Ions and Ionic Bonding
Dr. Davinder Bhachu
Teacher

Contents
What are Ions?
Ions are charged particles made when an atom gains or loses electrons.
If an atom loses electrons, it becomes positively charged → called a cation. There are more protons than electrons
If an atom gains electrons, it becomes negatively charged → called an anion. There are more electrons than protons
The charge on the ion tells you how many electrons were lost or gained.
Example: If an atom forms a 3+ ion, it has lost 3 electrons
i.e. there are 3 more protons than electrons. The number of protons and electrons is no longer equal.
Example: If an atom forms a 3- ion, it has gained 3 electrons
i.e. there are 3 more electrons than protons. The number of protons and electrons is no longer equal.
Example 1 – A Metal Atom Forming a Positive Ion (Cation)
A magnesium atom has 12 protons and 12 electrons.
Protons = +12 Electrons = –12 Total charge = 0
If it loses 2 electrons, it now has 12 protons and 10 electrons.
Protons = +12 Electrons = –10 Total = +2
So the ion has a 2+ charge and is written as Mg²⁺.
Example 2 – A Non-Metal Atom Forming a Negative Ion (Anion)
A fluorine atom has 9 protons and 9 electrons.
If it gains one electron, it now has 9 protons and 10 electrons.
Protons = +9 Electrons = –10 Total = –1
So the ion has a 1- charge and is written as F⁻.
Example 3 – Working Backwards from Charge to Electrons
An aluminium ion (Al³⁺) has a charge of +3.
We know all atoms of aluminium have 13 protons.
Question: How many electrons are in Al³⁺?
Protons = +13
Overall charge = +3
To solve this:
Electron number = Proton number - charge
_________ = 13 - (+3)
So, Al³⁺ has 10 electrons (it lost 3 from its neutral atom).
Why Do Atoms Form Ions?
Atoms form ions to get a full outer shell of electrons (a stable arrangement like the noble gases in Group 0.)
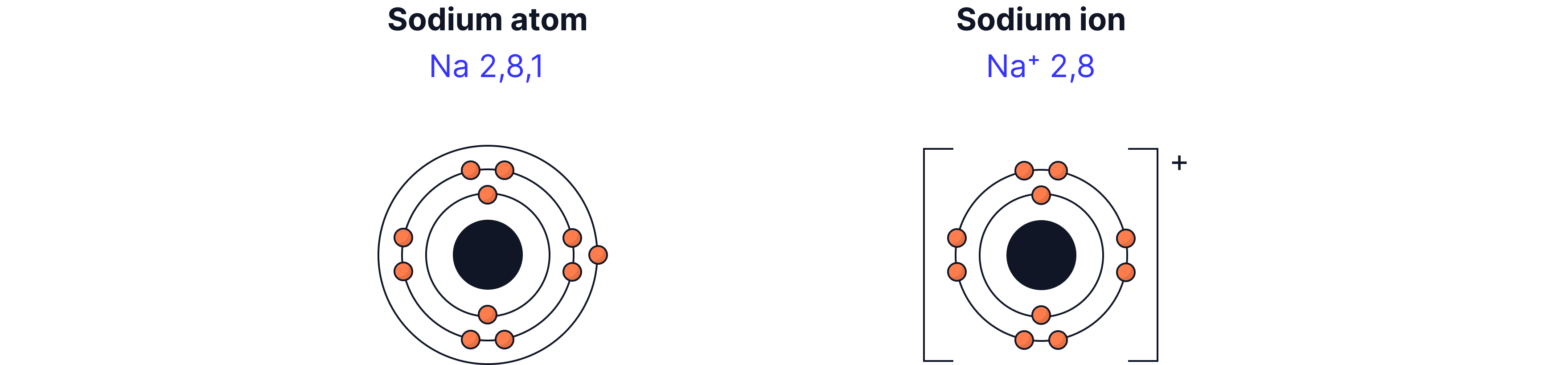
For example, a neutral sodium atom has 11 protons and 11 electrons (with a 2, 8, 1 electron configuration). To have a full outer shell there are two possibilities. We remove the outer most electron or we add seven electrons to the outer most shell. It is clearly easier to remove the outer most (valence electron) electron in this case.
