Properties of ionic compounds
Dr. Davinder Bhachu
Teacher

Contents
What Are Ionic Compounds?
When a metal reacts with a non-metal, electrons are transferred. This forms positive ions (cations) and negative ions (anions). These oppositely charged ions attract each other and build a huge 3-D structure called a giant ionic lattice.
The attraction between positive and negative ions is called a strong electrostatic force.
These forces act in all directions throughout the lattice.
A lattice contains billions of ions in a regular pattern.
Key idea: An ionic compound is not made of ion pairs. It is one continuous structure held together by strong electrostatic forces.
Models of Ionic Structures
Scientists use several types of diagrams to show ionic lattices. Each model is useful, but each has limitations.
a) Dot-and-Cross Diagrams
-
Show electron transfer and how ions form.
-
Good for understanding ionic bonding.
Limitation: They do not show the size of ions or the lattice structure.
b) 3-D Lattice Diagrams
-
Show a regular repeating pattern of ions.
-
Help students visualise the shape and structure of the lattice and see the relative sizes of the ions.
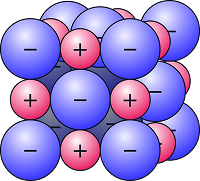
This is a 3D model of a small section of a sodium chloride crystal. The red spheres represent Na+ ions. The blue spheres represent Cl- ions. The oppositely charged ions are held together by strong electrostatic forces of attraction in all directions not just between one positive ion and one negative ion.
Limitation: Only a tiny part of the lattice can be shown. The model only lets us see the outer layer of the compound.
c) Ball-and-Stick Models
-
Show how each ion is connected and the arrangement in space.
-
Easy to see patterns, layers, and coordination.
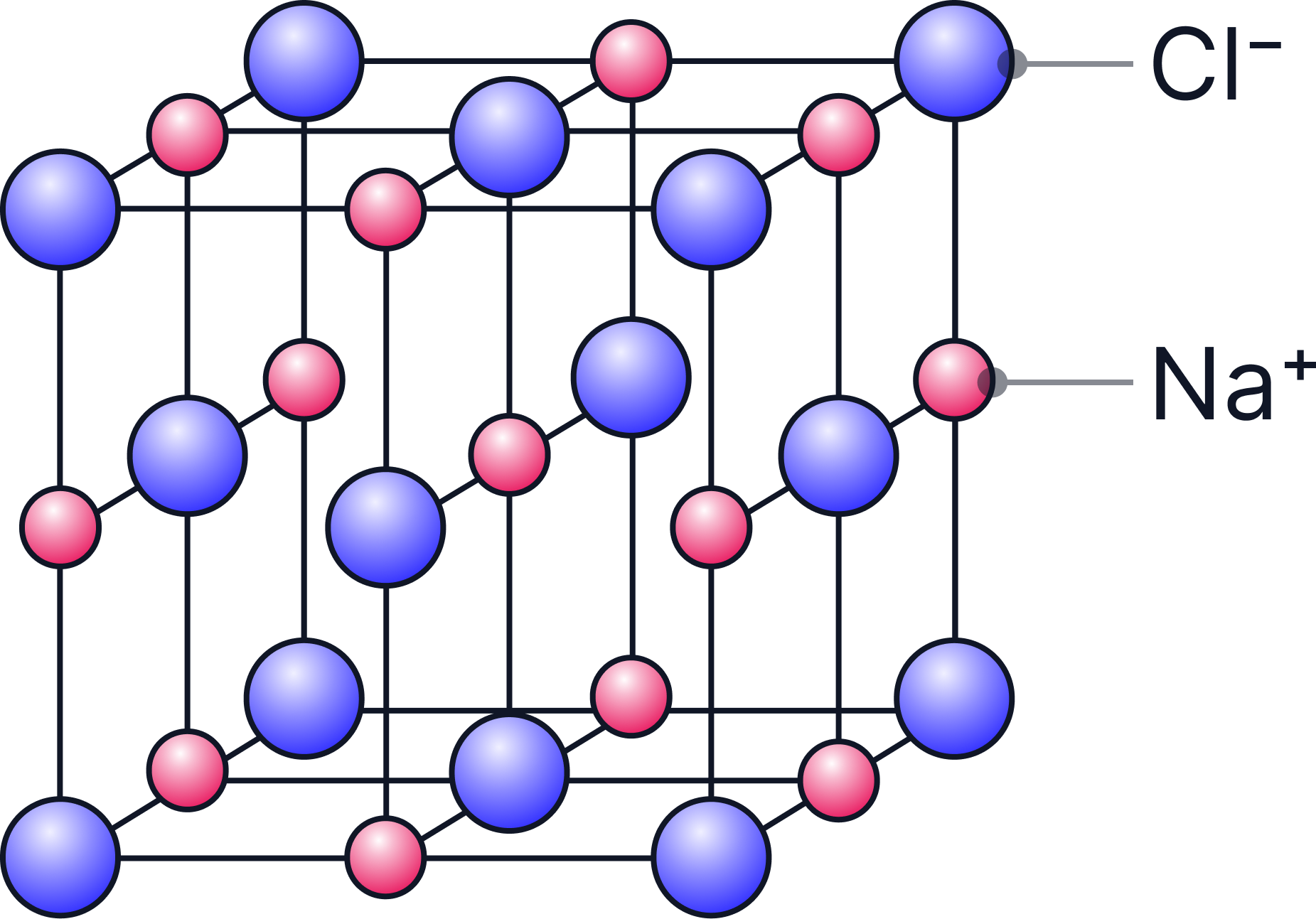
Limitations:
-
The “sticks” are not real bonds — ionic forces act in all directions, not as separate lines.
