Calculating rates of reactions
Dr. Davinder Bhachu
Teacher

Contents
What Do We Mean by “Rate of Reaction”?
The rate of a reaction tells us how quickly reactants are turned into products.
We can measure this change in different ways, depending on the reaction type.
Ways to Measure the Rate
What You Measure | How It’s Done | Example | Units |
(1) Mass loss | If a gas is released, the mass of the container decreases. | Calcium carbonate + acid → CO₂ | g/s |
(2) Gas volume | Use a gas syringe or an inverted measuring cylinder. | Magnesium + acid → Hydrogen | cm³/s |
(3) Colour change / cloudiness | Measure how long it takes for a mark to disappear through a mixture. | Sodium thiosulfate + acid | s⁻¹ (time taken used for comparison) |
Mass Loss Method
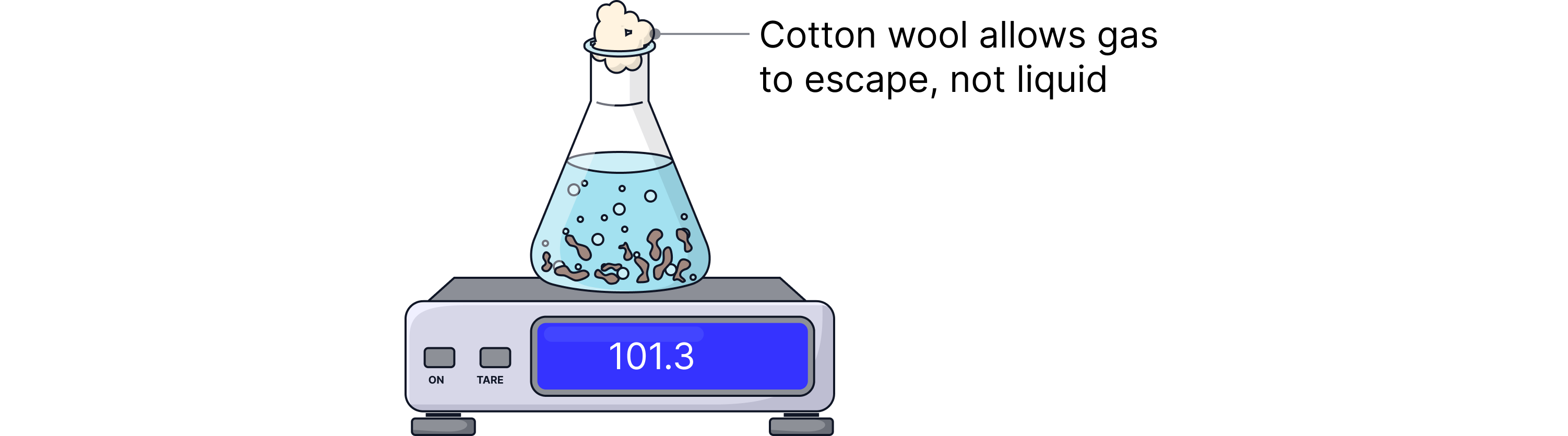
1. A conical flask containing magnesium and hydrochloric acid is placed on a digital balance.
2. As the reaction proceeds, hydrogen gas escapes from the flask.
3. The mass of the flask decreases over time due to the loss of gas.
4. The mass is recorded at regular time intervals (e.g. every 10 seconds).
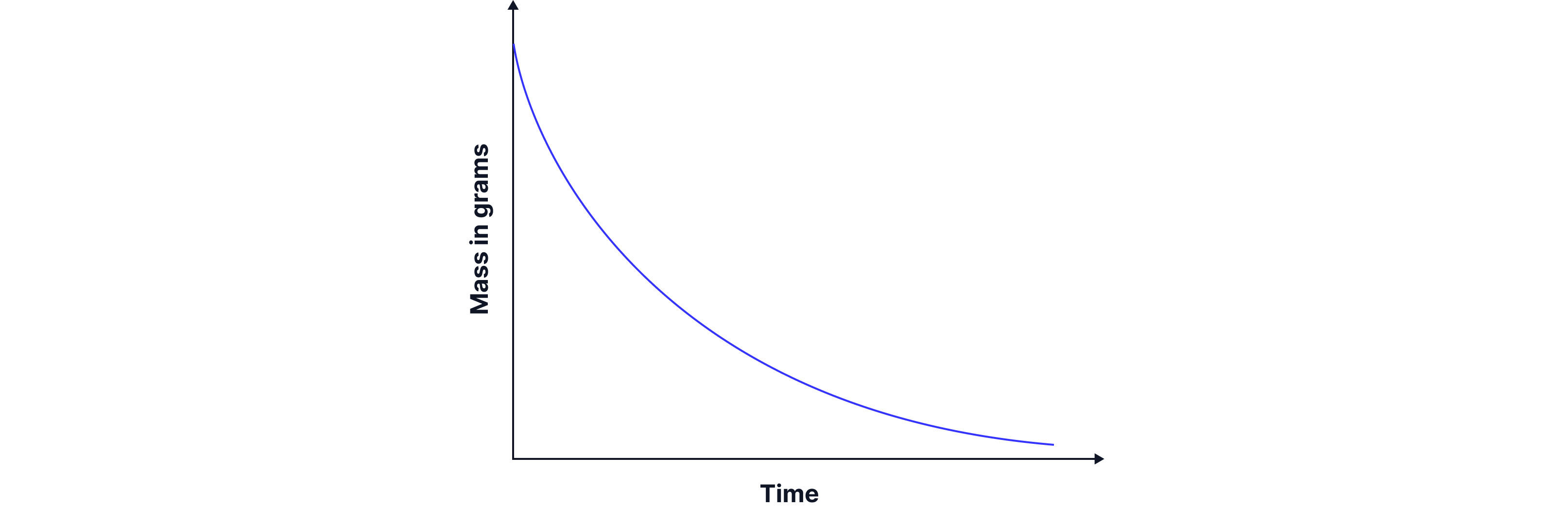
Typical graph of mass loss (in grams) with respect to time and key features
How It Measures Rate
