Cells and batteries (Triple Only)
Dr. Davinder Bhachu
Teacher

Contents
What is a Cell?
A cell is a device that uses a chemical reaction to produce electricity. It contains:
Two different metal electrodes (conduct electricity and release/accept electrons)
An electrolyte (a solution containing ions)
When the metals are placed into the electrolyte, a chemical reaction releases electrons. If the electrodes are joined by a wire, these electrons flow through the circuit, producing an electric current.
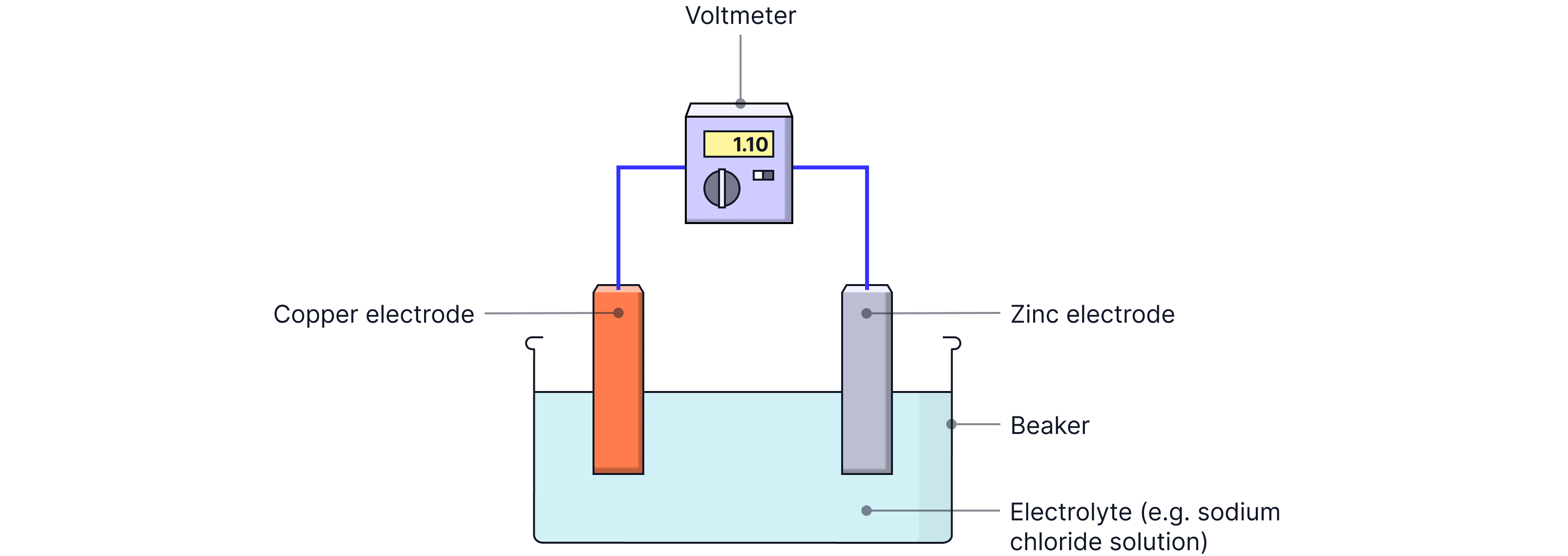 Key idea:
Key idea:
Electrons always flow from the more reactive metal to the less reactive metal.
Voltage and Reactivity
Each metal has a different tendency to lose electrons (a different reactivity). The more reactive metal:
Loses electrons more easily
Becomes the negative electrode (anode)
Pushes electrons around the circuit
The less reactive metal becomes the positive electrode (cathode). If the voltmeter shows a positive reading, meaning the more reactive metal is the negative electrode.
