States Of Matter
Dr. Davinder Bhachu
Teacher

Contents
What You Already Know
You’ve seen ice melt into water and watched water boil to make steam. These are examples of changes of state. The substance stays as H₂O, but its form changes. In this topic, you will learn how scientists explain these changes using the particle model.
Introducing the Particle Model
Scientists use the particle model to describe how materials behave.
It shows substances as made of tiny particles (atoms, ions or molecules) that move differently depending on the state of matter. Although the model uses simple spheres, it helps us to understand why materials have certain shapes, volumes and densities, and how they change state when heated or cooled.
Solids
Think about it: Why does an ice cube keep its shape even when you take it out of the tray?
Key Ideas
Particles are packed closely together in a regular pattern.
There are strong forces of attraction between them.
Particles vibrate in fixed positions but do not move freely.
Solids have a fixed shape and a fixed volume.
When heated, vibrations increase and the solid expands slightly.
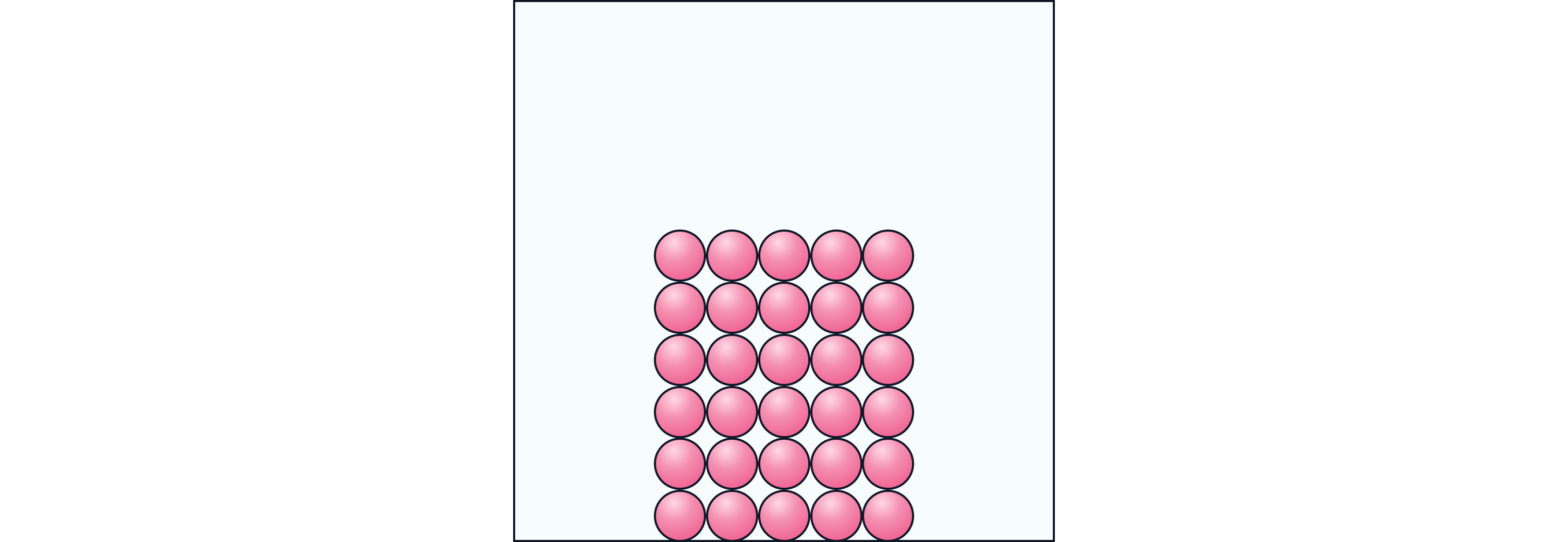
Visual Idea:
Imagine people standing close together in rows at assembly. They can shuffle a bit but not swap places.
Liquids
Think about it: When you pour water into a glass, it takes the shape of the glass. Why?
Key Ideas
Particles are close together, but not arranged in a regular pattern.
The forces between particles are weaker than in solids.
Particles can move and slide past each other, which allows the liquid to flow.
Liquids have a fixed volume but no fixed shape.
When heated, particles move faster and the liquid expands slightly.

Thermometers work because the liquid inside expands when heated, moving up the tube.
Visual idea:
Picture marbles in a box that can roll around each other. That’s how particles move in a liquid.
Gases
Think About It: When you open a bottle of perfume, why can someone smell it across the room?
Key Ideas
Particles are very far apart and move freely and quickly.
The forces of attraction between them are extremely weak.
Gas particles move in random directions and collide with each other and the container walls.
Gases have no fixed shape or volume and will spread out to fill any space.
When heated, particles move faster, collide more often and increase the pressure inside the container.
