
Conduct electricity and heat (electrons can move along the tube)
High melting point compared to C₆₀ (molecules are very long therefore there are stronger intermolecular forces than spherical fullerenes)
Uses:
Reinforcing materials (strong but lightweight)
Electronics and nanotechnology
Sports equipment, bike frames, tennis rackets
Summary
Carbon form | Structure Type | Conducts? | MP/BP | Notes |
C₆₀ fullerene | Simple molecular | Poor conductor | Low | Soft, spherical |
Nanotube | Simple molecular (long tubular molecules) | Good conductor | Medium | Very hard |
Diamond | Giant covalent | No | Very high | Very hard |
Graphite | Giant covalent (layered) | Yes | Very high | Layers slide |
Graphene | Giant covalent (single layer) | Yes | Very high | Strong + light |
Practice Questions
Recall
Q1. What is an allotrope?
An allotrope is a different structural form of the same element.
Allotropes have the same type of atoms but arranged differently, giving them different properties.
Q2. Why is diamond extremely hard?
Each carbon atom in diamond forms four strong covalent bonds arranged in a rigid 3-D tetrahedral structure. Because all atoms are strongly bonded throughout the lattice, a lot of energy is needed to break the structure, making diamond very hard.
Give one reason why nanotubes are useful in materials science.
Nanotubes are very strong but lightweight , so they can be used to reinforce materials such as sports equipment or aircraft composites.
Alternatives:
Nanotubes can conduct electricity, making them useful in nanoelectronics.
They have a large surface area, making them suitable as catalyst supports.
Which allotrope is a single layer of carbon atoms?
Graphene.
(Higher-tier note:)
Graphene is one layer of graphite arranged in a hexagonal lattice one atom thick.
Apply
Explain why graphite can conduct electricity.
In graphite, each carbon forms three covalent bonds.
This leaves one electron per atom delocalised.
These delocalised electrons can move through the layers and carry charge, so graphite conducts electricity.
Explain why buckminster fullerenes are useful as lubricants.
They have a spherical shape so the molecules can roll over each other
Challenge
What physical property do buckminster fullerenes have that is very different to the other allotropes of carbon? Explain your answer.
Buckminsterfullerenes have a low melting point compared to other allotropes of carbon.
Explanation:
Buckminsterfullerenes are made of simple molecular structures
There are weak intermolecular forces between the molecules
Only a small amount of energy is needed to overcome these forces