Giant covalent structures
Dr. Davinder Bhachu & MyEdSpace Chemistry
Teacher


Giant Covalent Molecules (Macromolecules)
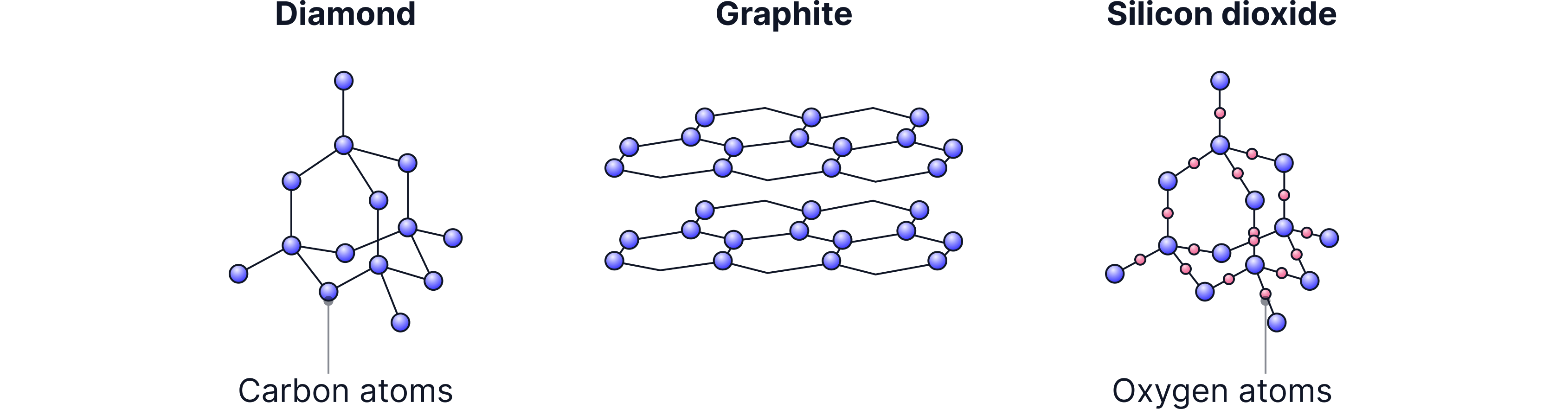
Giant covalent structures are huge 3-D networks in which all atoms are joined by strong covalent bonds. Instead of existing as small, separate molecules, the atoms are linked in a continuous structure (a lattice) that can contain thousands or even millions of atoms.
These substances are sometimes called macromolecules.
Key idea:
Simple molecules = small groups of atoms with weak forces between molecules.
Giant covalent structures = every atom covalently bonded (strongly) throughout, making one enormous structure.
Melting and Boiling Points
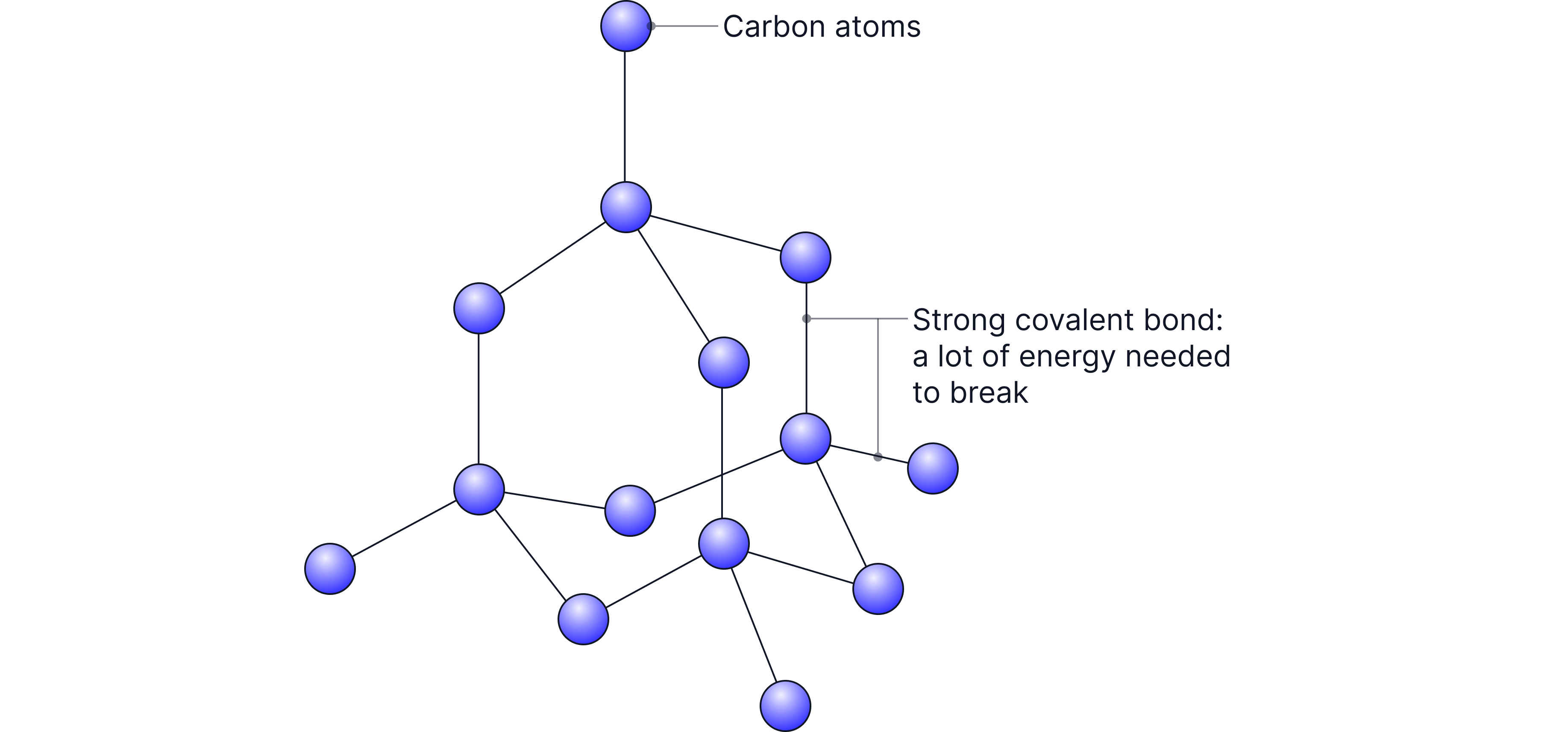
Giant covalent structures have very high melting and boiling points because:
Every atom is held in place by strong covalent bonds
These bonds must be overcome throughout the entire structure to melt or boil the substance
This needs a huge amount of energy
Therefore, giant covalent structures are solid at room temperature.
Electrical Conductivity
Most giant covalent structures do not conduct electricity because:
They have no free electrons
They have no ions to carry charge
Exception: some carbon allotropes (graphite) can conduct electricity — but this will be covered in your next note.
Solubility
Giant covalent substances are insoluble in water and other solvents. The covalent bonds are too strong to be pulled apart by water molecules.
Examples
Three common giant covalent structures you need to know are:
Substance | Elements in structure | Notes |
