Covalent Bonding
Dr. Davinder Bhachu
Teacher

Contents
Definition
Covalent bonding occurs when two non-metal atoms share pairs of electrons. Each atom involved in the bond counts the shared electrons as part of its own outer shell, helping both atoms achieve a full outer shell and become more stable (similar to noble gases).
The covalent bond itself is a strong electrostatic force of attraction between:
the positive nuclei of the atoms
and the shared pair(s) of electrons
Key idea:
Ionic bonding = electrons transferred.
Covalent bonding = electrons shared.
Covalent bonds are found in:
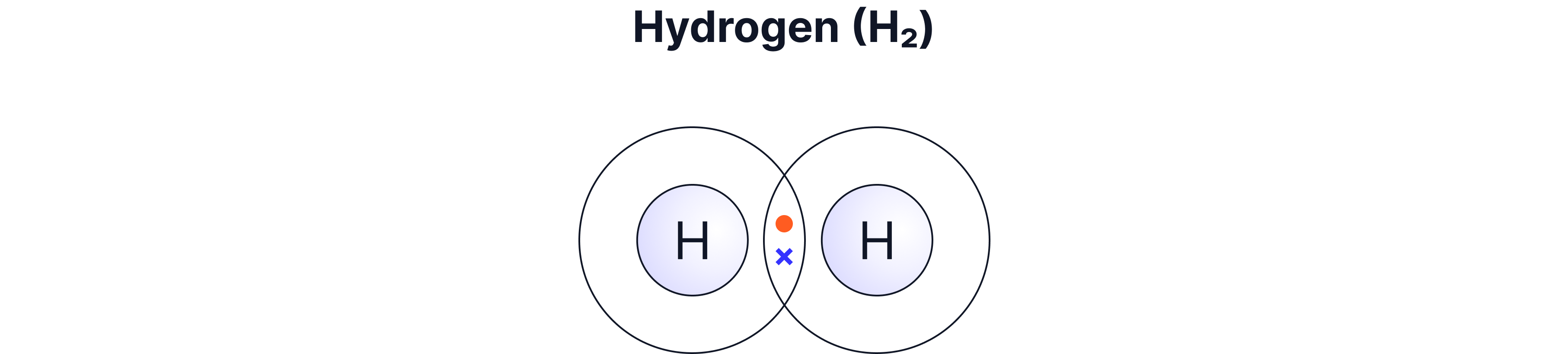
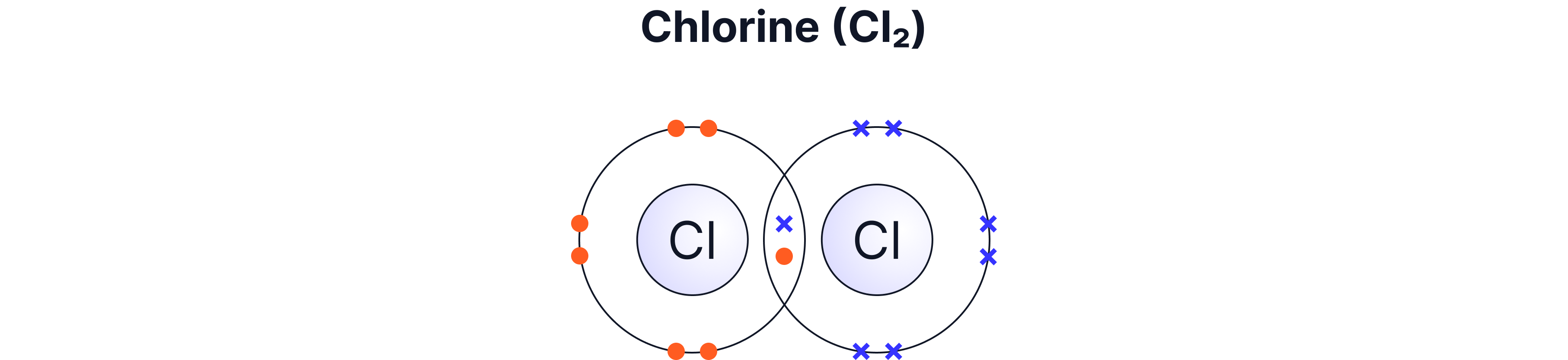