-
There are no gaps between the ions.
-
Ion sizes are often not to scale, which can be misleading.
When a metal reacts with a non-metal, electrons are transferred. This forms positive ions (cations) and negative ions (anions). These oppositely charged ions attract each other and build a huge 3-D structure called a giant ionic lattice.
-
The attraction between positive and negative ions is called a strong electrostatic force.
-
These forces act in all directions throughout the lattice.
-
A lattice contains billions of ions in a regular pattern.
Key idea: An ionic compound is not made of ion pairs. It is one continuous structure held together by strong electrostatic forces.
Working Out the Empirical Formula from a 3D model or ball and stick diagram
To find the formula of an ionic compound from a lattice diagram:
Steps:
1. Count how many of each type of ion is shown.
2. Write the ratio in the simplest whole numbers.
3. Write the metal first, then the non-metal.
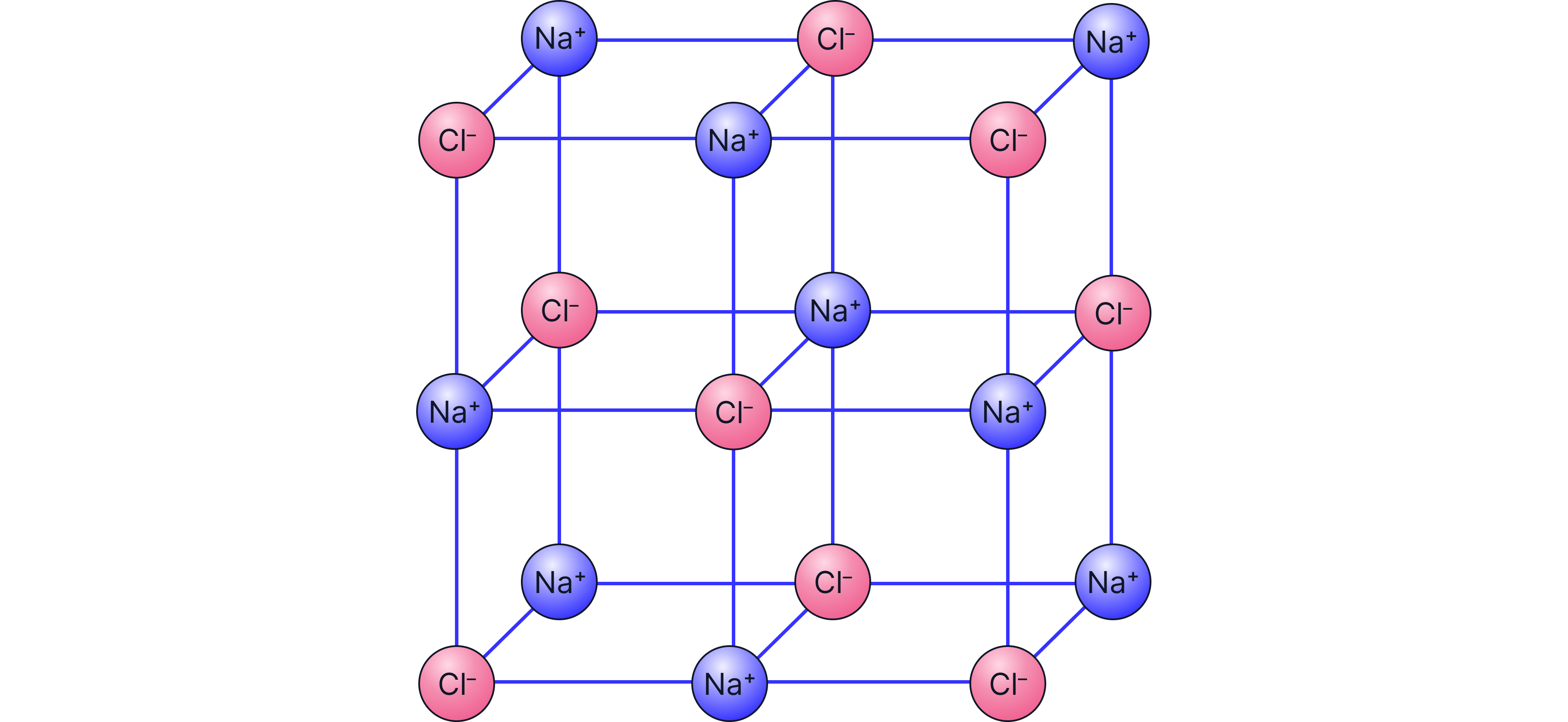
Example: NaCl
A lattice section contains:
-
9 Na⁺ ions
-
9 Cl⁻ ions
Ratio = 9:9
So the simplest ratio of ions is 1:1
Formula = NaCl
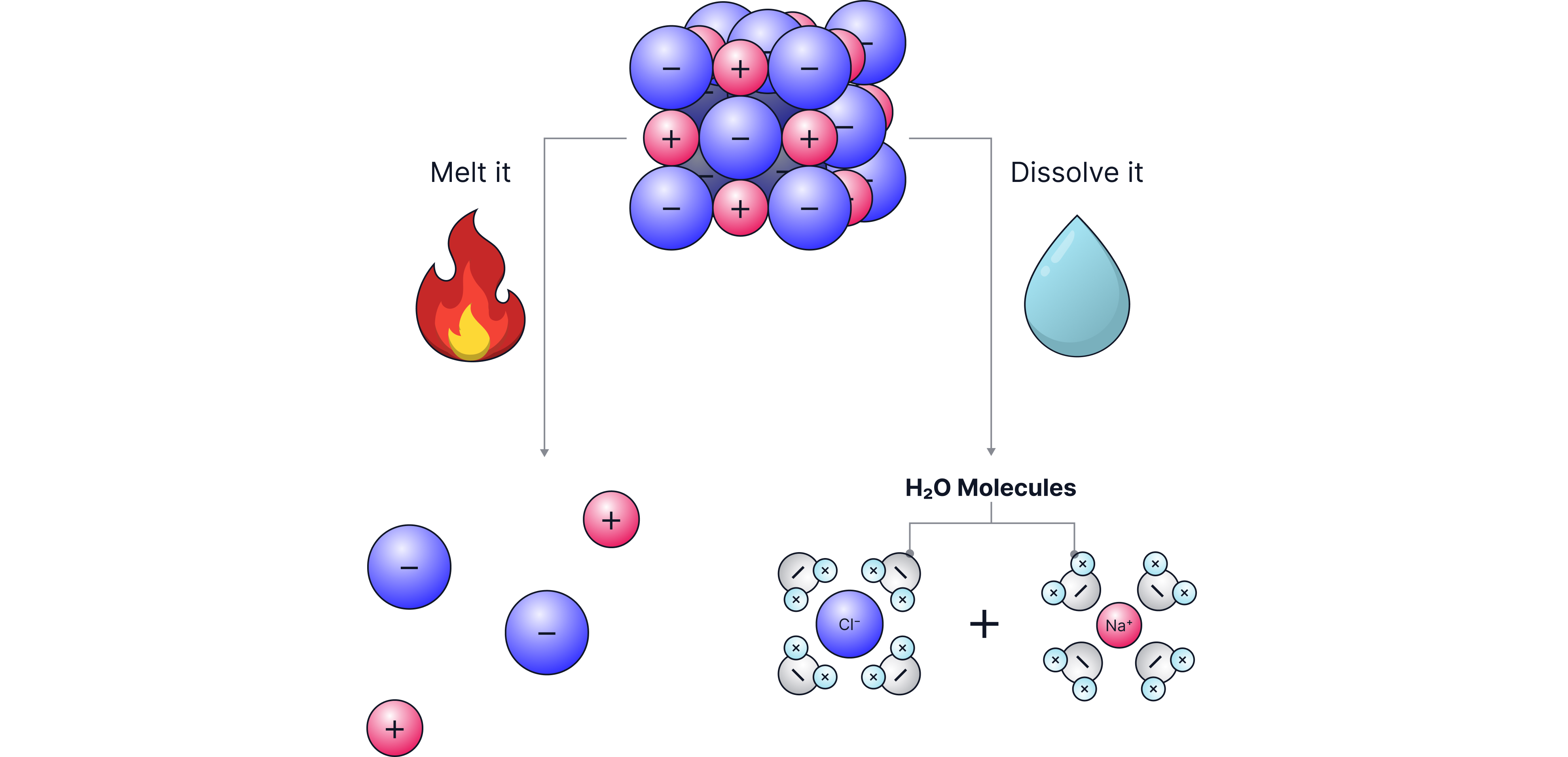
Properties of Ionic Compounds (Explained by Structure)
a) High Melting and Boiling Points
-
Ionic lattices contain strong electrostatic forces of attraction between the oppositely charged ions.