The result is a sodium ion. A sodium ion has 11 protons but 10 electrons. These are not equal and therefore there is an overall charge of 1+.
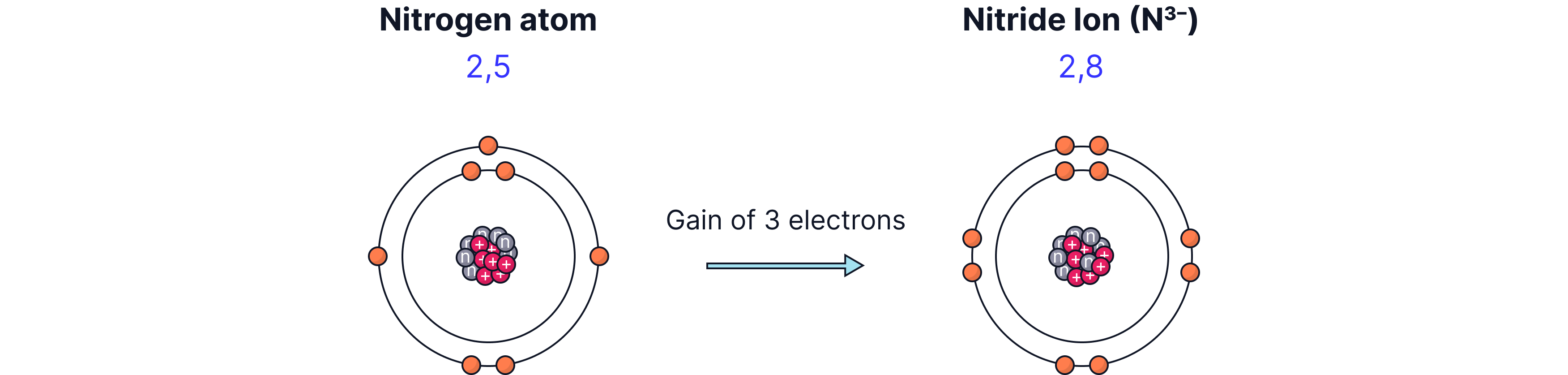
For example, a neutral nitrogen atom has 7 protons and 7 electrons (with a 2, 5 electron configuration). To have a full outer shell there are two possibilities. We remove the five outer most electron or we add three electrons to the outer most shell. It is clearly easier to add three electrons to the outer most (valence electron) electron in this case. The result is a nitride ion. A nitride ion has 7 protons but 10 electrons. These are not equal and therefore there is an overall charge of 3-.
Atoms will always gain or lose the smallest number of electrons needed to have an electron configuration like their nearest noble gas (full outer shell).
Charge on Ion and Group number
We don’t have to remember the charge of ions for elements. We can look at the periodic table. When metals react, they lose electrons forming cations. The charge on the cation is related to the group in which the metal is found on the periodic table. When non-metals form ions, they gain electrons, forming anions. The charge on the anion is given by the:
Charge = Group number - 8.

Metals (Groups 1, 2 and 3):
Group 1 lose 1 electron to get a full outer shell forming 1+ ions.
Group 2 lose 2 electrons to get a full outer shell forming 2+ ions.
Group 3 lose 3 electrons to get a full outer shell forming 3+ ions.
Non-Metals (Groups 5, 6 and 7):
Group 5 gain 3 electrons to get a full outer shell forming 3- ions.
Group 6 gain 2 electrons to get a full outer shell forming 2- ions.
Group 7 gain 1 electron to get a full outer shell forming 1- ions.