Key point:
Most reactive metal → negative electrode → releases electrons
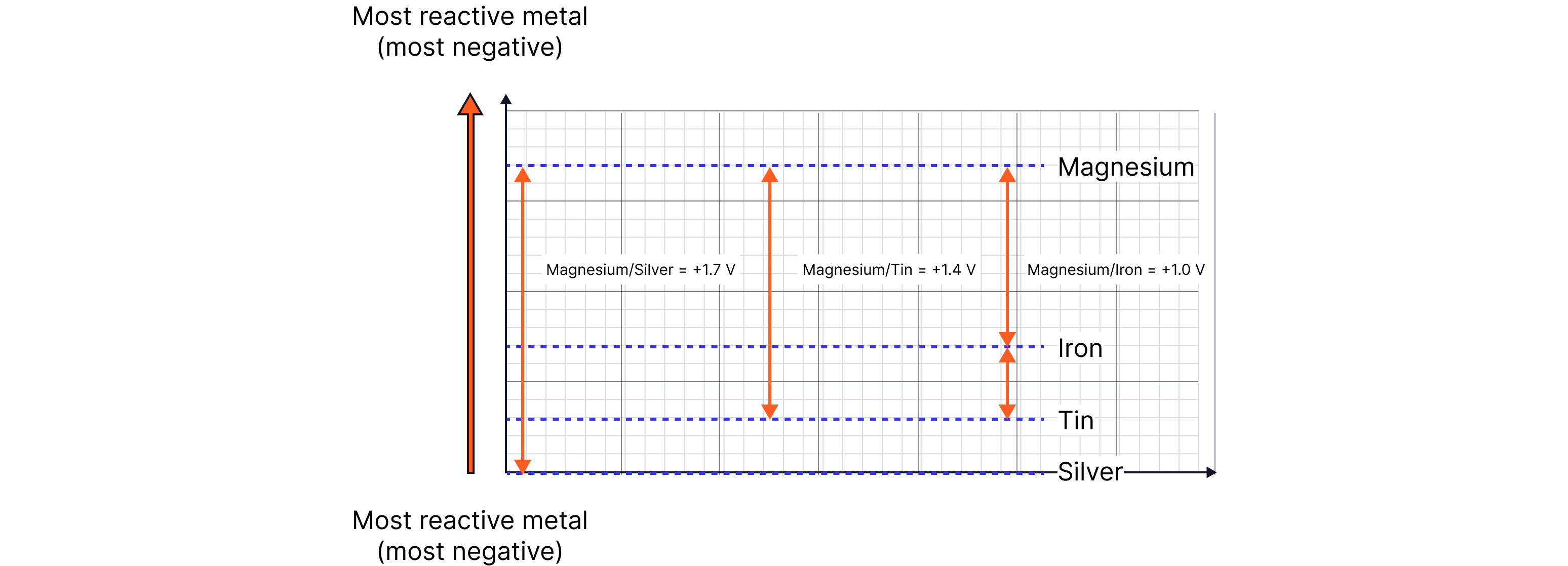
For example:
| Voltage/V | |
Magnesium | Tin | +1.4 |
Magnesium | Iron | +1.0 |
Magnesium | Silver | +1.7 |
Iron | Silver | +0.7 |
From this data we can conclude:
The combination of magnesium and silver gives the largest voltage. This is because of the difference in reactivity between magnesium and silver. So, which electrode is the negative electrode?
If the voltmeter shows a positive reading (+1.7 V in this case), this means that the more reactive metal is the negative electrode.
Therefore, the more reactive metal (out of magnesium and silver) in this case must be magnesium.
In fact, if we plot this on a graph, we can see a reactivity series presents itself.
Predicting Voltage
Once you know the order of reactivity, you can predict:
Which metal becomes negative
Whether the voltage is positive or negative
Which pair gives the largest voltage
If the less reactive metal is accidentally placed on the left-hand terminal, the voltage will be negative, because electron flow is reversed.