-
A lot of energy is needed to break all of these strong attractions between the ions.
-
Therefore, ionic compounds have high melting and boiling points.
-
We should also be able to predict whether NaCl or MgO will have the higher melting point, based purely on charges of ions at GCSE.
-
In MgO, the ions have higher charges (+2 and –2) compared to NaCl (+1 and –1).
-
This creates much stronger electrostatic attraction in the lattice so more energy is needed to separate the ions.
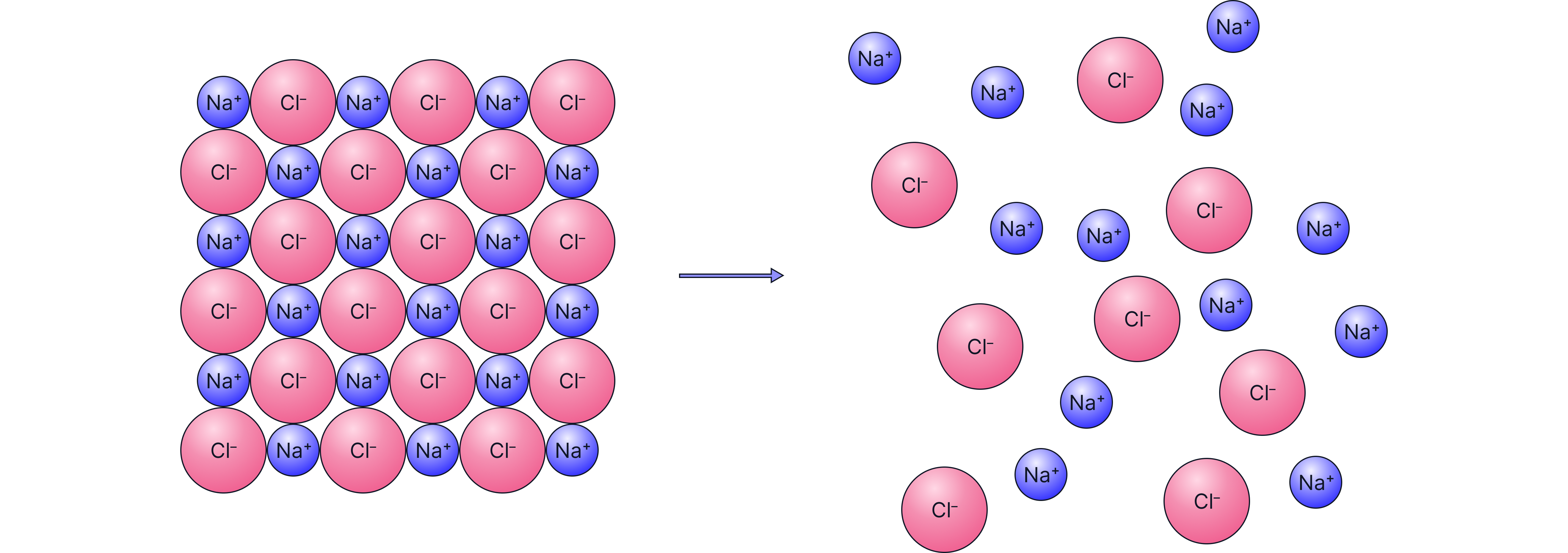
Caption: Melting requires overcoming enough electrostatic attraction between the sodium and chloride ions in this example, therefore allowing them to move.
