Properties of simple molecules (including polymers)
Dr. Davinder Bhachu
Teacher

Contents
What Are Simple Molecular Substances?
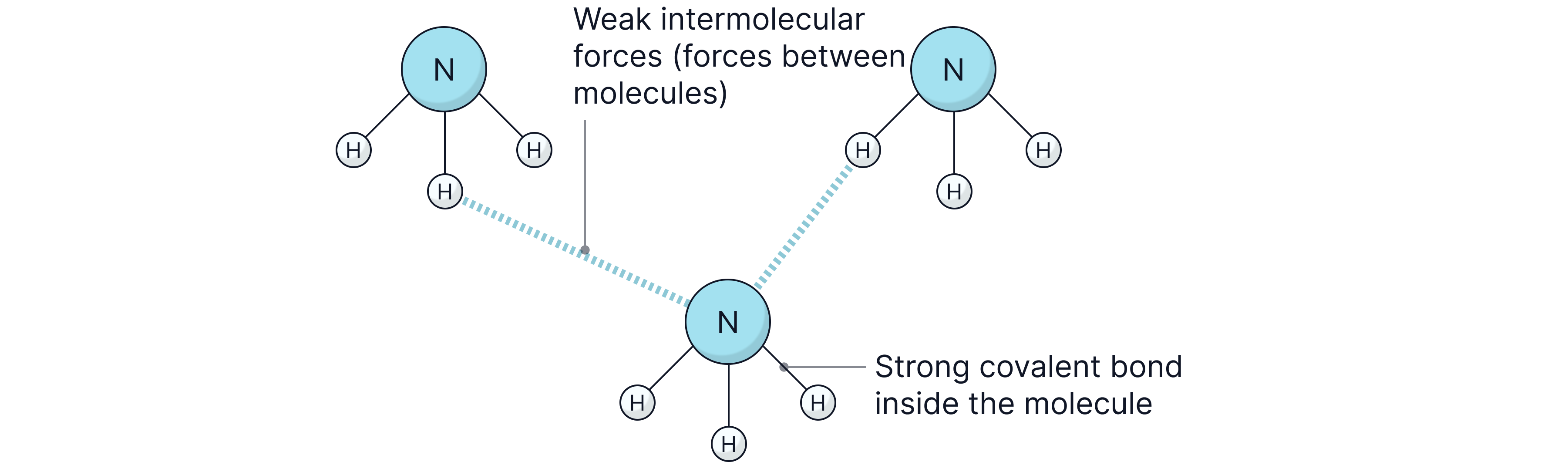
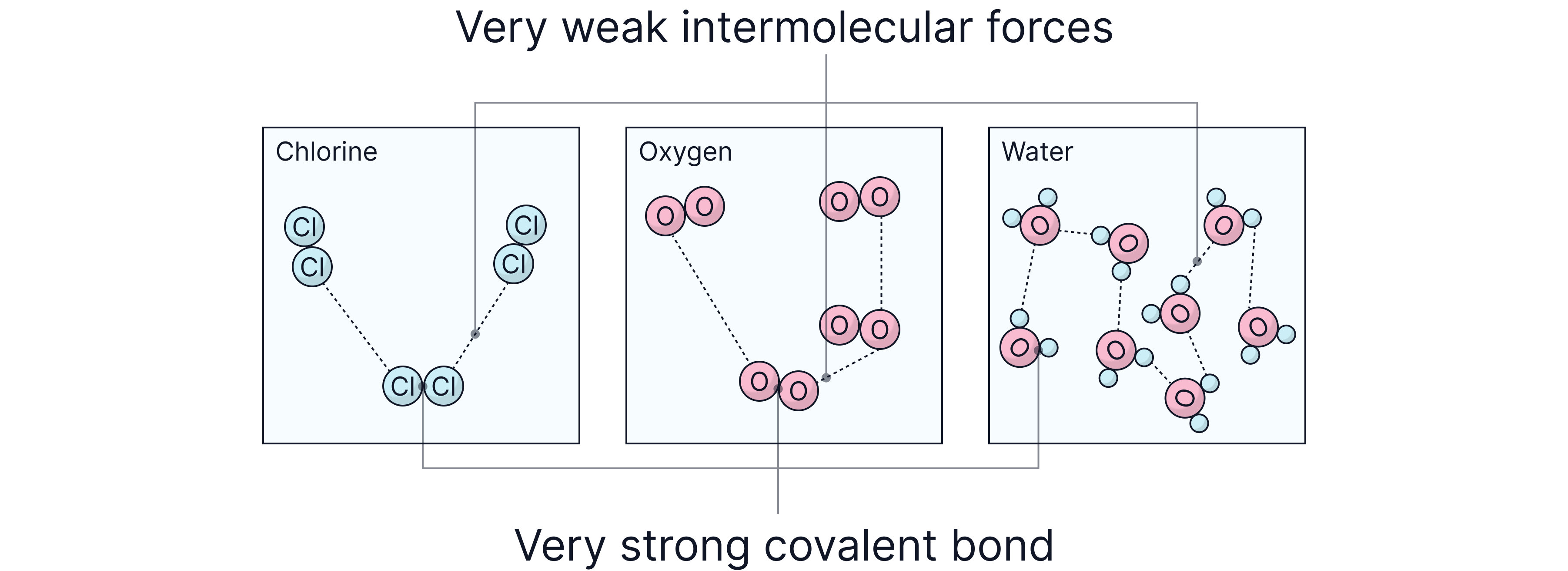
Simple molecular substances are made of small molecules containing a fixed number of non-metal atoms joined by strong covalent bonds. These molecules are held next to each other by weak intermolecular forces.