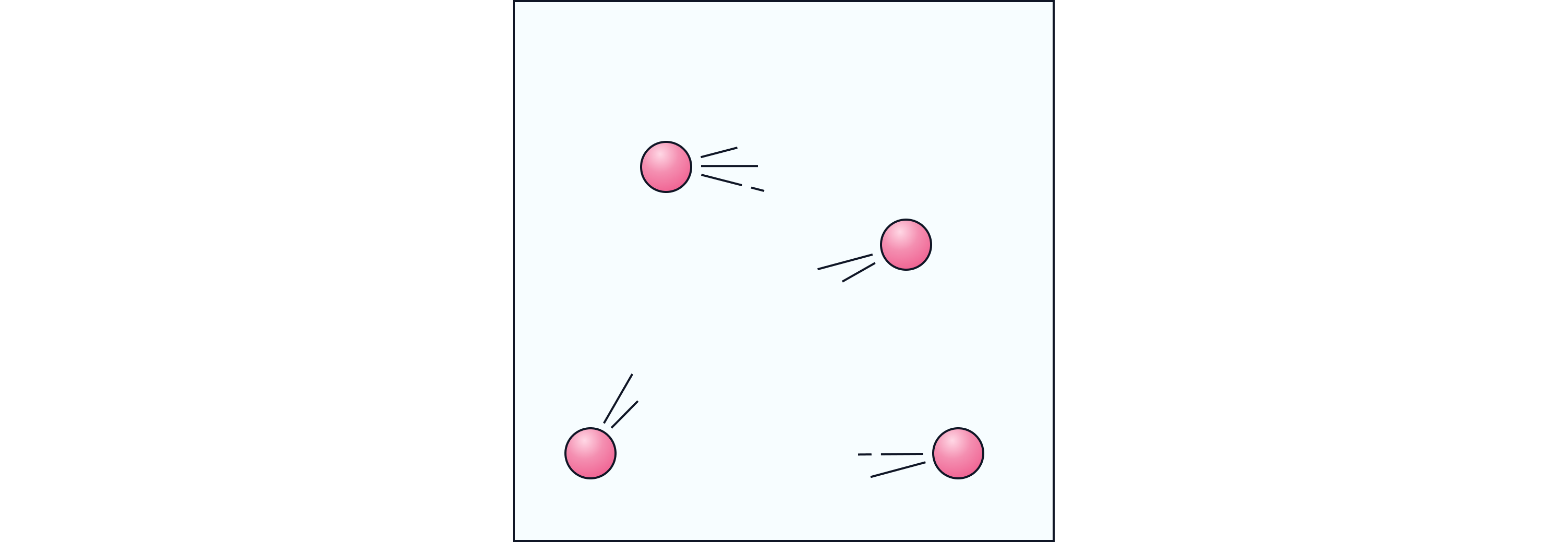
Visual Idea:
Think of ping-pong balls bouncing randomly around a box. That’s similar to how gas particles move.
Link: This random movement is called Brownian motion.
Comparing The Three States
| Property | Solid | Liquid | Gas |
| Forces between particles | Strong | Weaker | Very weak |
| Arrangement | Regular, fixed | Random, close | Random, far apart |
| Movement | Vibrate only | Slide past each other | Move freely |
| Shape | Fixed | Takes container shape | Fills container |
| Volume | Fixed | Fixed |
Not fixed |
Worked Example
Question: What happens to particles in a solid when it melts?
Answer (Step-by-step):
Heating gives the particles more energy.
They vibrate faster, weakening the forces between them.
Eventually, the particles break free from their fixed positions.
The solid becomes a liquid, where particles can move past each other.
Answer (Step-by-step):
1. Heating gives the particles more energy.
2. They vibrate faster, weakening the forces between them.
3. Eventually, the particles break free from their fixed positions.
4. The solid becomes a liquid, where particles can move past each other.
Limitations Of The Particle Model
The particle model is useful, but it isn’t perfect.
It doesn’t show:
The forces between particles or how strong they are.
The true shapes or sizes of atoms and molecules.
The empty space between particles accurately.
Bulk and Atomic Properties
A sample of a material contains billions of atoms or molecules. These particles give the material its bulk properties, such as density, melting point and colour.
Each tiny part of the material behaves the same way because it contains identical particles.
Nanoparticles contain only a few hundred atoms. They can behave differently from the same substance in bulk form and are often used in medicines and advanced materials.
State Symbols in Equations
Symbol | Meaning | Example |
|---|---|---|
(s) | Solid | NaCl(s) – solid salt |
(l) | Liquid | H₂O(l) – liquid water |
(g) | Gas | CO₂(g) – carbon dioxide |
(aq) | Aqueous (dissolved in water) | NaCl(aq) – salt solution |
Check Your Understanding
Recall
a) Name the three states of matter.
Answer:
Solid, Liquid and Gas
b) Describe how particles move in a solid.
Answer:
The particles in a solid vibrate in fixed positions but do not move from place to place
c) Why do gases fill their containers completely?
Answer:
The particles in a gas move freely in all directions and spread out until they fill all the space available
d) What happens to particle movement when temperature increases?
Answer:
When the temperature increases, particles gain more energy, move faster and vibrate more strongly
Apply Your Knowledge
e) Explain why a liquid has a fixed volume but not a fixed shape.
Answer:
In a liquid, the particles are close together so the forces between them keep the volume fixed. However, the particles can move and slide past each other, so the liquid takes the shape of its container
f) Describe what happens to gas pressure when a gas is heated in a sealed container.
Answer:
When a gas is heated, the particles move faster and collide with the walls of the container more often and with more force. This increases the pressure inside the container
Challenge Yourself
g) Explain one limitation of the particle model.
Answer:
The particle model shows particles as solid spheres, but real atoms and molecules are not perfect spheres, and the model does not show the forces between them
More Practice
Try to answer these practice questions from the TikTok videos on your own, then watch the videos to see how well you did!