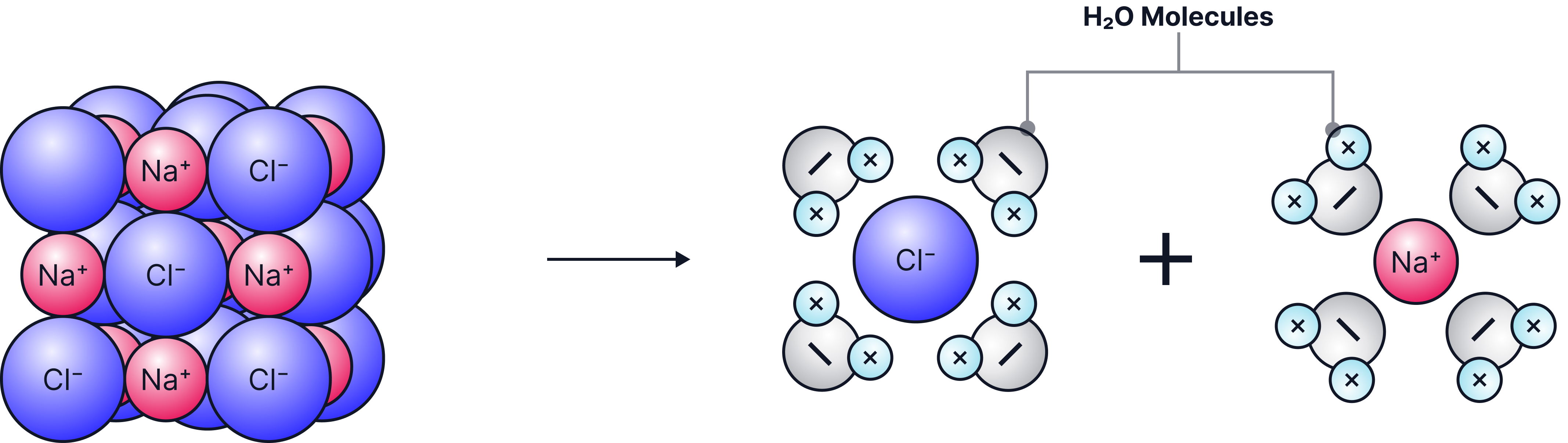
b) Solubility in Water
-
Water molecules are slightly charged.
-
They can pull ions away from the lattice.
-
Many ionic compounds dissolve in water.
c) Electrical Conductivity
-
Solid ionic compounds do not conduct electricity. The ions are fixed in place and cannot move.
-
Molten or dissolved ionic compounds do conduct — the ions are free to move.
-
Free ions are needed to carry charge as shown in the diagram.
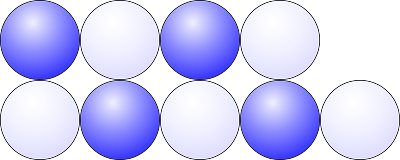
Ionic compounds are brittle
The diagram below shows oppositely charged ions arranged in a lattice structure. They are held in place by strong electrostatic forces of attraction between oppositely charged ions.
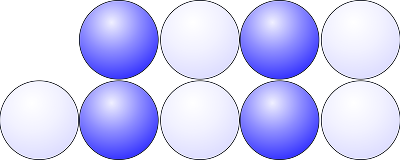
If we imagine applying a force to the second row so that the row slides along; we can see that ions of like charge are now arranged next to each other. This results in repulsion (as like charges repel). So much so that the lattice is shattered. Metals on the contrary are malleable whereas ionic compounds are not.
Practice Questions
Recall
What holds ions together in a giant lattice?
Strong electrostatic attraction between oppositely charged ions.
Explain why ionic compounds have high melting points.
Because the giant ionic lattice contains lots of strong electrostatic forces of attraction that need a lot of energy to break.
Apply
A lattice contains 2 Al³⁺ ions and 3 O²⁻ ions. What is the formula?
Formula = Al₂O₃ (charges balance 2×+3 and 3×–2).
Does solid copper(II) chloride conduct electricity? Explain your answer.
No, not when solid as the ions cannot move.
Challenge
Name one advantage and one limitation of a ball-and-stick model.
Advantage: shows arrangement clearly. Limitation: sticks are not real bonds and there are no gaps between the ions.
Why do many ionic compounds dissolve in water?
Water molecules are slightly charged, so they attract ions and pull them apart from the lattice.
Predict whether Na₂O will conduct electricity when molten and justify your answer.
Yes. When molten, the ions are free to move, so they can carry charge.