Non-metal elements such as H₂, O₂, Cl₂
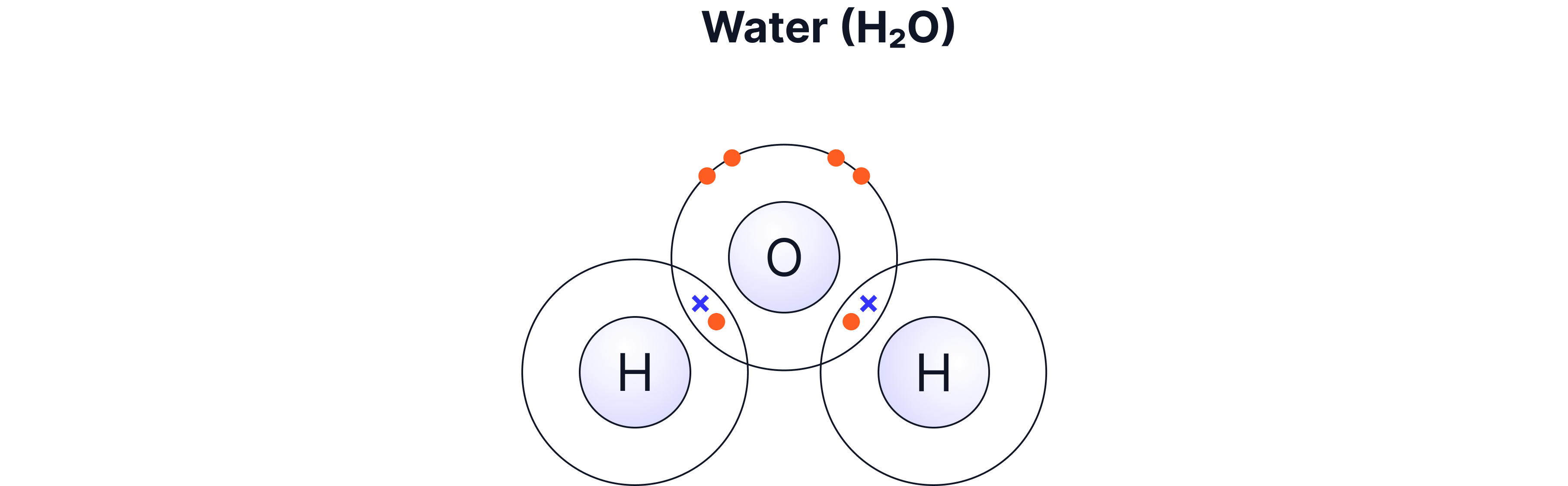
Compounds of non-metals such as H₂O, CH₄, CO₂
Double and Triple Covalent Bonds
Sometimes, atoms must share more than one pair of electrons to fill their outer shells. This creates double bonds (2 shared pairs) or triple bonds (3 shared pairs).
Type of bond | Electrons shared | Example |
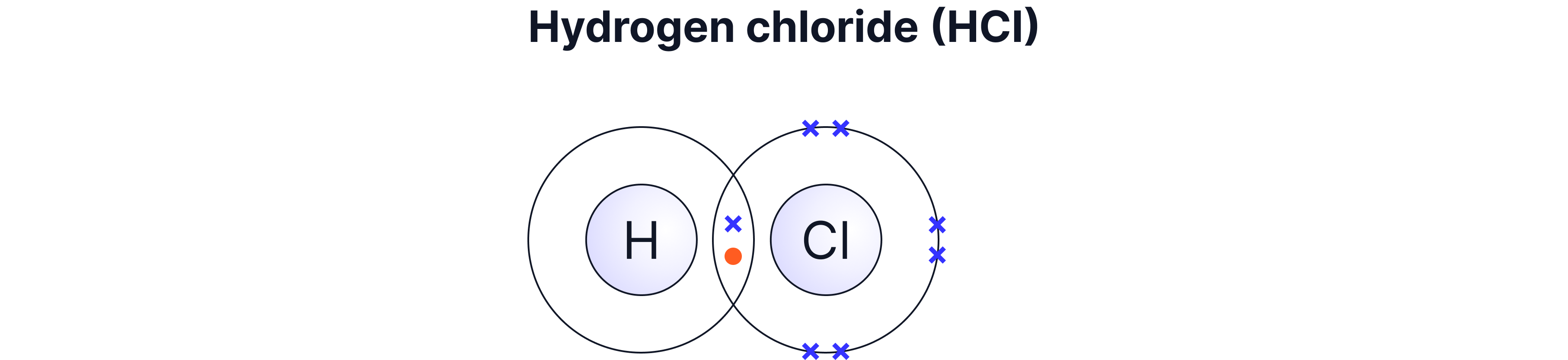
Single | 1 pair | H₂, HCl, CH₄ |
Double | 2 pairs | O₂, CO₂ |
Triple | 3 pairs | N₂ |
Multiple bonds are stronger and shorter than single bonds. The method for dot and cross diagrams for molecules with multiple bonds is the same as before:
Step 1 — Count outer electrons