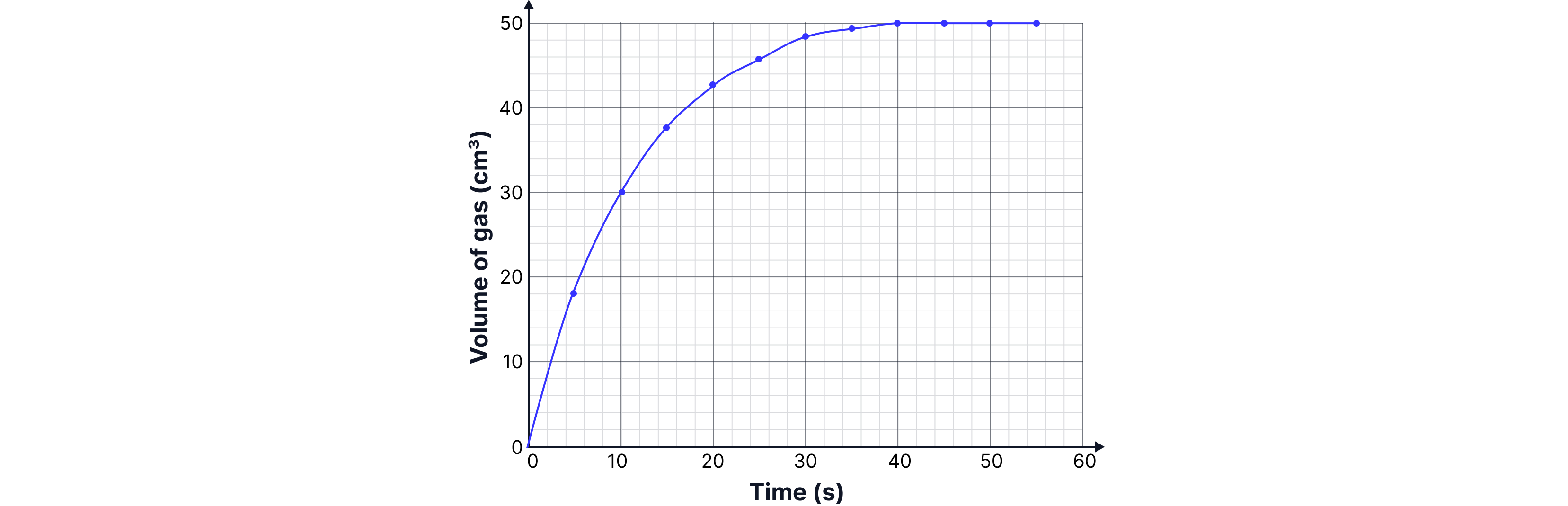
The faster the mass decreases, the faster the reaction is occurring.
A graph of mass lost vs time can be plotted.
The gradient of the curve at any point gives the rate of reaction.
The initial gradient (at t = 0) represents the initial rate.
Gas Volume Methods
Gas syringe method
How it works:
A gas syringe is connected to a sealed reaction vessel.
As gas forms, it pushes the plunger outward.
Volume is recorded at regular time intervals.
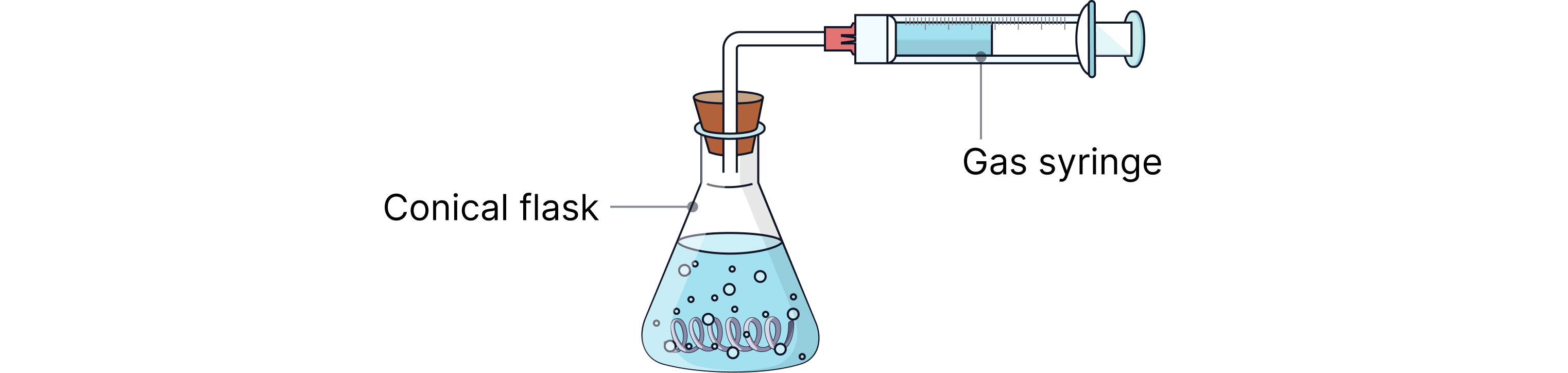
Suitable for:
Most gases
Accurate volume measurement (up to 100 cm³ or more)
Ideal for:
Insoluble gases like hydrogen, oxygen, or carbon dioxide.
Rate determined by:
Plotting volume vs time, then calculating gradient.
Displacement of water
How it works:
Gas is directed into an inverted measuring cylinder filled with water and placed in a water trough.
The gas displaces the water downward as it collects at the top of the cylinder.
The volume of gas collected can be measured using the cylinder’s graduations.
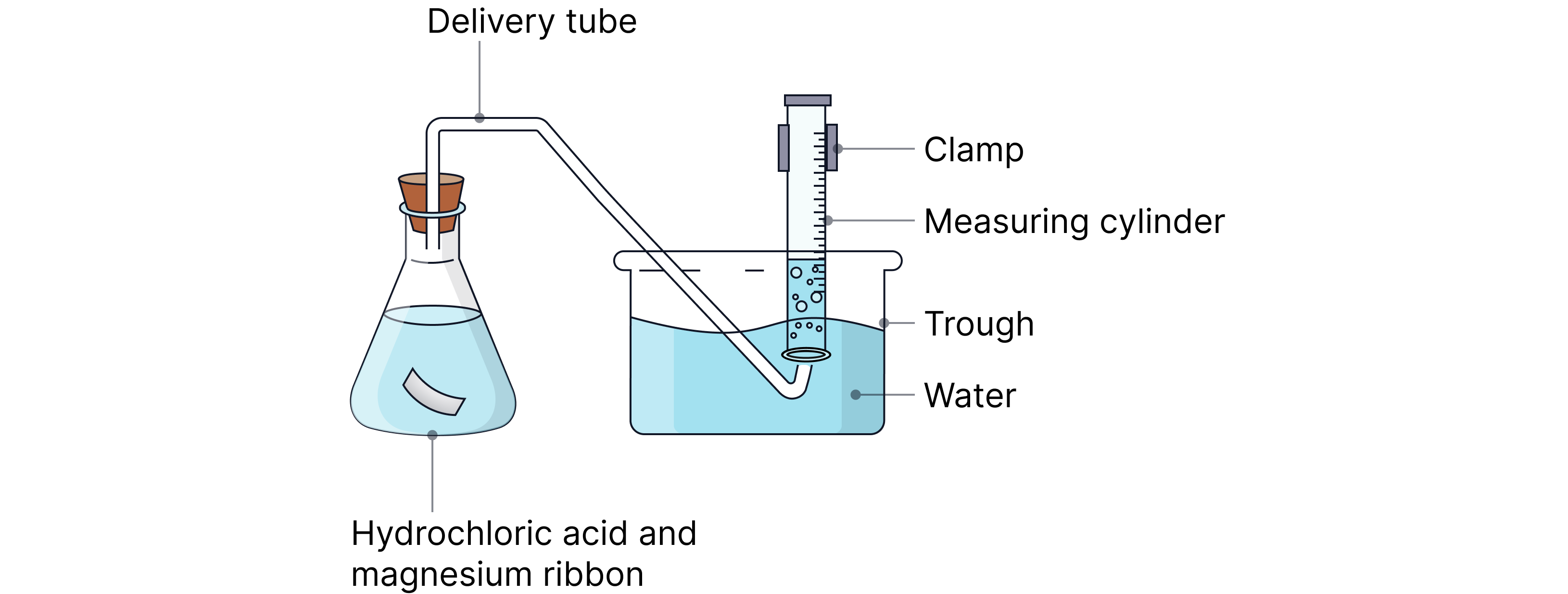
Examples:
Chlorine (Cl₂)
Carbon dioxide (CO₂)
Hydrogen (H₂)
Hydrogen chloride (HCl)
Rate determined by:
Tracking volume collected vs time, then finding gradient.