Examples include ammonia (NH₃), fluorine (F₂), and water (H₂O) etc.
Key idea:
Inside each molecule = strong covalent bonds
Between molecules = weak intermolecular forces
Electrical Conductivity
Simple molecular substances do not conduct electricity in any state.
They have no ions and no free electrons
There is nothing that can move and carry charge
Therefore, simple molecular substances are electrical insulators.
Melting and Boiling Points
Even though covalent bonds inside the molecules are strong, the forces between molecules (intermolecular forces) are very weak.
Because these weak forces are easy to overcome:
Little energy is needed to separate molecules
Melting and boiling points are low
Many simple molecular substances are gases or liquids at room temperature
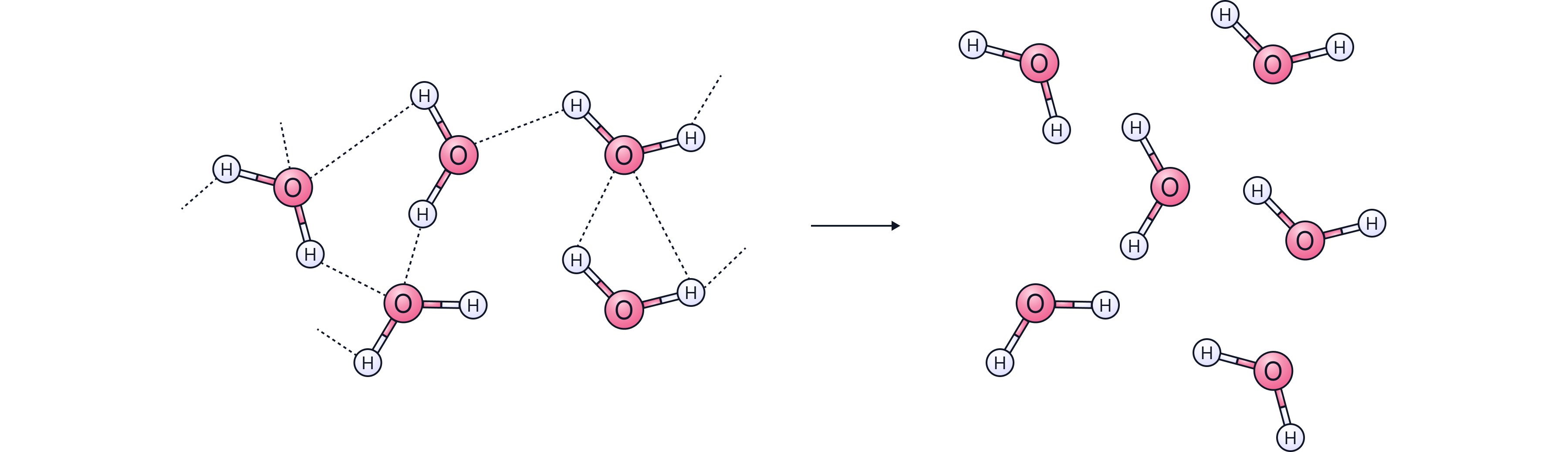
Exam tip: When melting or boiling simple molecules, you break intermolecular forces — not covalent bonds.
Why Do Some Simple Molecules Have Higher Melting Points Than Others?
As molecules get bigger, they have more electrons, which increases the strength of intermolecular forces.
So:
Molecular Size | Intermolecular Forces | Melting & Boiling Points |
small | very weak | very low |
larger | stronger (still weak overall) | higher (but still low compared to ionic/giant covalent) |
Example Trend (Group 7 molecules comparison)
Molecule | State at Room Temp | Reason |
F₂ | gas | smallest molecules, weakest forces |
Br₂ | liquid | bigger molecule, stronger forces |
I₂ | solid | biggest molecules, strongest forces |
Polymers — Larger Covalent Molecules
Polymers are very long molecules made when many small molecules (monomers) link together.
Atoms in a polymer are joined by strong covalent bonds.
Polymer chains are much longer than simple molecules.