Step 2 — Work out how many electrons are needed
Step 3 — Add shared pairs (1 pair = 1 bond)
Step 4 — Add remaining lone pairs
Example A — Oxygen (O₂) — Double Bond
Each oxygen atom has 6 electrons in its outer shell
Each oxygen atom needs 2 more electrons
So they share 2 pairs → a double bond
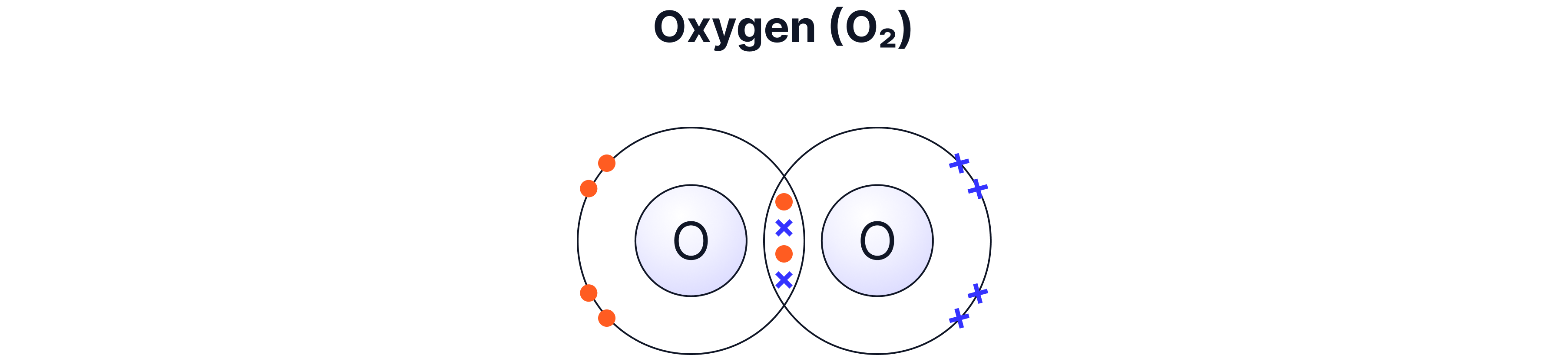
Oxygen forms two bonds because it needs two more electrons to complete its outer shell.
Example B — Nitrogen (N₂) — Triple Bond
Each nitrogen atom has 5 outer electrons
Each nitrogen atom needs 3 more electrons
So they share 3 pairs → a triple bond
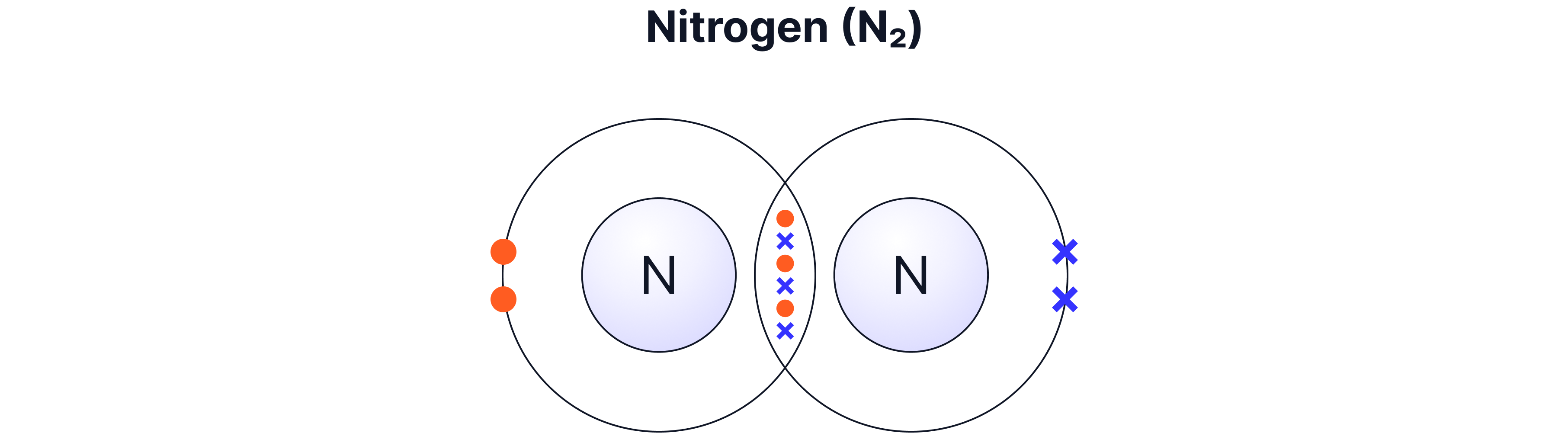
Nitrogen forms three bonds because it needs three electrons to fill its outer shell.
