Names and formulae of ionic compounds
Dr. Davinder Bhachu
Teacher

Naming Ionic Compounds Made from Two Elements
When an ionic compound contains just two elements (a metal and a non-metal):
The metal’s name comes first (it stays the same).
The non-metal’s name comes second, but its ending changes to –ide.
Metal | Non-metal | Compound Name |
Sodium | Chlorine | Sodium chloride |
Magnesium | Oxygen | Magnesium oxide |
Potassium | Nitrogen | Potassium nitride |
Calcium | Sulfur | Calcium sulfide |
Aluminium | Bromine | Aluminium brom |
Examples – Determine the name from the formula
1. Formula: LiBr
Metal: lithium
Non-metal: bromine → change ending to –ide
Name: lithium bromide
2. Formula: MgO
Metal: magnesium
Non-metal: oxygen → –ide
Name: magnesium oxide
3. Formula: AlN
Metal: aluminium
Non-metal: nitrogen → –ide
Name: aluminium nitride
Naming Ionic Compounds with Three or More Elements
If a compound contains more than two elements, one of them is usually oxygen. These compounds often contain a compound/polyatomic ion (a small cluster of atoms that stay together and carry a charge). You have to remember these!
Group Ion | Formula | Ending | Example |
Nitrate | NO₃⁻ | –ate | Sodium nitrate (NaNO₃) |
Sulphate | SO₄²⁻ | –ate | Calcium sulphate (CaSO₄) |
Carbonate | CO₃²⁻ | –ate | Zinc carbonate (ZnCO₃) |
Hydroxide | OH⁻ | –ide | Sodium hydroxide (NaOH) |
Ammonium | NH₄⁺ | (acts as metal) | Ammonium chloride (NH₄Cl) |
Naming Compounds with Transition Metals
Some metals (like iron, copper, lead) can form more than one type of ion (e.g. Fe²⁺ and Fe³⁺). We show which ion is used with Roman numerals in the name.
Metal Ion | Example Compound | Compound Name |
Fe²⁺ | FeO | Iron(II) oxide |
Fe³⁺ | Fe₂O₃ | Iron(III) oxide |
Cu⁺ | CuCl | Copper(I) chloride |
Cu²⁺ | CuCl₂ | Copper(II) chloride |
Trick to remember:
Roman numerals = charge on the metal ion (not how many atoms).
So Iron(III) = Fe³⁺, Copper(II) = Cu²⁺, etc.
Writing Ionic Formulae
Recap: What You Already Know
You now know how to name ionic compounds by combining the names of positive (metal) and negative (non-metal) ions.
Now we’ll learn how to write their chemical formulae — showing exactly how many of each ion are present in the compound.
Ionic Compounds Are Always Charge Neutral
Every ionic compound must have no overall charge. That means the total positive charge from the metal ions must balance the total negative charge from the non-metal ions.
Think of it like a seesaw — both sides must balance.
Example 1 – Magnesium Chloride
Mg forms Mg²⁺ ions
Cl forms Cl⁻ ions
To balance the +2 charge on magnesium, you need two chloride ions (each –1).
Formula: MgCl₂
Check:
(+2) + (2 × –1) = 0 ✔️
Method 1: Lowest Common Multiple (LCM) Method
This method uses the lowest common multiple of the ion charges.
Steps:
1. Write the ions with their charges.
2. Find the lowest common multiple (LCM) of the charges.
3. Work out how many of each ion you need so the total charge = 0.
4. Write the formula without charges.
Example 2 – Calcium Fluoride
Ca²⁺ and F⁻
LCM of 2 and 1 is 2
→ Need 2 × F⁻ to balance 1 × Ca²⁺
Formula: CaF₂
Example 3 – Aluminium Oxide
Al³⁺ and O²⁻
LCM of 3 and 2 is 6
→ Need 2 × Al³⁺ (+6 total) and 3 × O²⁻ (–6 total)
Formula: Al₂O₃
Example 4 – Iron(III) Chloride
Fe³⁺ and Cl⁻
LCM of 3 and 1 is 3
→ Need 1 × Fe³⁺ and 3 × Cl⁻
Formula: FeCl₃
Method 2: The “Drop and Swap” Shortcut
This is a quick method to get the same result as the LCM method.