e.g
This reaction produces hydrogen gas, making it ideal for monitoring rate by measuring gas volume over time.
Typical graph of volume of gas produced with respect to time and key features
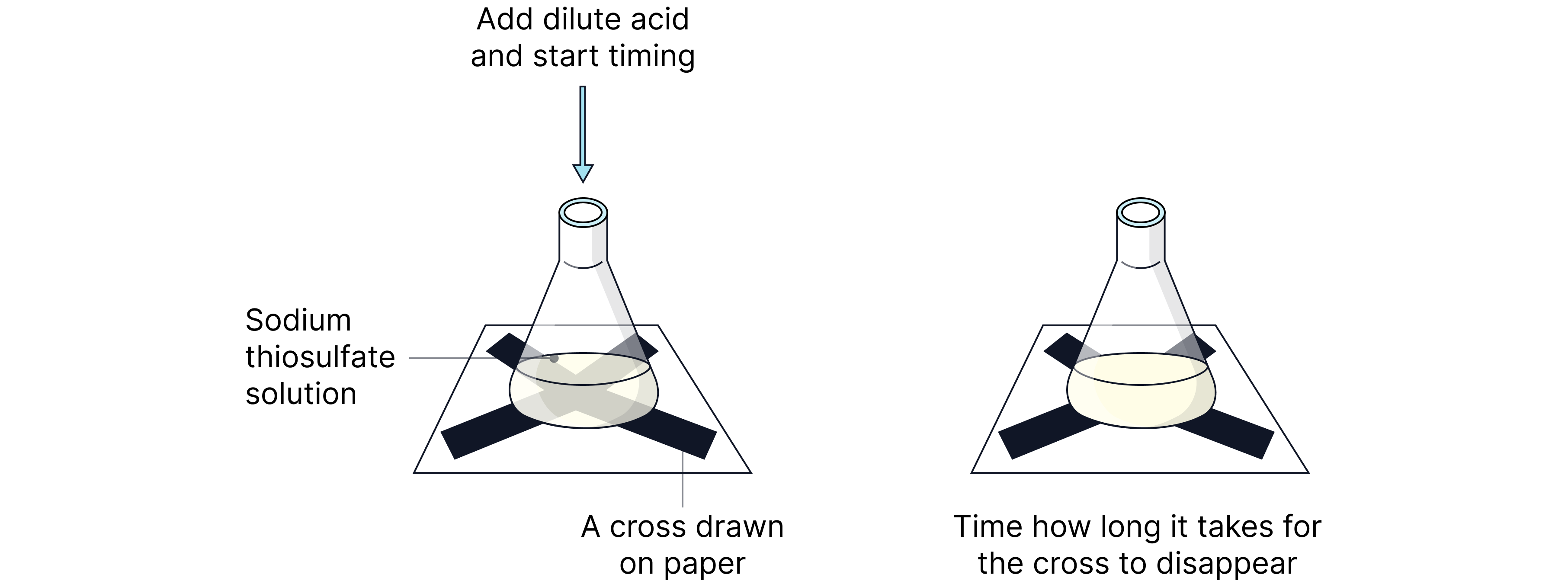
Disappearing Cross
A classic method to compare rates without using concentration directly:
Reaction:
In this reaction:
Sulfur forms as a cloudy precipitate.
A cross drawn beneath the reaction vessel becomes obscured over time.
The time taken for the cross to disappear is recorded.