Example C — Carbon Dioxide (CO₂) — Two Double Bonds
Carbon has 4 outer electrons → needs 4 more
Each oxygen has 6 outer electrons → needs 2 more
Carbon shares 2 pairs with each oxygen
This makes two double bonds
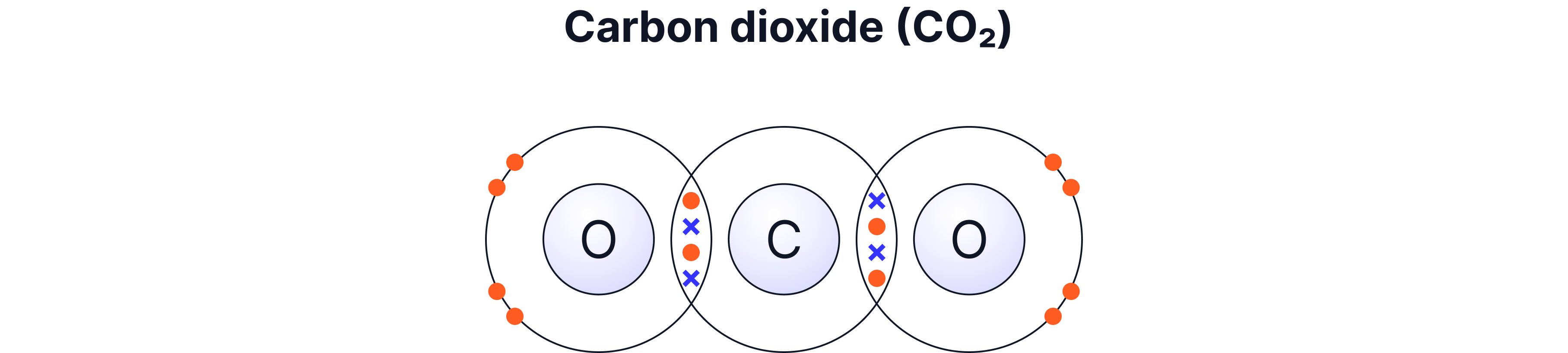
Carbon forms four shared electrons in total, making two double bonds — one to each oxygen atom.
Representing Covalent Molecules
There are several different ways to show covalent bonding. Each model highlights some helpful features of molecules, but no model shows everything. You have already seen dot-and-cross diagrams above, so the models below build on that idea and show the molecules in different ways.
