Factors Which Affect The Rates Of Chemical Reactions
Dr. Davinder Bhachu
Teacher

What Do We Mean by “Rate of Reaction”?
Core idea
The rate of a chemical reaction describes how quickly reactant particles are transformed into product particles.
Some reactions take place almost instantly, while others unfold over days, months, or even years.
Examples:
Slow reaction: Copper metal gradually turning dull when it oxidises in moist air.
Moderate reaction: A piece of zinc reacting with dilute acid to release hydrogen gas steadily.
Very fast reaction: Flash powder igniting to produce a burst of light.
The Four Major Factors That Influence Rate
1. Temperature
2. Concentration (or pressure, for gases)
3. Surface area of solid reactants
4. Catalysts
Each factor changes the number or success rate of particle collisions.
Collision Theory
Key principle
For a reaction to occur, particles must:
1. Collide with each other.
2. Collide with enough energy to break old bonds and form new ones.
Why rate changes
Anything that increases:
How often particles collide, or
How energetic those collisions are, will make the reaction go faster.
Increasing the Frequency of Collisions
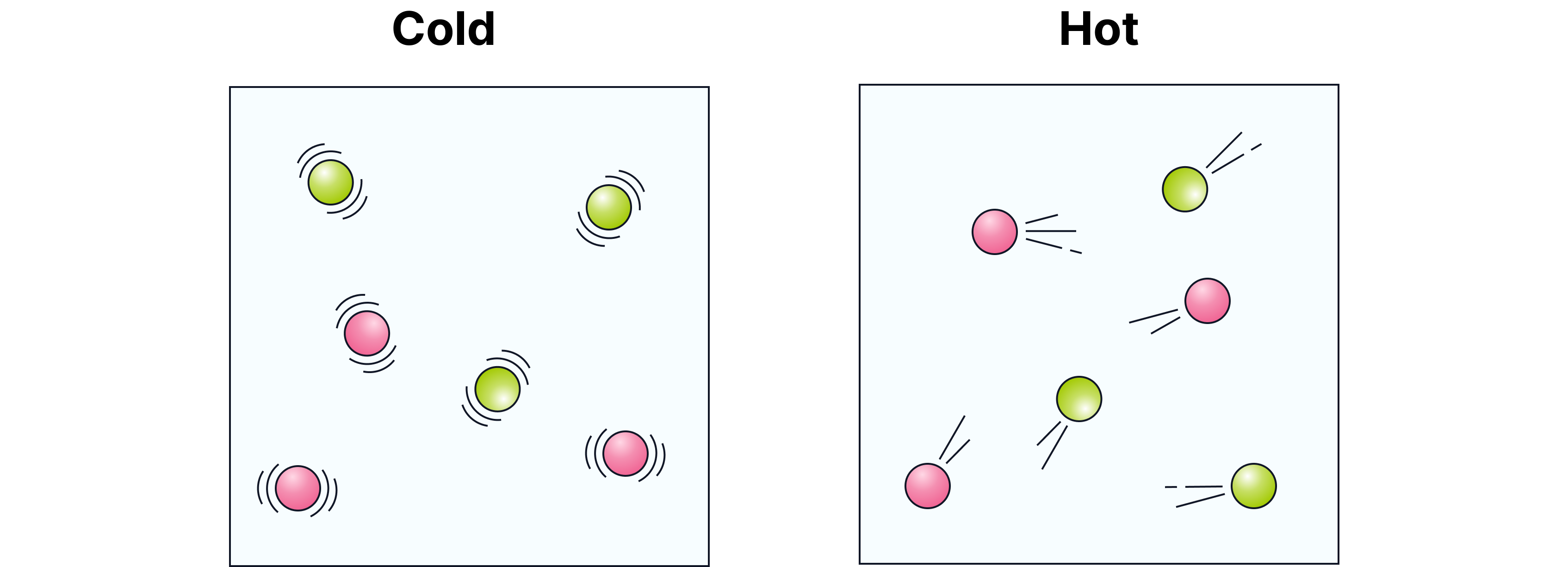
Temperature
Explanation
When the temperature rises:
Particles move faster.
Faster movement means they meet more often.
More frequent collisions → faster reaction.