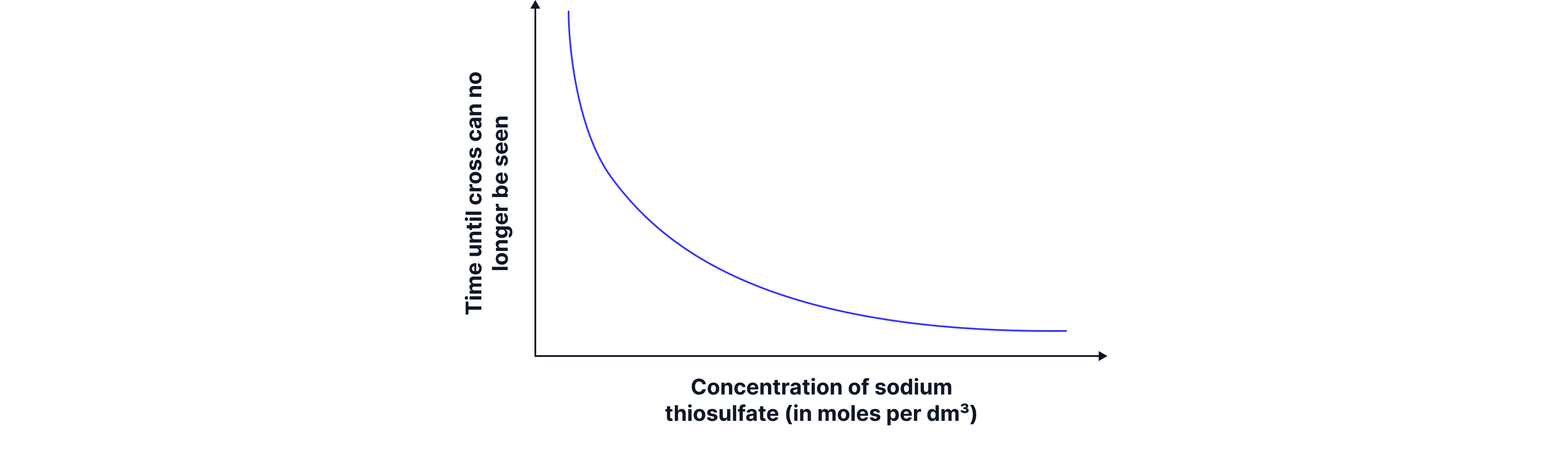
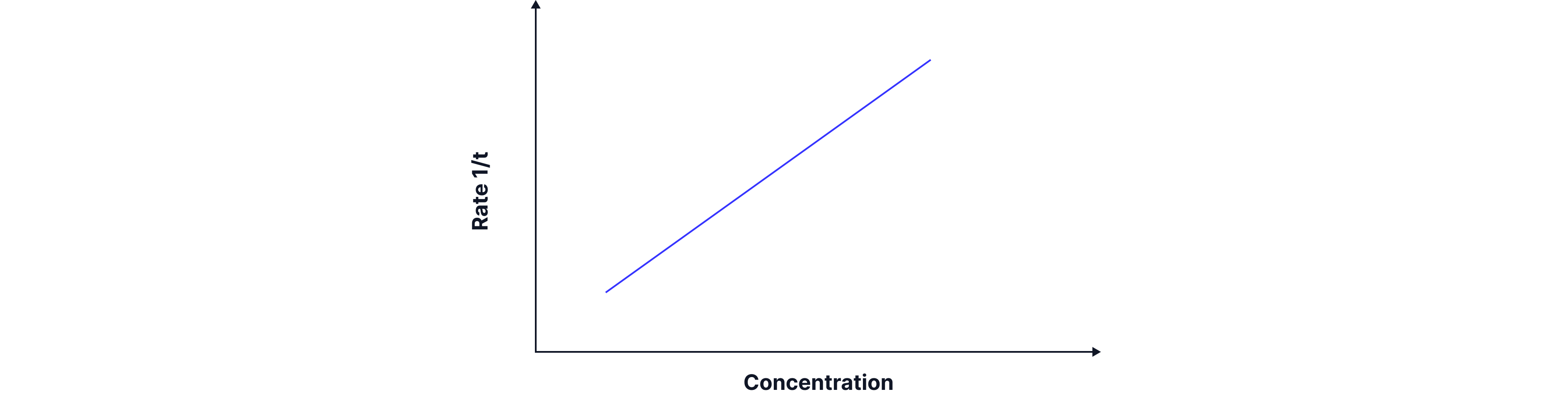
Typical graphs from the disappearing cross experiment:
The graph on the below shows the time taken for the cross to disappear. When sodium thiosulphate of higher concentration is used then less time is taken for the cross to disappear, so the rate of reaction is higher. We can also see the corresponding rate (1/t) vs concentration graph.
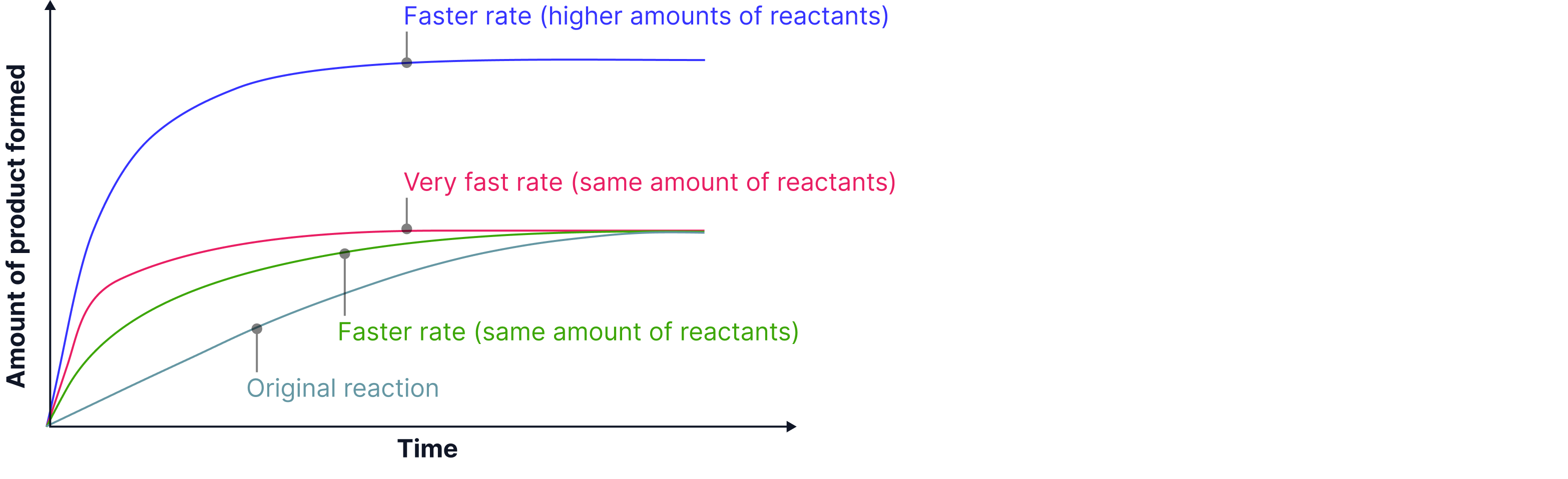
Comparing Rate Graphs
Once you’ve plotted data for how much reactant is used up or product is formed, you can compare different reaction rates on the same axes.
Key idea: The steeper the curve, the faster the reaction.
Each curve shows how conditions like temperature, concentration, or surface area affect the reaction rate.
Step-by-Step Comparison
1. Look at the slopes:
The steepest line = the fastest initial rate (reaction starts quickly).
Lines that start steep but flatten quickly show reactions finishing faster
2. Check where they level off:
If all lines finish at the same height, the total amount of product formed is the same.
A line finishing higher means more product was formed, usually because more reactants were present.
3. Interpret the shape:
Curves that rise steeply then flatten show that the reaction slowed down as reactants were used up.
A horizontal line means the reaction has stopped completely.
Example: Comparing Reactions
A student reacted magnesium with hydrochloric acid at different temperatures:
30°C → slow reaction (gentle slope)
40°C → faster reaction (steeper curve)
50°C → even faster reaction
50°C with double the magnesium → fastest and highest curve (more product overall)
Increasing the temperature or reactant quantity increases the reaction rate and, in some cases, total yield.