a) Dot-and-Cross Diagram
What it shows well:
Which electrons are shared
Which electrons are lone pairs
Strength: Clear for bonding and electron placement
Limitation: Not 3-D and not realistic for shape
b) Displayed Formula

What it shows:
Each covalent bond as a line (1 line = 1 shared pair)
Pros:
Very clear and quick to interpret
Cons:
Doesn’t show lone pairs or 3-D arrangement
c) Ball-and-Stick Model
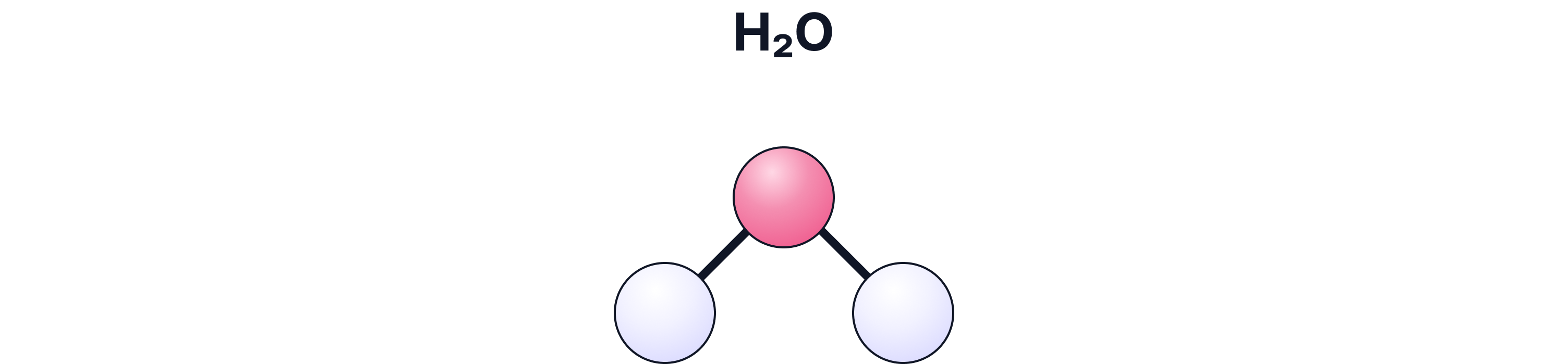
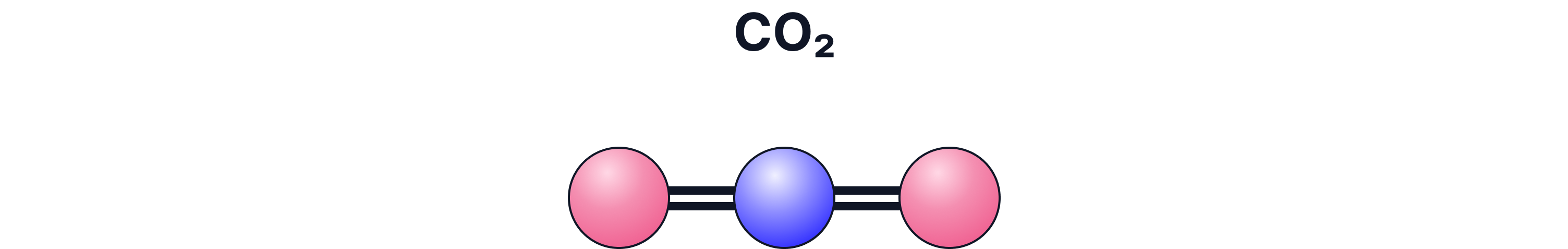
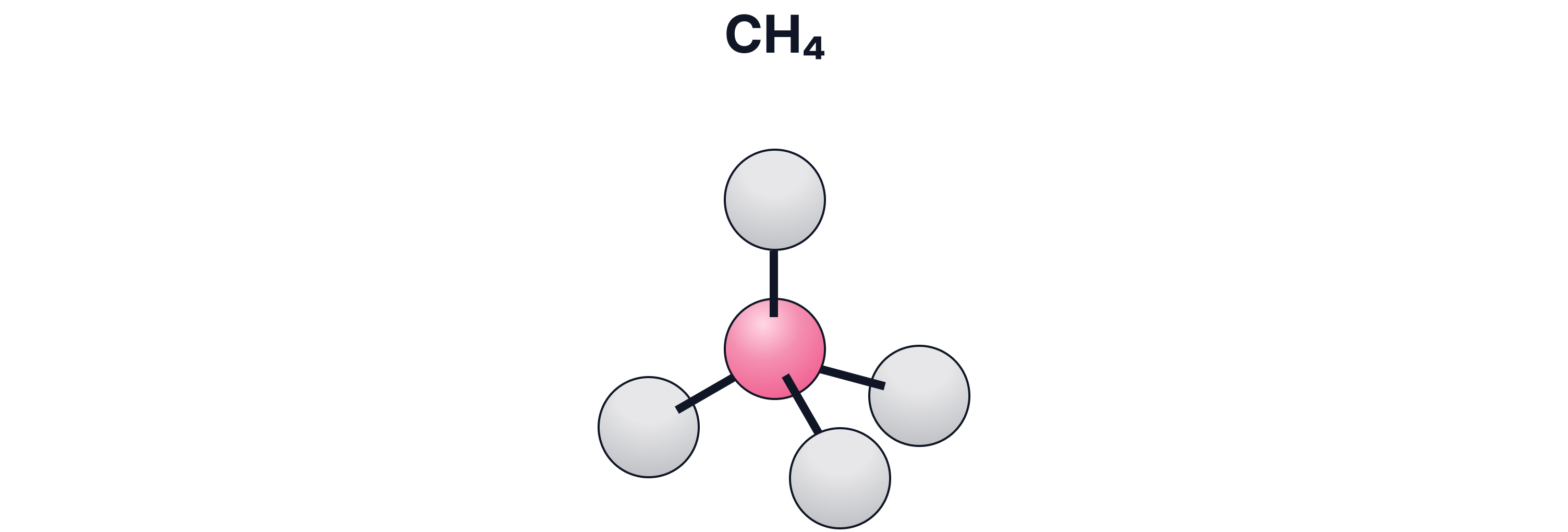
What it shows:
3-D structure and bond angles
Pros:
Helps visualise shape
Cons:
Bonds look like stiff sticks (not realistic)
Atoms not to scale
d) Space-Filling / 3-D Model
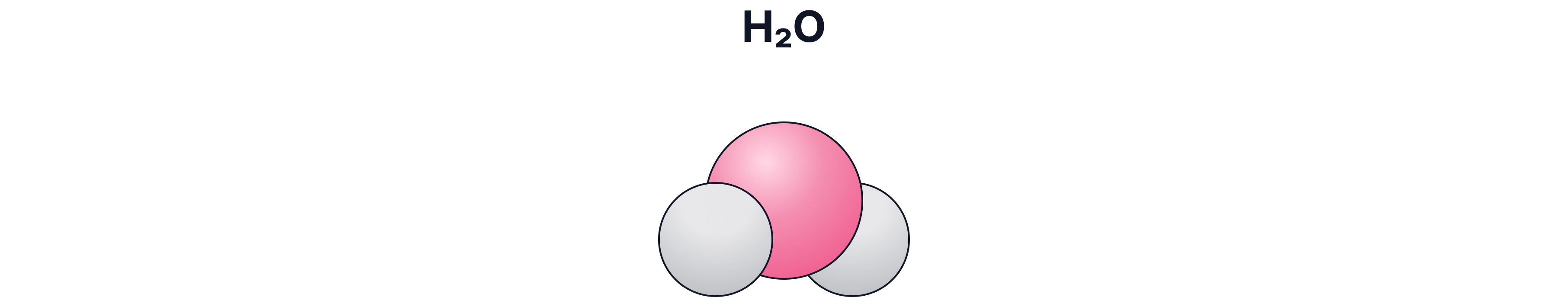
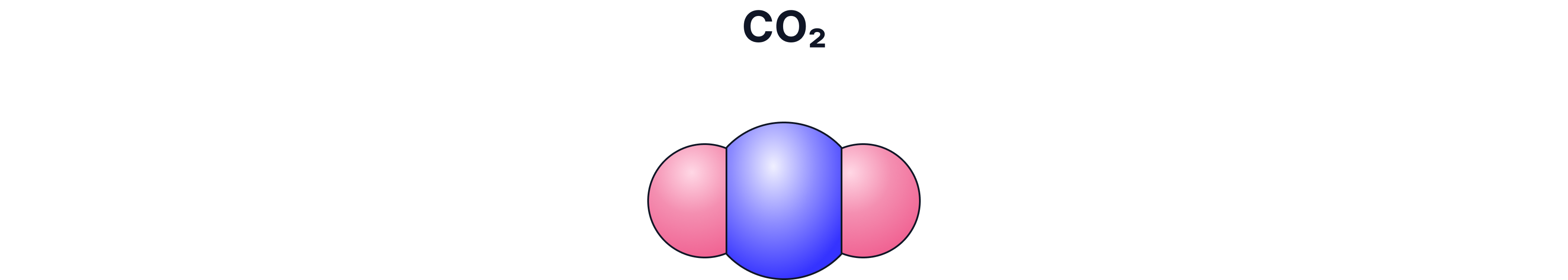
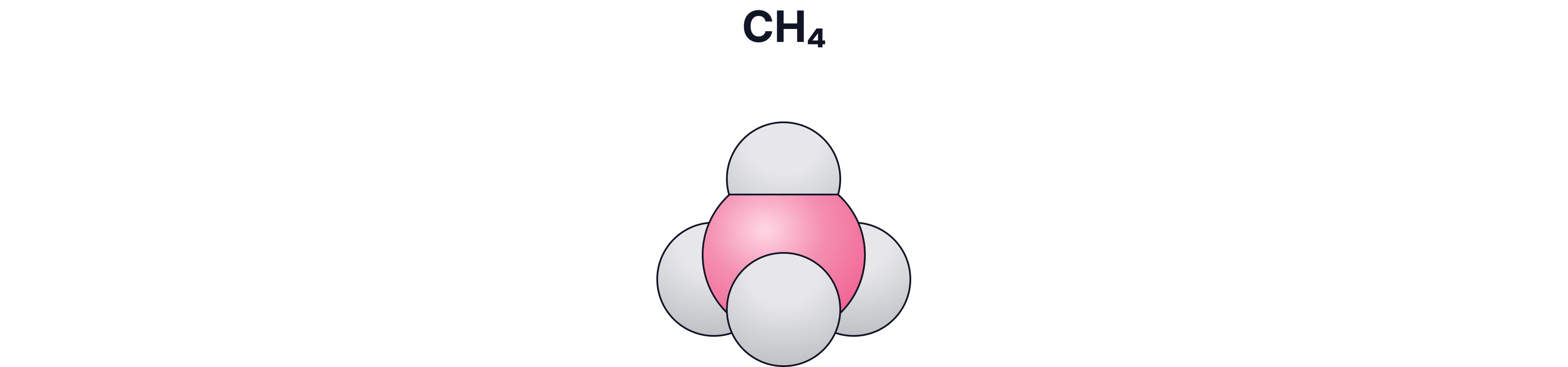
What it shows:
The real space taken up by atoms
Pros:
Good for showing size and packing
Cons:
Bonds are hidden
Model Comparison Table
Model | Best for… | Limitation |
Dot-and-cross | Electrons + lone pairs | Not 3-D |
Displayed | Counting bonds | Doesn’t show shape |
Ball-and-stick | 3-D shape and angles | Unrealistic sticks |