Polymer chains have stronger intermolecular forces than simple molecules (due to their length and size).
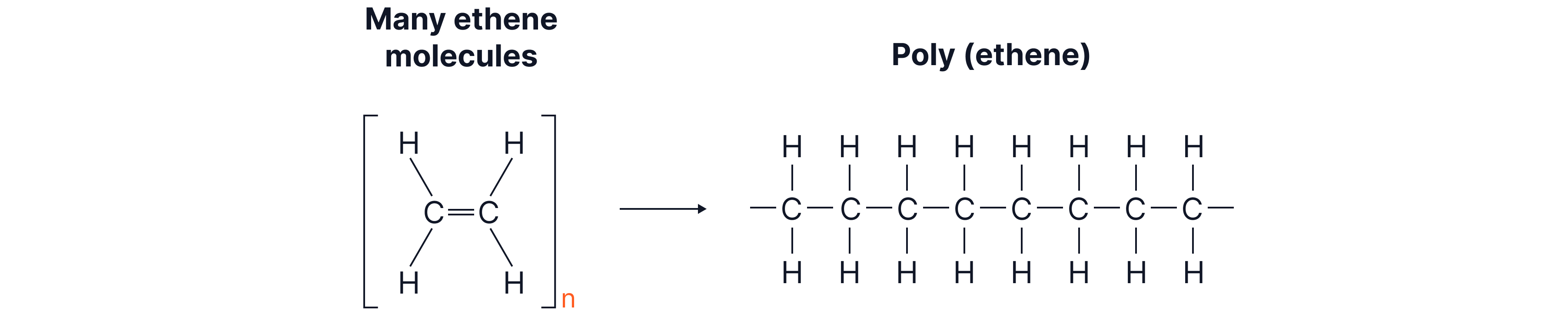
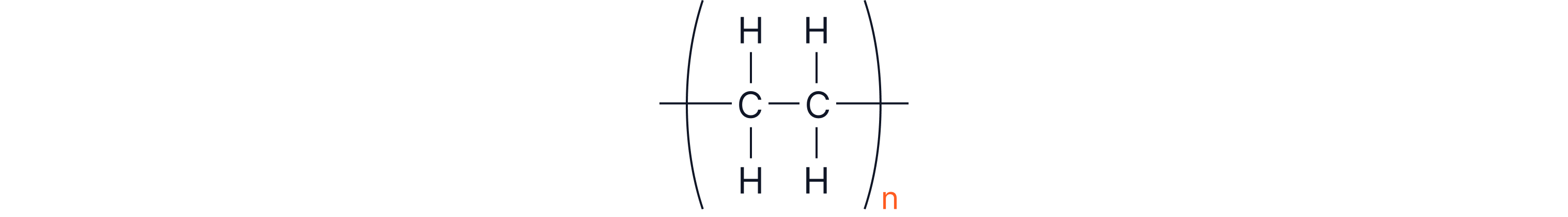
This shows a repeat unit of polyethene. This is the way of representing lots of ethene units bonded together. To show this, we draw the unit that repeats lots of times i.e. with brackets around it and the bonds that go through the brackets show them joining up to the neighbouring repeat units. To highlight that there are many repeat units, n is written outside of the brackets (n is a large number which tells us that this unit is repeated many times).
As a result:
Polymers are usually solids at room temperature, even though the forces between the chains are still weaker than those in giant covalent or ionic lattices.
Melting and Boiling Points of Polymers
Compared to simple molecules:
Polymer melting/boiling points are higher (stronger intermolecular forces between the long chains)
But they are still lower than giant covalent structures such as diamond or silicon dioxide
Substance Type | Bonds Inside | Forces Between | MP/BP | Conducts? |
Simple molecular | Strong covalent | Weak intermolecular | Low | No |
Polymer | Strong covalent | Moderate intermolecular | Medium | No |
Giant covalent | Strong covalent (network) | — | Very high | No (except graphite) |
Practice Questions
Recall
What type of forces exist between simple molecules?
Weak intermolecular forces
What happens to intermolecular forces when molecules get bigger?
They become stronger because there are more electrons and stronger attractions between molecules as a result
Apply
Why don’t simple molecular substances conduct electricity?
No ions or free electrons → nothing to carry charge.
Why do simple molecular substances have low melting points?
Only weak forces between the molecules → so very little energy needed to separate the molecules.
Which has stronger intermolecular forces: CO₂ or C₆H₁₄? Explain why.
C₆H₁₄ → larger molecule => more electrons, therefore stronger intermolecular forces.
Challenge
Why is iodine a solid at room temperature but fluorine is a gas?
I₂ has more electrons → stronger intermolecular forces → higher melting/boiling points.
Why do polymers have higher melting points than simple molecules?
Polymer chains are long → more intermolecular forces → more energy needed to separate chains.