Fuel Cells (Triple Only)
Dr. Davinder Bhachu
Teacher

Contents
Fuel Cells
1. What is a fuel cell?
A fuel cell is a device that converts chemical energy directly into electrical energy. Unlike a normal cell (which eventually runs out of reactants), a fuel cell works continuously, as long as it is supplied with:
- a fuel (such as hydrogen), and
- oxygen (from air)
This produces a voltage that can power external devices (e.g. a motor in a car).
Key idea
Fuel cells are not recharged. They keep producing electricity as long as fuel and oxygen are supplied.
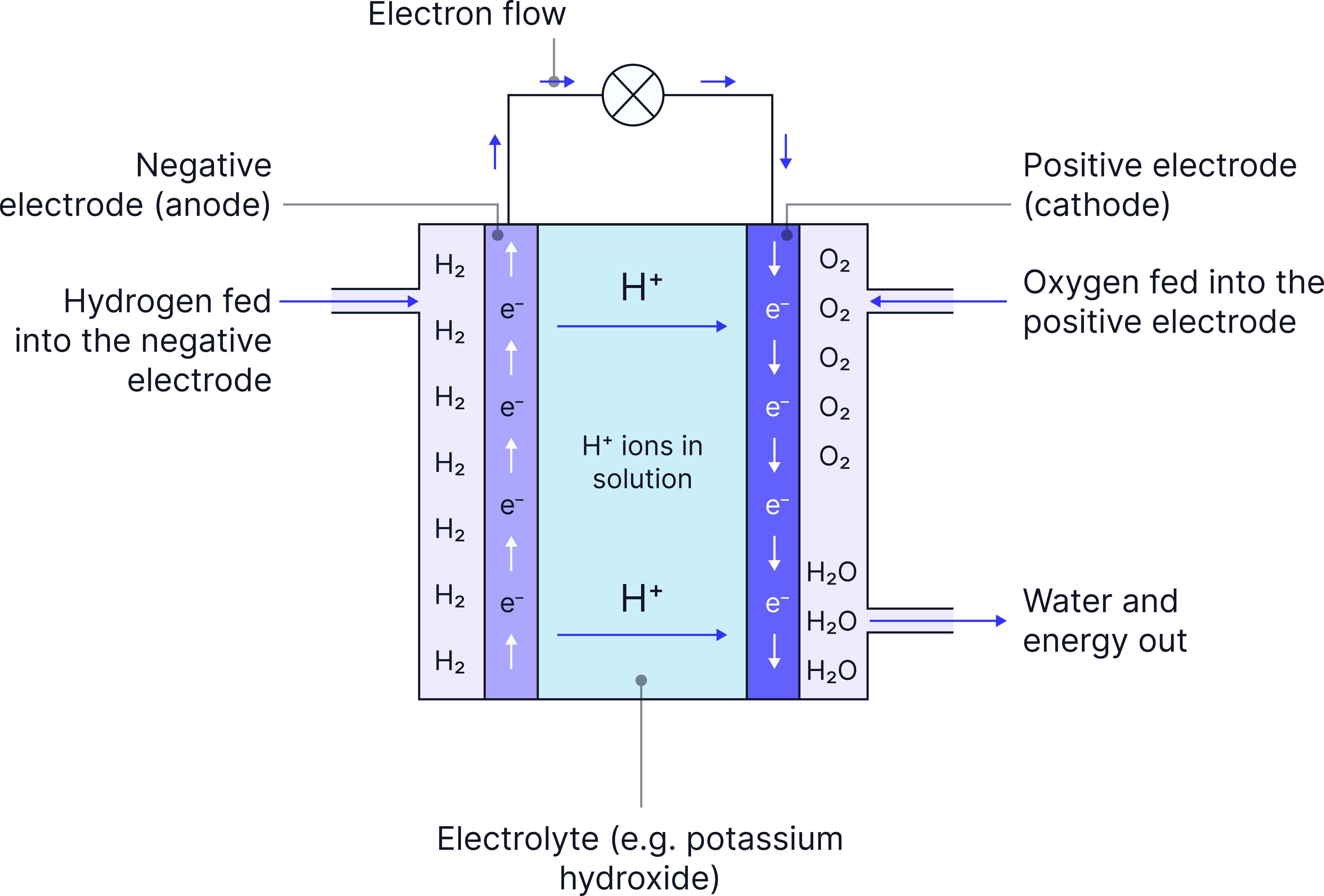
The Hydrogen-Oxygen Fuel Cell
- Hydrogen goes to the negative electrode (anode)
- Hydrogen loses electrons, forming H⁺ ions
- The electrons travel through the external circuit, making electricity
- At the cathode, oxygen gains electrons
- H⁺ ions (from the electrolyte) and oxygen combine to make water
- The only product is H₂O (no CO₂ released)
|
Electrode |
What happens? |
Type of reaction |
|
Negative electrode (anode) |
Hydrogen loses electrons |
Oxidation |
|
Positive electrode (cathode) |
Oxygen gains electrons |
Reduction |
The electrolyte is often an alkaline solution, such as potassium hydroxide, which allows ions to move.
The Electrode Reactions (Higher Tier)
At the anode (oxidation):
H₂ → 2H⁺ + 2e⁻
At the cathode (reduction):
O₂ + 4H⁺ + 4e⁻ → 2H₂O
Overall reaction:
2H₂ + O₂ → 2H₂O
Because hydrogen is oxidised, it must be at the anode. Oxygen is reduced at the cathode.
Advantages And Disadvantages Of Hydrogen Fuel Cells
|
Advantages |
Explanation |
|
Only product is water |
No CO₂ or air pollutants at point of use |
|
Continuous supply |
Works as long as fuel and oxygen are supplied |
|
Lightweight for the energy they provide |
Good for vehicles and spacecraft |
|
Disadvantages |
Explanation |
|
Hydrogen is difficult to store |
Needs high-pressure tanks |
|
Hydrogen is flammable |
Safety risk |
|
Hydrogen often made from fossil fuels |
Can still create CO₂ overall |
Check Your Understanding
Recall
a) What is the product of a hydrogen fuel cell?
Answer:
Water
b) Which electrode is the anode in a hydrogen fuel cell?
Answer:
The negative electrode
c) What type of reaction occurs at the cathode?
Answer:
Reduction
Application
d) Why can a fuel cell run continuously?
Answer:
Fuel and oxygen are constantly supplied, so reactants do not run out
e) What is the role of the electrolyte?
Answer:
It allows ions (H⁺) to move between electrodes
f) Give one advantage and one disadvantage of using hydrogen fuel cells in vehicles.
Answer:
Advantage: no CO₂ at point of use. Disadvantage: hydrogen is difficult to store
Challenge (HT)
g) Explain why hydrogen must be supplied to the anode and oxygen to the cathode.
Answer:
Hydrogen is oxidised at the anode (loses electrons), while oxygen is reduced at the cathode (gains electrons)
h) Write the half-equation for the reaction at the anode.
Answer:
H₂ → 2H⁺ + 2e⁻
i) Write the half-equation for the reaction at the cathode.
Answer:
O₂ + 4H⁺ + 4e⁻ → 2H₂O
More Practice
Try to answer the practice question from the TikTok video on your own, then watch the video to see how well you did!