Group 7
Group | Action | Ion Charge | Example |
1 | Lose 1 electron | +1 | Li⁺, Na⁺, K⁺ |
2 | Lose 2 electrons | +2 | Mg²⁺, Ca²⁺ |
3 | Lose 3 electrons | +3 | Al³⁺ |
6 | Gain 2 electrons | –2 | O²⁻, S²⁻ |
7 | Gain 1 electron | –1 | F⁻, Cl⁻ |
Ionic bonding
When a metal reacts with a non-metal, electrons are transferred from the metal to the non-metal.
The metal loses electrons and becomes a positive ion (cation).
The non-metal gains electrons and becomes a negative ion (anion).
The oppositely charged ions are held together by a strong electrostatic attraction.
This is called ionic bonding.
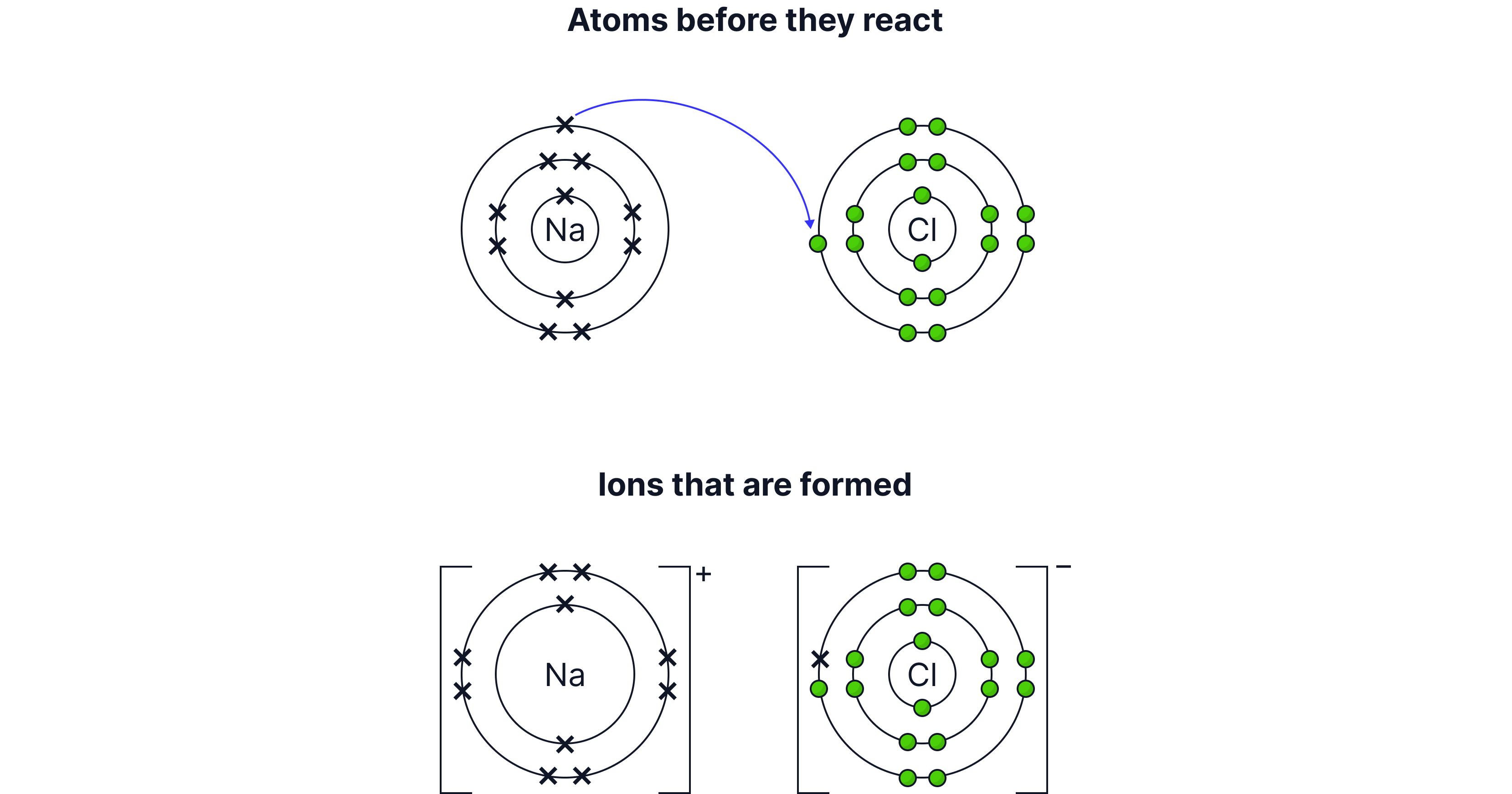
Representing ionic bonding with dot and cross diagrams:
Worked Example 1: Sodium chloride
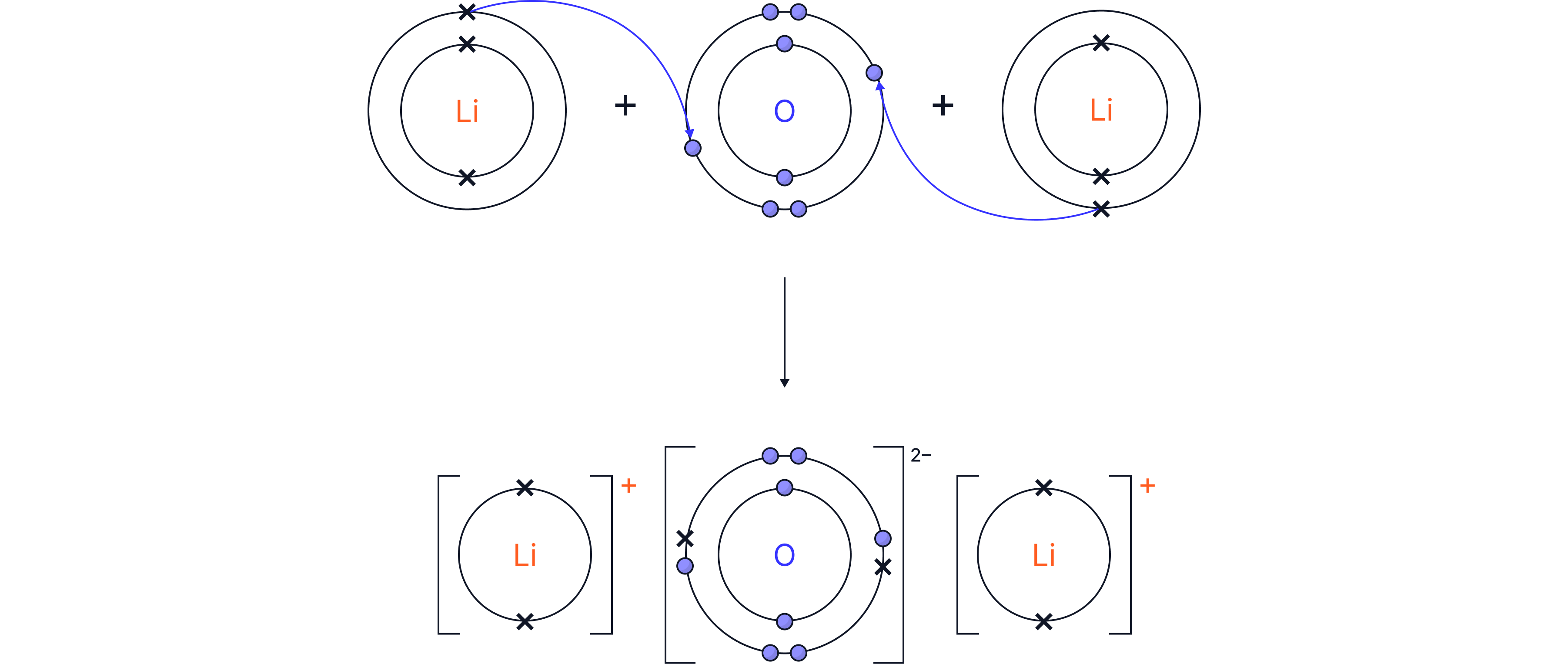