Worked Example:
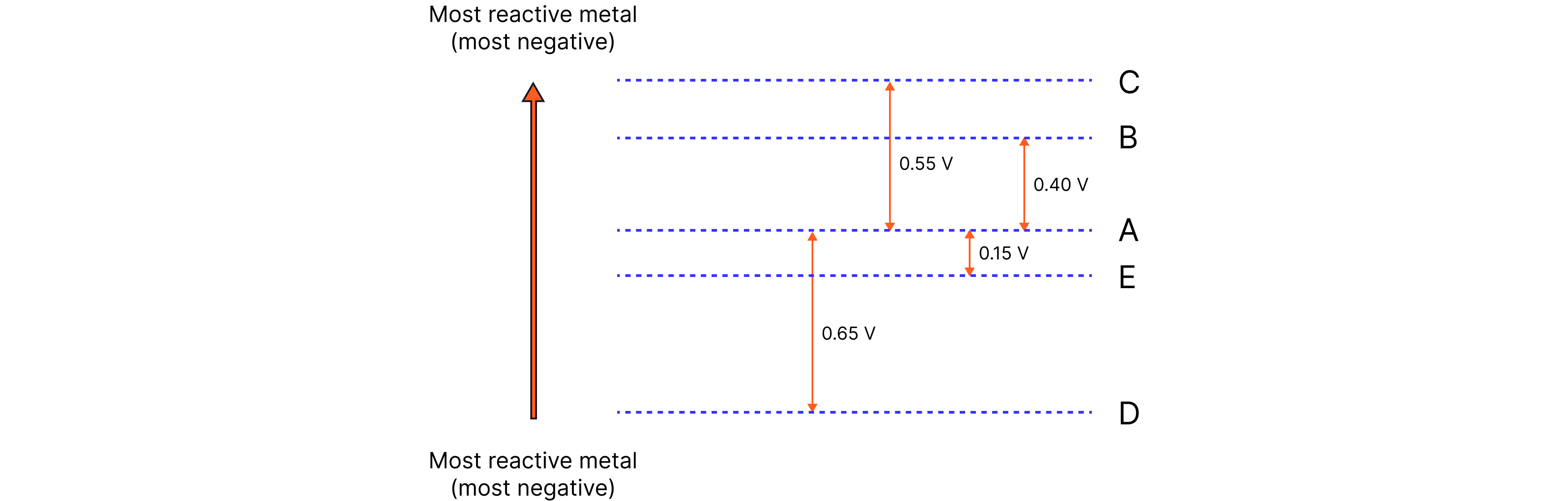
Metals A, B, C and D are connected in cells and the potential difference is measured.
Metal connected to positive terminal of voltmeter | Metal connected to negative terminal of voltmeter | Potential difference (V) |
A | B | +0.40 |
A | C | +0.55 |
A | D | –0.65 |
A | E | –0.15 |
a) Put the metals in order of reactivity (most reactive first).
b) Calculate the potential difference of a cell made from C and E, with C connected to the positive terminal of the voltmeter.
Answer: To work this out simply add up the values of the lines from C E. So 0.55 V + 0.15 V = 0.70 V. As C (the most reactive metal) is connected to the positive terminal them the value will be -0.7 V.
c) Calculate the potential difference of a cell made from D and E, with D connected to the positive terminal of the voltmeter.
Answer:
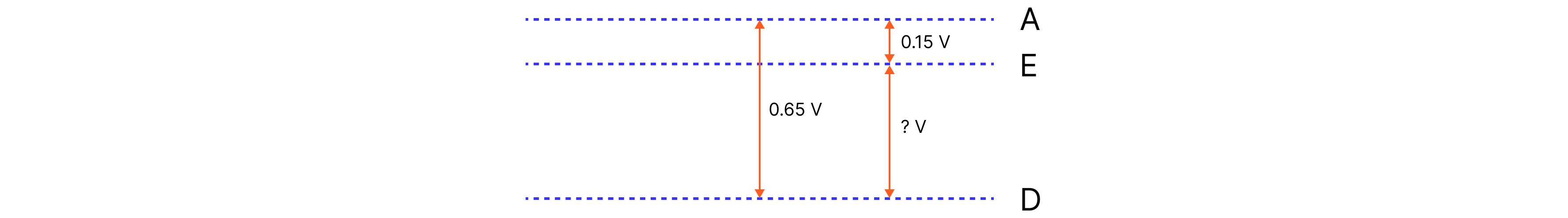
We can see:
1. ? + 0.15 V = 0.65V. If we rearrange for ?, then
2. ? = 0.65 V - 0.15 V = 0.5V
As D is the positive electrode (and the less reactive metal in this case) the value or D connected to E will be + 0.5 V.
How Electrolytes Affect Voltage
The electrolyte must contain mobile ions. It allows charge to move through the cell.
If ion concentration decreases → voltage drops
If the electrolyte dries out → cell stops working
Different electrolytes → different voltages (even with the same metals)
Misconception alert:
The electrolyte does NOT produce the electrons. It allows ions to move and completes the circuit.
Batteries
A battery is formed when two or more cells are connected in series.