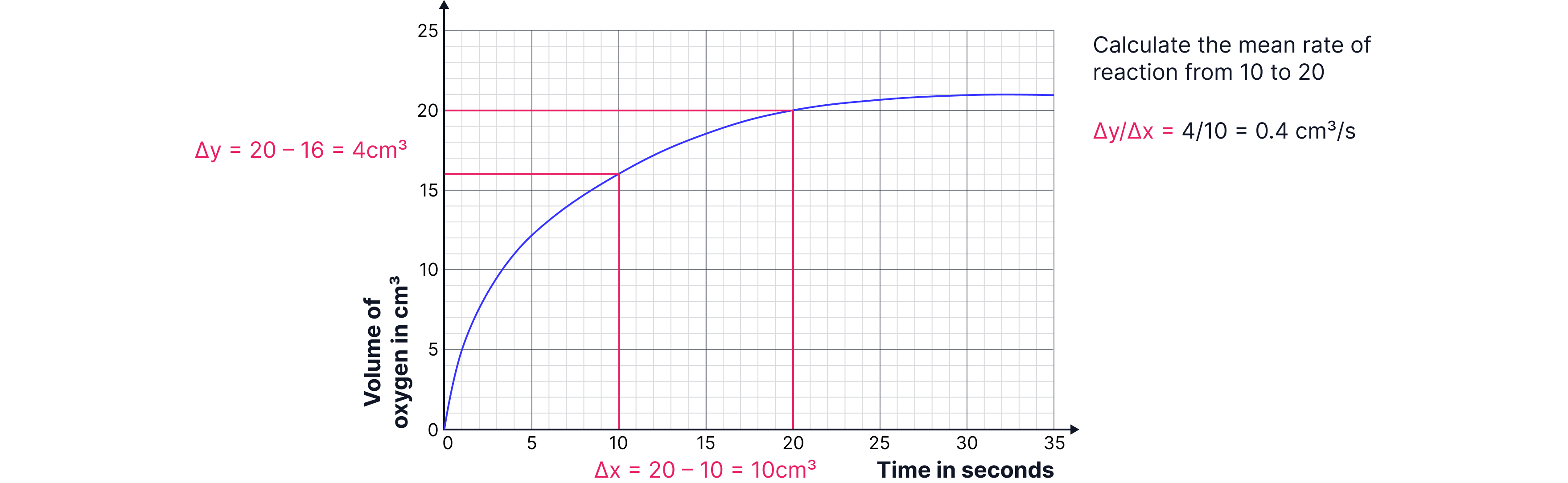
Finding Mean Rate from Graphs
You can calculate the mean rate of reaction using any two points on your graph:
Example:
Between 10 s and 20 s, the volume of gas increased from 16 cm³ to 20 cm³.
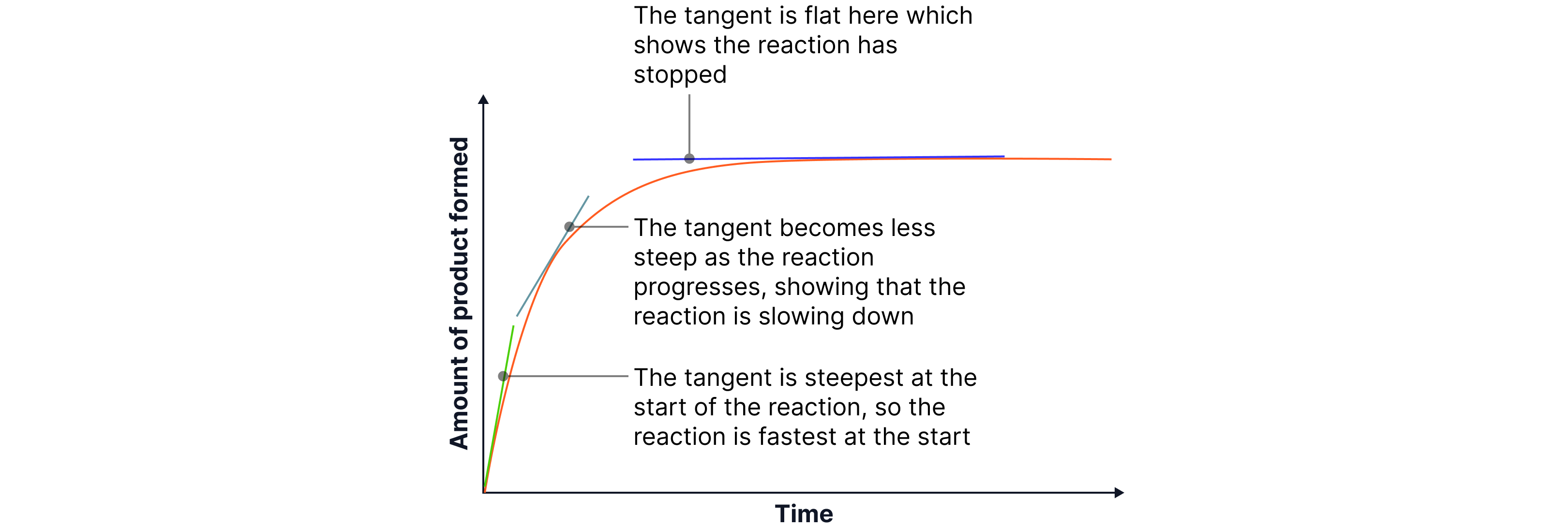
Interpreting rate graphs:
A graph of “quantity of product formed” or “reactant used up” against time shows how fast the reaction occurs.
A steep slope → fast reaction (high rate)
A gentle slope → slow reaction (low rate)
A flat line → reaction complete
Key idea: The slope (gradient) of the graph at any point represents the rate at that moment.