Worked example 2: Lithium oxide
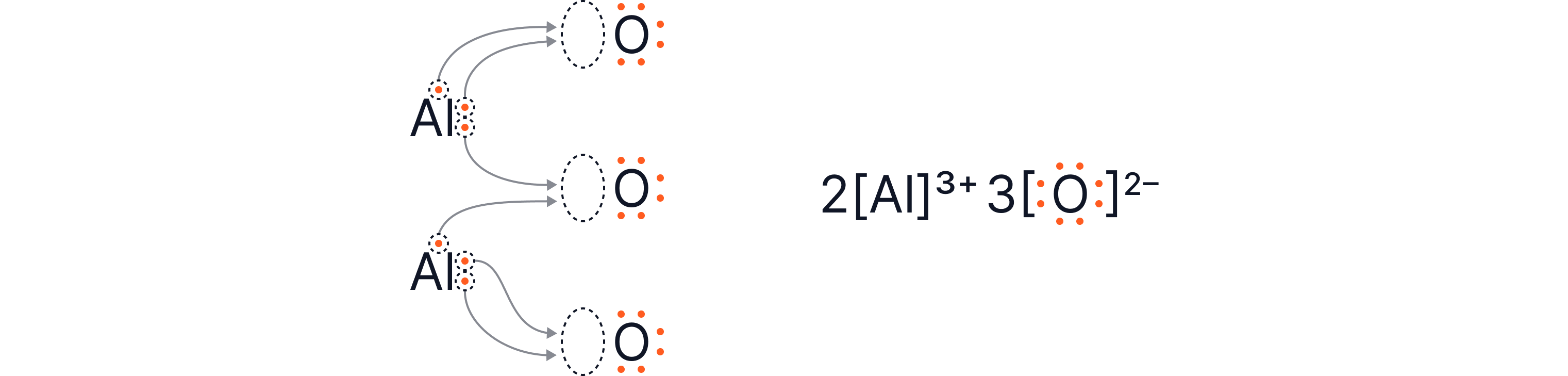
Worked Example 3: Aluminium oxide
Question 1 – Lithium Sulfide
1. Each lithium atom: 2,1 → loses 1 electron → Li⁺
2. Sulfur atom: 2,8,6 → gains 2 electrons → S²⁻
3. Need 2 Li⁺ ions to balance one S²⁻
→ Formula = Li₂S
Question 2 – Magnesium Nitride
Mg: 2,8,2 → loses 2 electrons → Mg²⁺
N: 2,5 → gains 3 electrons → N³⁻
3 × Mg²⁺ (total +6) and 2 × N³⁻ (total –6)
→ Formula = Mg₃N₂
Practice Questions
Recall
a) What is an ion?
Answer:
a) An ion is a charged particle formed when an atom gains or loses electrons.
b) How do metal atoms form ions?
Answer:
b) Metal atoms lose electrons to form positive ions (cations).
c) How do non-metals form ions?
Answer:
c) Non-metals gain electrons to form negative ions (anions).
Apply Your Knowledge
d) A magnesium ion has 10 electrons. How many did the atom have originally?
Answer:
d) Mg²⁺ has 10 electrons; original Mg atom had 12, so it lost 2.
e) Write the electron transfer equation for the formation of lithium iodide.
Answer:
e) Li → Li⁺ + e⁻ and I + e⁻ → I⁻; Li⁺ + I⁻ → LiI
f) Draw a dot-and-cross diagram for aluminium fluoride (AlF₃).
Answer:
f) Each Al loses 3 electrons to 3 fluorine atoms; final ions are Al³⁺ and 3F⁻.
Challenge
g) Explain, using ions, why ionic compounds are always electrically neutral.
Answer:
g) The total positive and negative charges balance, giving no overall charge.
h) Predict the ions and formula for a compound formed between calcium and oxygen.
Answer:
h) Ca²⁺ and O²⁻ → CaO.