Changes of State
Dr. Davinder Bhachu
Teacher

Contents
What You Already Know
You’ve seen butter melt on toast or water freeze into ice cubes. These are all examples of changes of state. When a substance changes state, the particles don’t change, only their arrangement and movement do.
In this note, you’ll use the particle model to explain what happens when substances melt, freeze, boil or condense.
Why Do Substances Change State?
A change of state is a physical change. This means no new substance is formed. Only the energy or arrangement of the particles changes.
The main changes of state are:
Melting (solid → liquid)
Freezing (liquid → solid)
Boiling (liquid → gas)
Condensing (gas → liquid)
Subliming (solid → gas directly, e.g. dry ice)
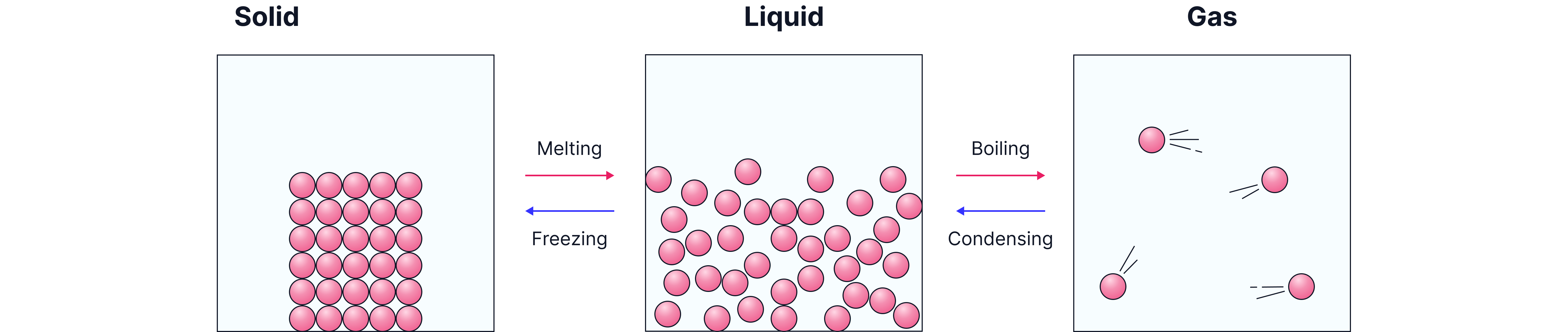
From Solid to Liquid – Melting

When a solid is heated, the particles gain energy. They vibrate faster and the forces of attraction between them become weaker. At a certain temperature, called the melting point, the particles have enough energy to break free from their fixed positions. The solid becomes a liquid.
Example:
Ice melts at 0°C because water molecules gain enough energy to move freely.
From Liquid to Gas – Boiling
When a liquid is heated, the particles gain even more energy. They move faster and the forces holding them together become weaker. At the boiling point, particles have enough energy to completely overcome the attractive forces between them.
The liquid becomes a gas.
Remember: Boiling happens throughout the liquid, not just at the surface.
From Liquid to Gas - Evaporation
Evaporation is when a liquid changes to a gas below its boiling point.
Some particles at the surface have enough energy to escape into the air.
Example:
A puddle dries up on a sunny day even though the water never boiled.
The Difference Between Boiling and Evaporation
Although both involve a liquid changing into a gas, they happen in different ways:
Feature | Boiling | Evaporation |
Temperature | Happens at the boiling point only (e.g. 100°C for water). | Can happen at any temperature below the boiling point. |
Where it happens | Happens throughout the whole liquid. | Happens only at the surface of the liquid. |
Energy input | Needs a constant heat source to keep supplying energy. | Uses energy from the surroundings (air, skin, etc.) to let surface particles escape. |
Speed | Happens quickly when boiling point is reached. | Happens slowly at everyday temperatures. |
Visible sign | Bubbles form throughout the liquid. | No bubbles — particles escape one by one from the surface. |
From Gas to Liquid – Condensing
As a gas cools, the particles lose energy. They move more slowly and can no longer overcome the forces between them. When these forces are strong enough to hold the particles together, the gas becomes a liquid.
This happens at the boiling point, but in reverse.
Example:
When you breathe out on a cold day, water vapour condenses to form tiny droplets – that’s why you can “see your breath”.
From Liquid to Solid – Freezing
When a liquid cools, the particles move more slowly. There isn’t enough energy to overcome the attraction between them, so they move closer together.
At the melting point, the forces between the particles become strong enough to keep them in fixed positions.
The liquid becomes a solid.
Example:
Water freezes at 0°C to form ice.
What Controls Melting and Boiling Points?
The stronger the forces between the particles, the more energy is needed to break them apart.
This means the melting and boiling points are higher.
Type of substance | Type of bonding | Typical melting/boiling points |
Simple molecular (e.g. oxygen, water) | Weak intermolecular forces | Low |
Ionic (e.g. sodium chloride) | Strong electrostatic forces | High |
Metallic (e.g. copper, iron) | Strong metallic bonds | Very high |
Predicting the State of a Substance
You can work out the state of a substance if you know its melting and boiling points.
Example 1: Bromine
Melting point = –7°C Boiling point = 59°C

Example 2: Oxygen
Melting point = –7°C Boiling point = 59°C

Practice Questions
Recall
a) What is meant by a change of state?
Answer:
a) A change of state is a physical change where the same substance changes between solid, liquid and gas forms.
b) Describe what happens to particles when a solid melts.
Answer:
b) When a solid melts, particles gain energy, vibrate faster and break free from their fixed positions.
c) How is boiling different from evaporation?
Answer:
c) Boiling happens throughout the liquid at its boiling point, while evaporation only happens at the surface and at any temperature below the boiling point.
Apply Your Knowledge
d) Carbon dioxide sublimes at –78°C. Predict its state at:
i) –100°C
ii) 25°C
Answer:
d) i) –100°C = solid, ii) 25°C = gas.
e) Explain why substances with strong forces between particles have higher melting points.
Answer:
e) Stronger forces between particles need more energy to break, so the melting and boiling points are higher.
f) Water boils at 100°C and freezes at 0°C. What state is it at –10°C, 50°C and 120°C?
Answer:
f) –10°C = solid (ice), 50°C = liquid (water), 120°C = gas (steam).
Challenge Yourself
g) Explain how sweating cools you down using the particle model.
Answer:
g) When sweat evaporates, the fastest-moving particles escape, taking heat energy away from the skin and cooling it.
h) Why do droplets form on the outside of a cold glass on a warm day?
Answer:
h) Warm air contains water vapour. When it touches a cold surface, the vapour cools, loses energy and condenses into liquid droplets.