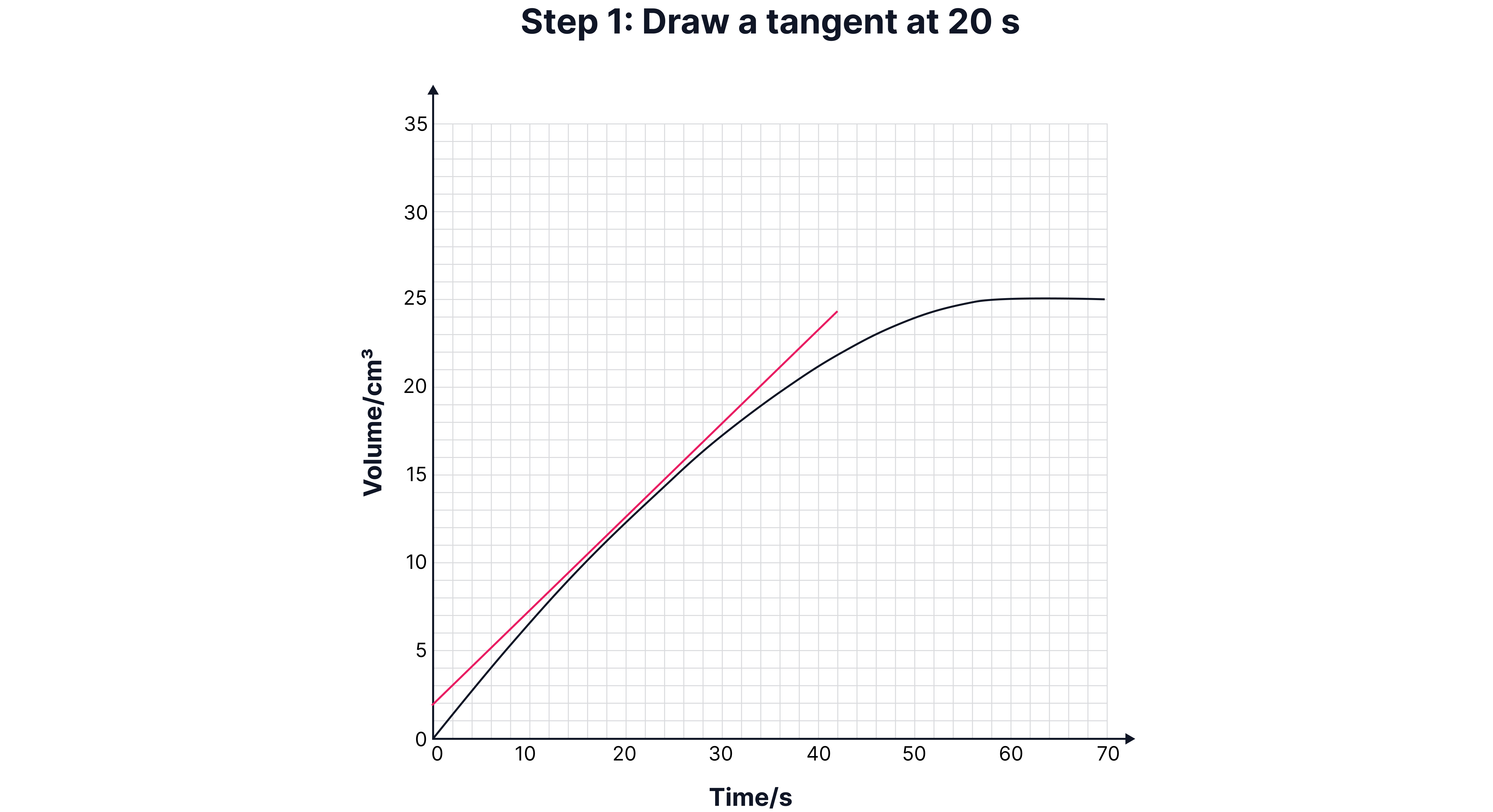
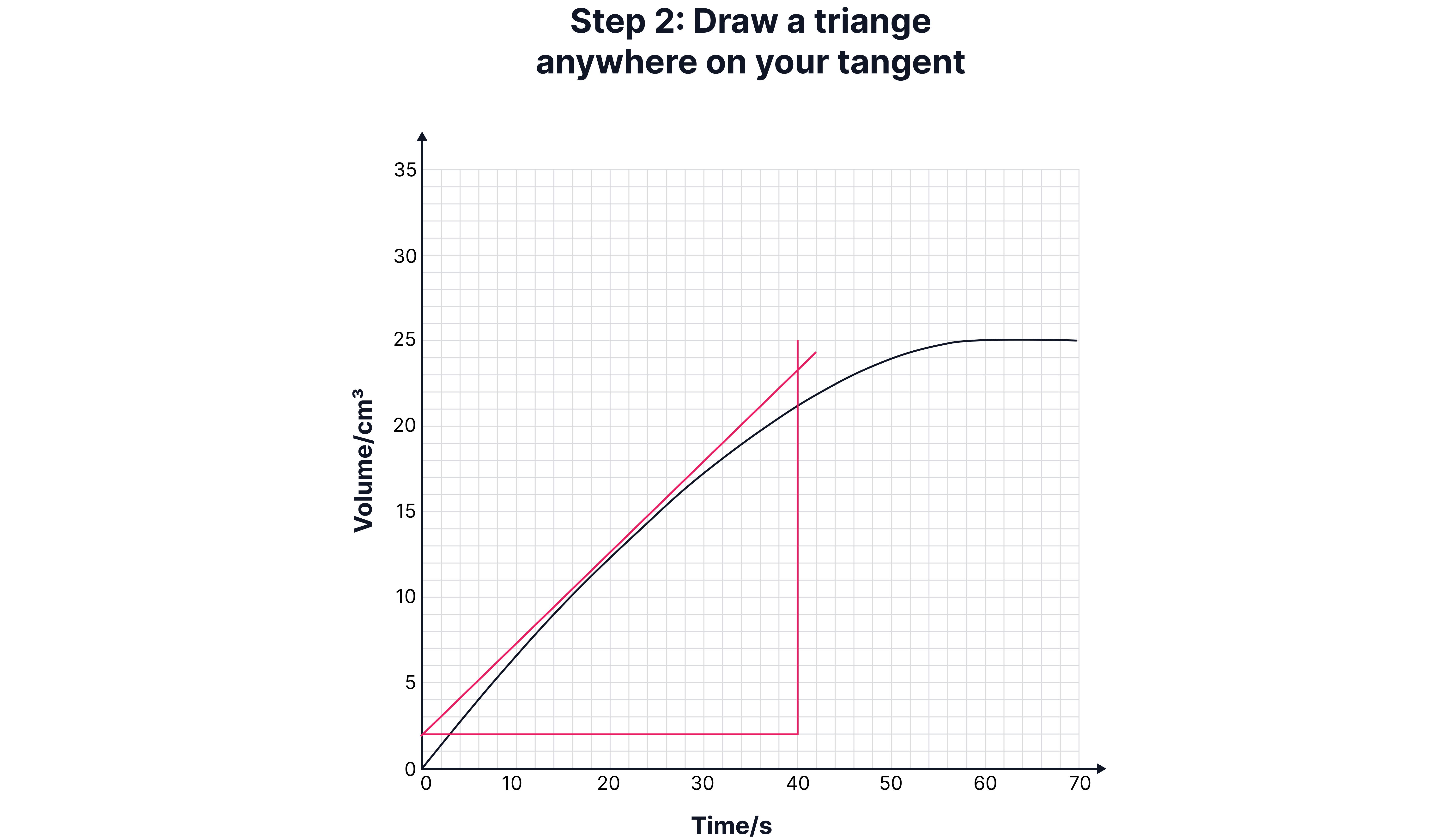
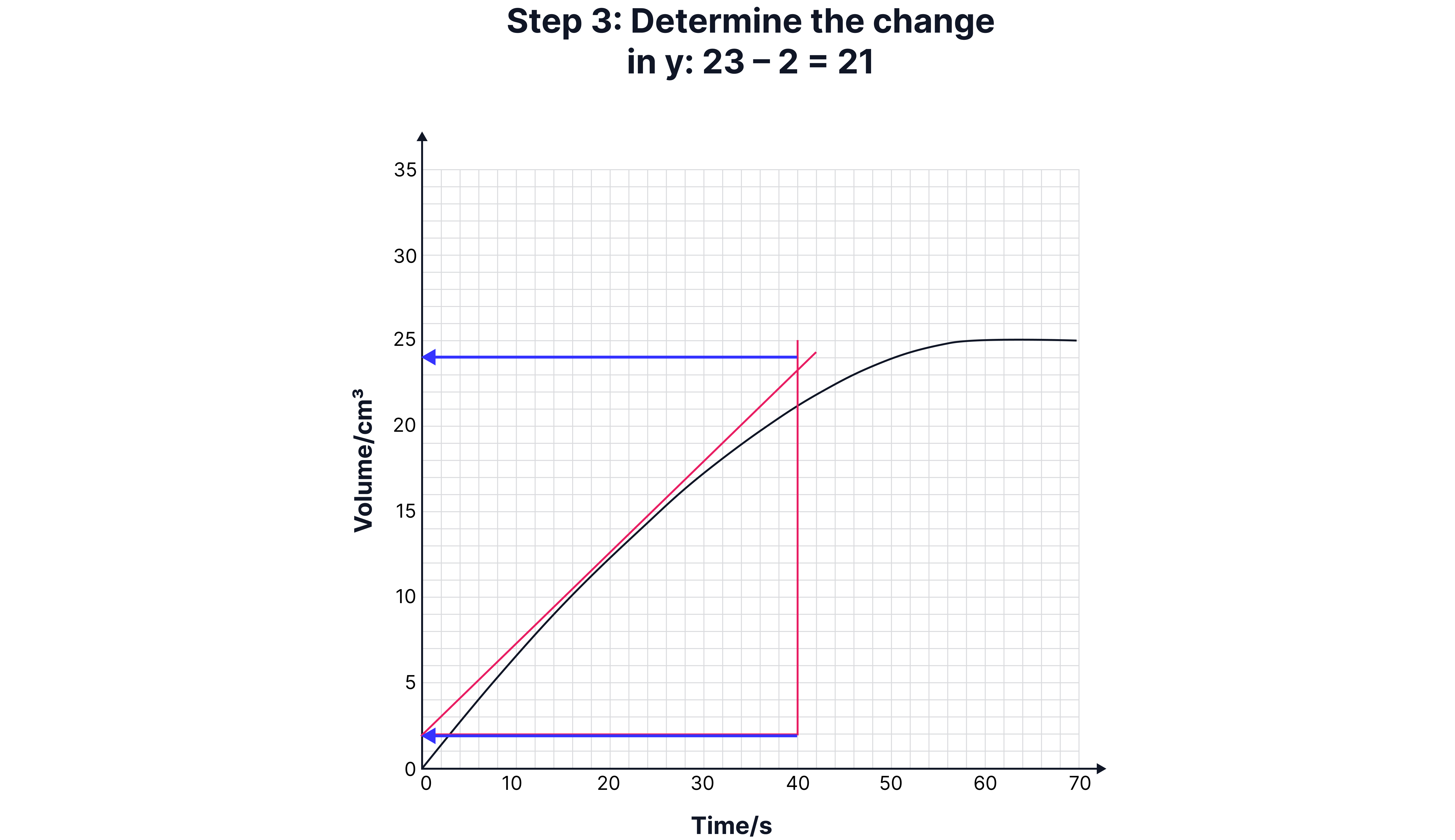
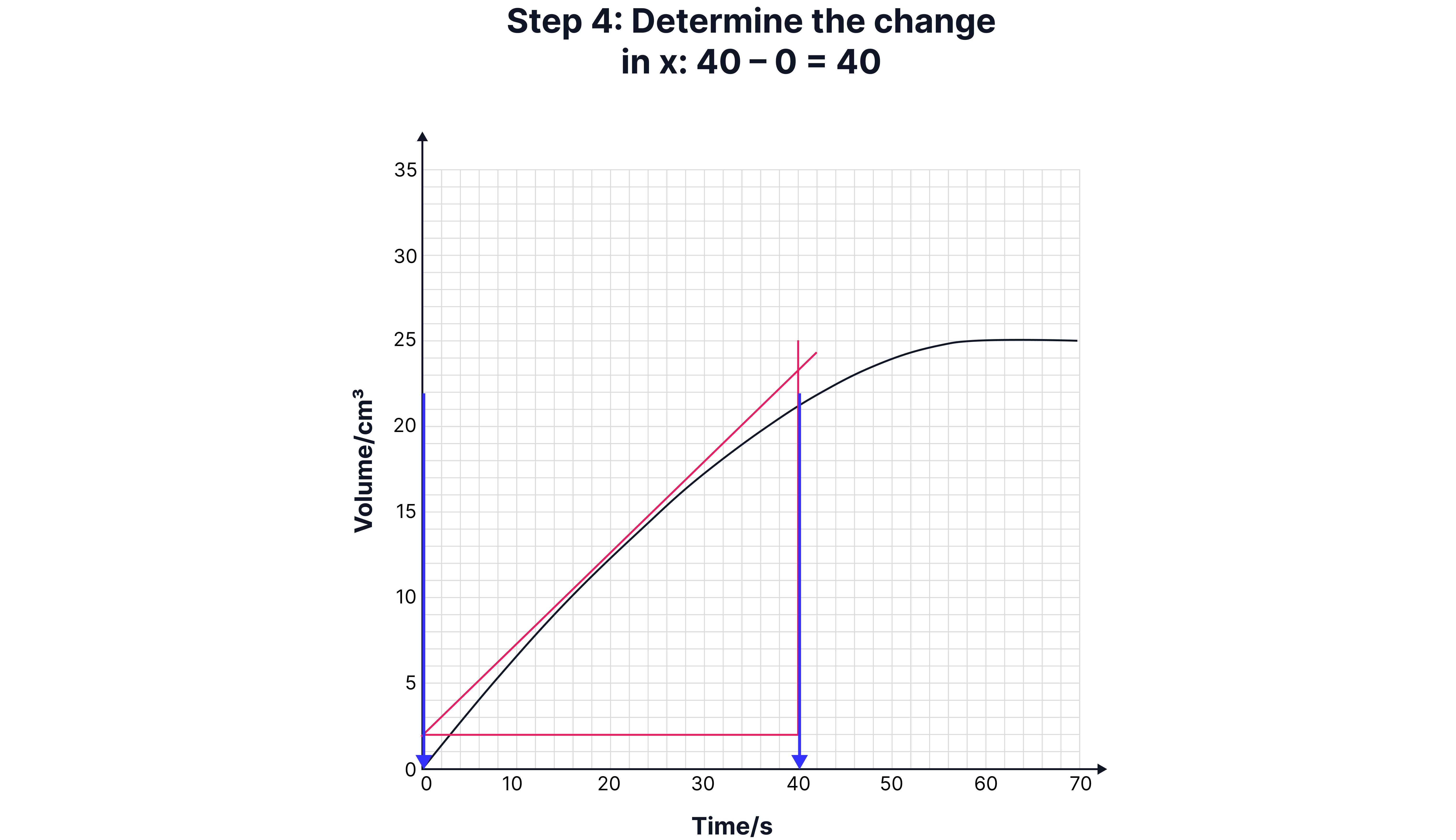
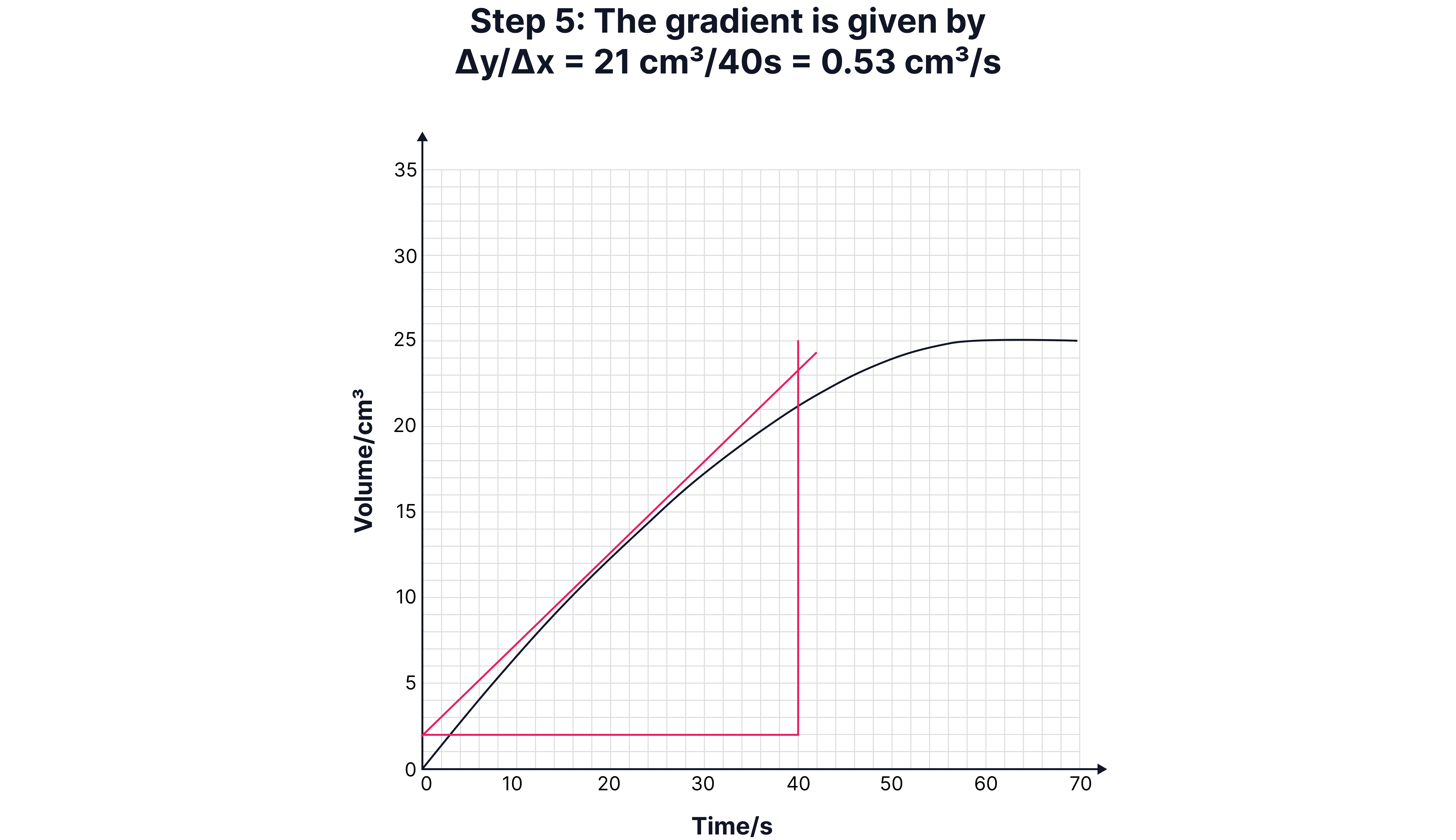
Calculating Instantaneous Rate (Using a Tangent)
Sometimes, you need the rate at a specific time (e.g., 15 seconds).
1. Draw a tangent to the curve at that point.
2. Construct a right-angled triangle under the tangent.
3. Calculate the gradient:
Calculating Instantaneous Rate (Using a Tangent)
Sometimes, you need the rate at a specific time (e.g., 15 seconds).
Draw a tangent to the curve at that point.
Construct a right-angled triangle under the tangent.
Calculate the gradient:
Worked Example:
Practice Questions
Recall
a. What is meant by the rate of a reaction?
a. How quickly reactants are converted into products
b. State three different methods used to measure the rate of a reaction.
b. Mass loss; gas volume (gas syringe or water displacement); colour change/cloudiness/turbidity (disappearing cross)
c. What does the gradient of a graph with volume-time (x-axis) represent?
c. The rate of reaction
Application
d. Explain why the mass of a flask decreases during the reaction between magnesium and hydrochloric acid.
d. Hydrogen gas is produced; gas escapes; mass decreases
e. A volume-time graph shows a steep slope at the start and then gradually levels off. Explain what this shows about the reaction.
e. Steep slope = fast initial rate; curve levels off as reactants are used up; flat line means reaction complete
f. Explain why the initial rate of reaction is usually the highest and decreases over time, using collision theory.
f. At the start concentration is highest; more frequent collisions; more successful collisions; as reactants are used up concentration decreases; fewer successful collisions; rate decreases
Challenge (HT)
g. Describe how you would calculate the instantaneous rate of reaction at 20 seconds using a graph.
g. Draw a tangent at 20 s; construct a right-angled triangle; calculate gradient (change in quantity ÷ change in time)