Space-filling | Atom size and packing | Bonds not visible |
Practice Questions
Recall
Q1. What is a covalent bond?
A covalent bond is the electrostatic force of attraction between a shared pair of electrons (of two non-metal atoms) and positive nuclei.
Q2. State whether each of the following substances is ionic or covalent:
a) CO₂
b) MgCl₂
c) N₂
d) H₂O
e) KBr
a) Covalent
b) Ionic
c) Covalent
d) Covalent
e) Ionic
Apply
Q3. Why do non-metal atoms share electrons instead of transferring them?
Non-metal atoms share electrons because they have similar attraction for electrons and both need to gain electrons to fill their outer shells. Sharing allows each atom to achieve a full outer shell and greater stability.
Q4. Oxygen has 6 electrons in its outer shell. Explain why an oxygen molecule (O₂) contains a double bond.
Oxygen needs two more electrons to complete its outer shell. Each oxygen atom shares two pairs of electrons, which forms a double covalent bond so both atoms achieve full shells.
Q5. Hydrogen chloride (HCl) contains a single covalent bond. Explain how this bond forms in terms of electrons.
Hydrogen needs one more electron and chlorine needs one more electron. They share one pair of electrons, and this shared pair counts for both atoms, giving each a full outer shell. This shared pair is the single covalent bond.
Q6. A student says: “Covalent bonding happens when electrons are transferred between atoms.” Explain why this statement is incorrect.
The statement is incorrect because electrons are shared in covalent bonding, not transferred. Electron transfer happens in ionic bonding, not covalent bonding.
Q7. For each molecule below, state how many covalent bonds it contains:
a) NH₃
b) CO₂
c) CH₄
d) N₂
a) NH₃ → 3 covalent bonds
b) CO₂ → 2 double bonds (4 shared pairs total)
c) CH₄ → 4 covalent bonds
d) N₂ → 1 triple bond (3 shared pairs)
Challenge
Q8. Describe one advantage and one limitation of using a displayed formula to represent covalent molecules.
Advantage: It clearly shows the number and placement of bonds.
Limitation: It does not show the shape or 3-D structure of the molecule.
Q9. Which bond is stronger and shorter — a single bond or a double bond? Explain why.
A double bond is stronger and shorter than a single bond because more electrons are shared, creating a stronger electrostatic attraction between the nuclei and the bonding electrons.