Steps:
1. Write the ion symbols with their charges.
2. Drop the number of each charge.
3. Swap them to become the number of the other ion.
4. Remove the charges and simplify if needed.
Example 5 – Magnesium Nitrate
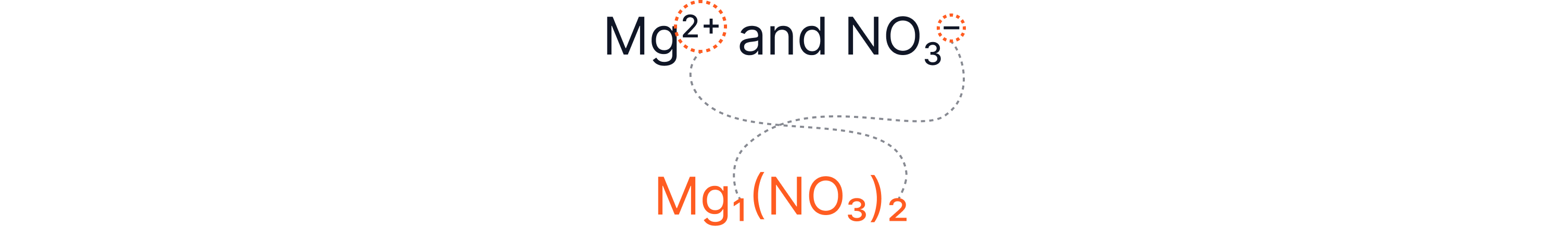
Brackets are needed around the nitrate ion because there’s more than one of that group ion.
Mg(NO₃)₂
Example 6 – Aluminium Sulphate
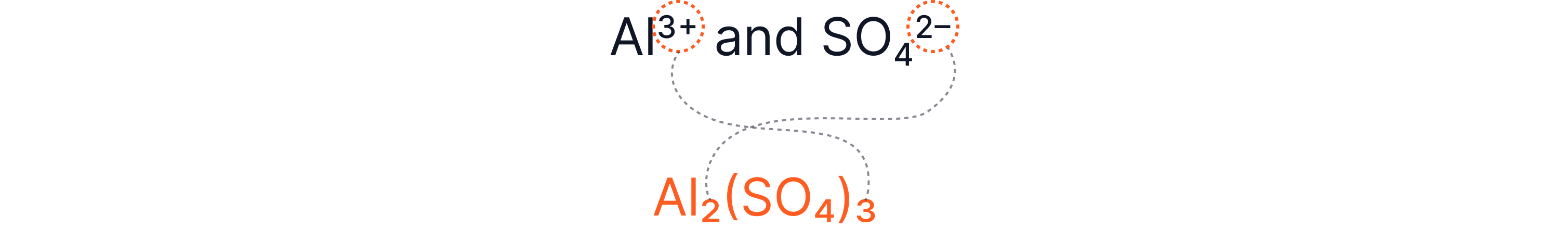
Al₂(SO₄)₃
Example 7 – Ammonium Phosphate
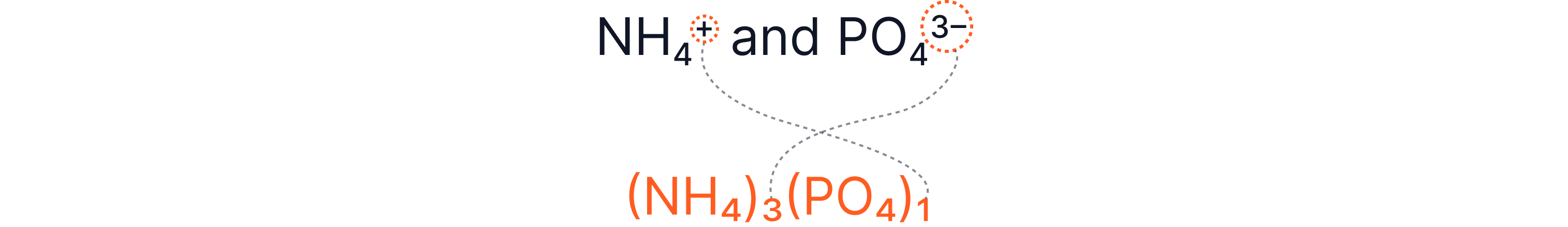
So (NH₄)₃PO₄
When the Drop-and-Swap Method Doesn’t Change Anything
Sometimes, the charges on the ions already balance perfectly 1:1 — so when you “drop and swap,” you might end up writing unnecessary numbers.
Example – Magnesium Oxide
Ions:
Mg²⁺ O²⁻
If you drop and swap:
This give
That looks wrong — because Mg₂O₂ simplifies to MgO.
You don’t need two of each; one magnesium ion and one oxide ion already balance the charges.
Correct formula: MgO
Check:
(+2) + (–2) = 0 ✔️
Practice Questions
1️. Name these ionic compounds:
a) Na₂S
Answer:
a) Sodium sulphide
b) CaCl₂
b) Calcium chloride
c) K₂O
c) Potassium oxide
d) AlBr₃
d) Aluminium bromide
2️. Now name these compounds that contain oxygen:
e) Na₂SO₄
e) Sodium sulphate
f) Cu(NO₃)₂
f) Copper(II) nitrate
g) ZnCO₃
g) Zinc carbonate
h) NH₄OH
h) Ammonium hydroxide
Challenge (name these):
i) Fe₂O₃
i) Iron(III) oxide
j) Pb(NO₃)₂
j) Lead(II) nitrate
k) CuCl
k) Copper(I) chloride
l) FeSO₄
l) Iron(II) sulphate