Silicon dioxide (SiO₂) | Silicon + oxygen | Found in sand, glass, quartz |
Silicon (Si) |
Silicon only | Used in computer chips and solar cells |
Carbon allotropes (diamond, graphite, graphene) | Carbon only | — |
Comparison Recap:
Type of substance | Bonding | Forces between particles | MP/BP | Conducts? |
Simple molecular | Strong covalent inside molecules; weak forces between molecules | Weak intermolecular | Low | No |
Giant covalent | Strong covalent bonds throughout structure | — | Very high
| No |
Exam tip:
When explaining high melting points for giant covalent structures, do not talk about breaking intermolecular forces — mention strong covalent bonds throughout the lattice.
Practice Questions
Recall
Q1. What is a giant covalent structure?
A substance where atoms are joined by strong covalent bonds in a continuous 3-D network.
Q2. Explain why giant covalent substances have such high melting points.
Because many strong covalent bonds must be broken throughout the whole structure, which needs a lot of energy.
Apply
Q3. Do giant covalent substances conduct electricity? Give a reason.
No — there are no free electrons or ions to carry charge.
Q4. Why are giant covalent substances insoluble in water?
The covalent bonds are too strong to break, so water cannot separate the atoms.
Q5. Which would have the higher melting point: silicon dioxide or oxygen (O₂)? Explain your answer.
SiO₂, because it is a giant covalent structure, not simple molecular — many strong covalent bonds require lots of energy to be broken.
Challenge
Q6. Put these in order of melting point (lowest → highest): O₂, SiO₂, poly(ethene)
O₂ (lowest) → poly(ethene) → SiO₂ (highest)