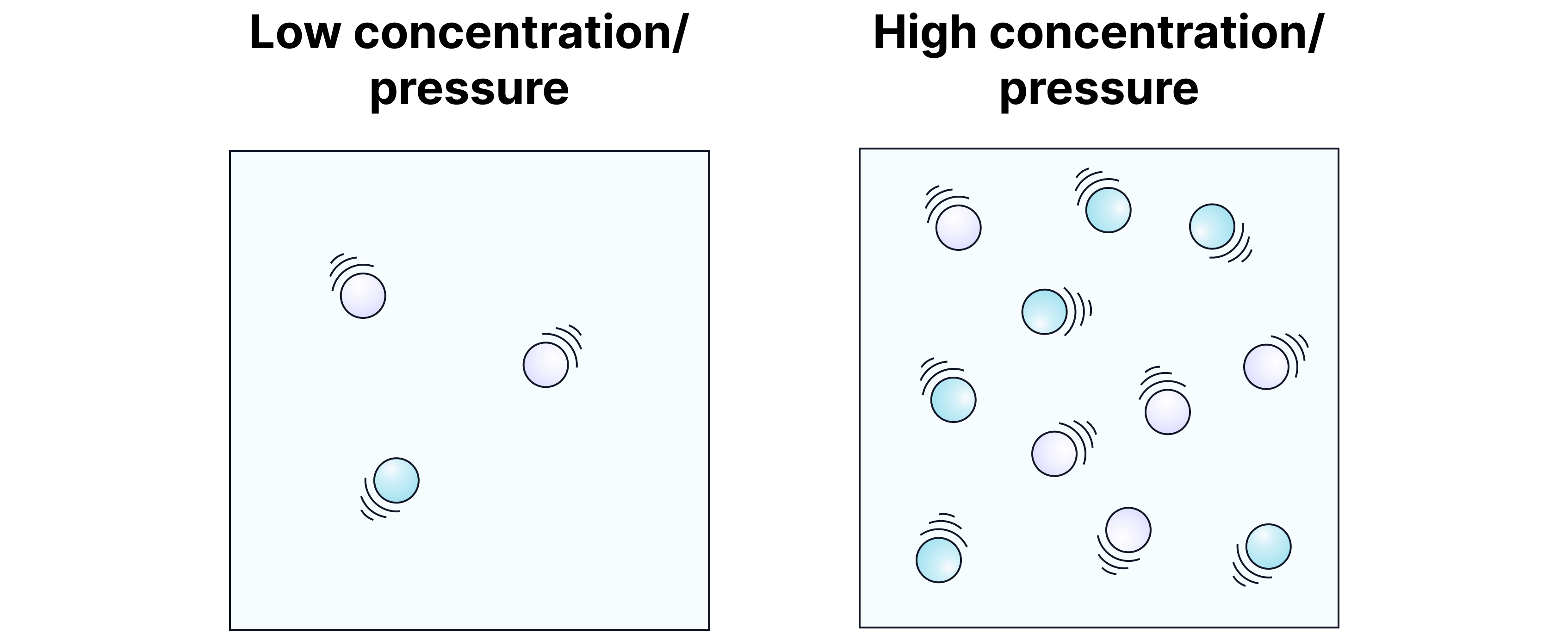
Concentration (or Pressure for Gases)
Explanation
In a more concentrated solution, there are more reactant particles in the same space.
More particles → more chances of collisions.
For gases: increasing pressure compresses particles together, raising collision frequency.