The total voltage = sum of the individual cell voltages.
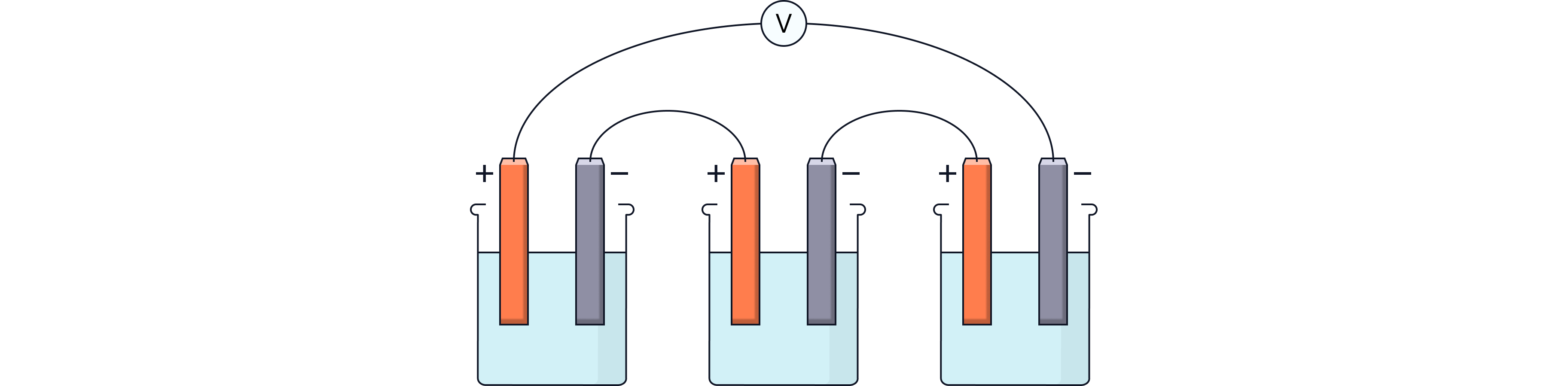
Example:
0.6 V + 0.6 V + 1.2 V = 2.4 V
Rechargeable vs Non-Rechargeable Cells
Type | Key idea | Reactions | Reusable? |
Non-rechargeable | Chemicals are used up | Irreversible reactions | No |
Rechargeable | External current restores reactants | Reversible reactions | Yes |
Examples:
Non-rechargeable: alkaline battery
Rechargeable: lithium-ion battery (phones), NiMH (rechargeable AA)
Practice Questions
Recall
a) What is the role of the electrolyte?
a) Allows ions to move and complete the circuit.
b) Why is the most reactive metal always the negative electrode?
b) It loses electrons more easily, so becomes the anode and pushes electrons through the circuit.
c) What is meant by a battery?
c) A battery is two or more cells connected in series.
d) What happens to voltage when more cells are connected in series?
d) The total voltage increases (it is the sum of the individual voltages).
Application
e) A student gets a negative voltage reading. What does this tell you about the metals?
e) The less reactive metal is the negative electrode, so the polarity has been reversed.
f) Two cells of 1.1 V and 0.9 V are connected in series. What is the total voltage?
f) 1.1 + 0.9 = 2.0 V
g) Which pair of metals would produce a higher voltage: magnesium + copper or iron + copper? Explain why.
g) Magnesium + copper, because magnesium is more reactive, giving a larger difference in reactivity and higher voltage.
h) What would happen to the voltage if the electrolyte started to dry out?
h) The voltage would decrease and eventually stop as ions can no longer move.
Challenge (HT)
i) Why does a cell eventually stop producing electricity?
i) Reactants are used up / ion concentration falls, so the reaction stops.
j) Why can rechargeable cells be reused but non-rechargeable ones cannot?
j) Rechargeable cells have reversible reactions; non-rechargeable cells have irreversible reactions so the reactants are used up.
k) Explain why a greater difference in reactivity between two metals leads to a higher voltage.
k) A larger difference in reactivity means a greater tendency for electrons to flow, increasing potential difference (voltage).
l) A cell uses two metals but produces almost no voltage. Suggest two possible reasons.
l) Possible reasons:
The metals have very similar reactivities
The electrolyte is weak, low concentration, or dried out
Poor connections in the circuit