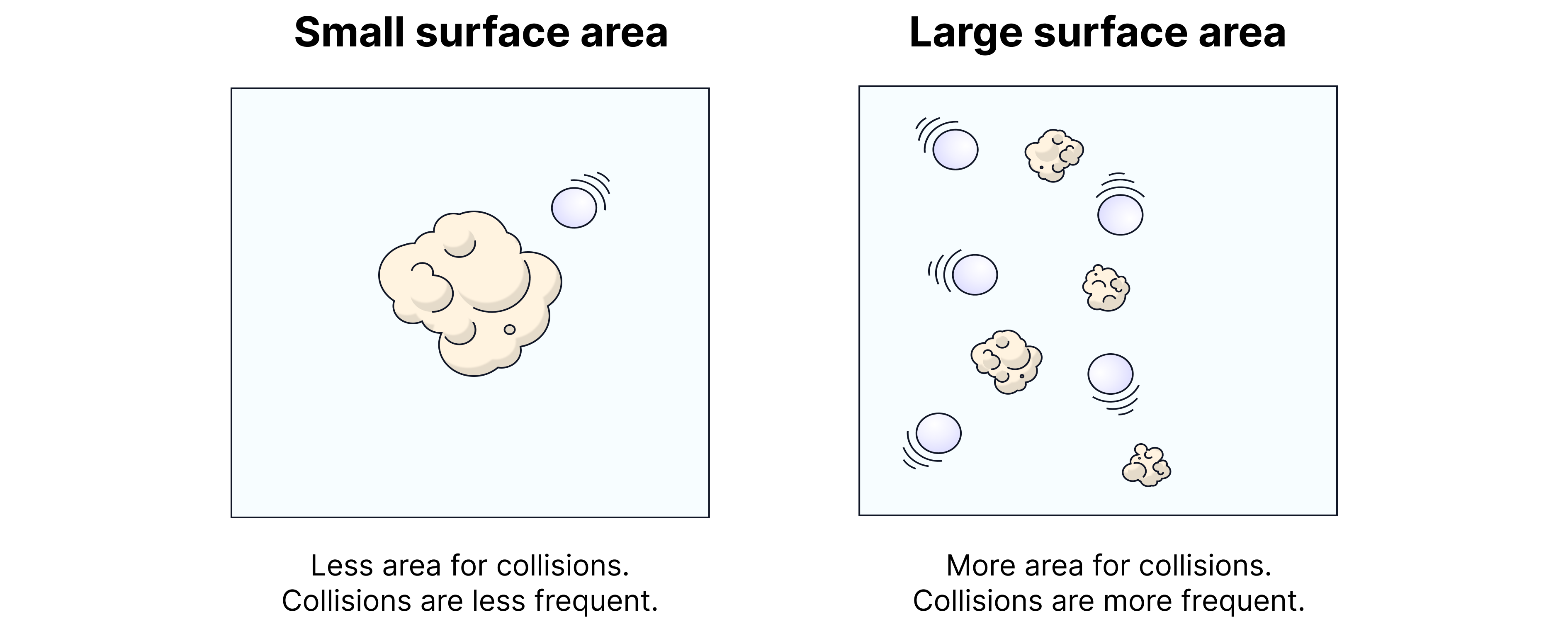
Surface Area
Explanation
When a solid reactant is broken into smaller pieces or ground into powder:
More particle surfaces are exposed.
Reactant particles in the solution or gas phase can collide with the solid more easily.
More exposed area → more collisions → faster reaction.

Increasing the Energy of Collisions
Activation energy
Reactions only occur when collisions have at least a minimum amount of energy known as the activation energy.
Why temperature matters again
Increasing temperature:
Not only makes collisions more frequent,
It also makes a higher proportion of collisions have energy equal to or greater than the activation energy.
Thus, temperature has a double effect on rate.
Rate and Proportionality
The rate of a reaction is directly proportional to the number of successful collisions each second.
Example
During the reaction between sodium thiosulfate and hydrochloric acid, the time until the solution clouds over is recorded at various temperatures. If increasing the temperature from 20 °C to 30 °C cuts the reaction time in half, then the frequency of successful collisions has roughly doubled.
Check for Understanding
Recall
a) What is meant by the rate of a chemical reaction?
Answer:
How quickly reactants are converted into products
b) State the four major factors that affect the rate of reaction.
Answer:
Temperature; concentration (or pressure for gases); surface area; catalyst
c) According to collision theory, what two conditions must be met for a reaction to occur?
Answer:
Particles must collide; collisions must have sufficient energy (equal to or greater than activation energy)
Application
d) Explain why increasing concentration increases the rate of reaction.
Answer:
More particles in same volume; more frequent collisions; more successful collisions
e) Explain why breaking a solid into smaller pieces increases the rate of reaction.
Answer:
Greater surface area exposed; more collisions between particles; increased collision frequency
f) Explain why increasing temperature has a greater effect on rate than simply increasing concentration.
Answer:
Temperature increases particle speed; increases collision frequency; increases proportion exceeding activation energy; double effect
Challenge (HT)
g) Explain, using activation energy and collision theory, why increasing temperature increases the proportion of successful collisions.
Answer:
Activation energy is the minimum energy required; increasing temperature increases kinetic energy; more particles have energy ≥ activation energy; greater proportion of successful collisions per second
More Practice
Try to answer the practice question from the TikTok video on your own, then watch the video to see how well you did!