Chemical tests for ions
Lajoy Tucker
Teacher

Introduction and Definition
Chemical tests allow chemists to identify ions in unknown samples.
Tests can be used to identify the positive metal ions (cations) and negative ions (anions) that make up ionic compounds.
Test for Metal Ions (Cations) Using Sodium Hydroxide Solution
Sodium hydroxide solution (NaOH) is added to metal-ion solutions to form insoluble metal hydroxide precipitates.
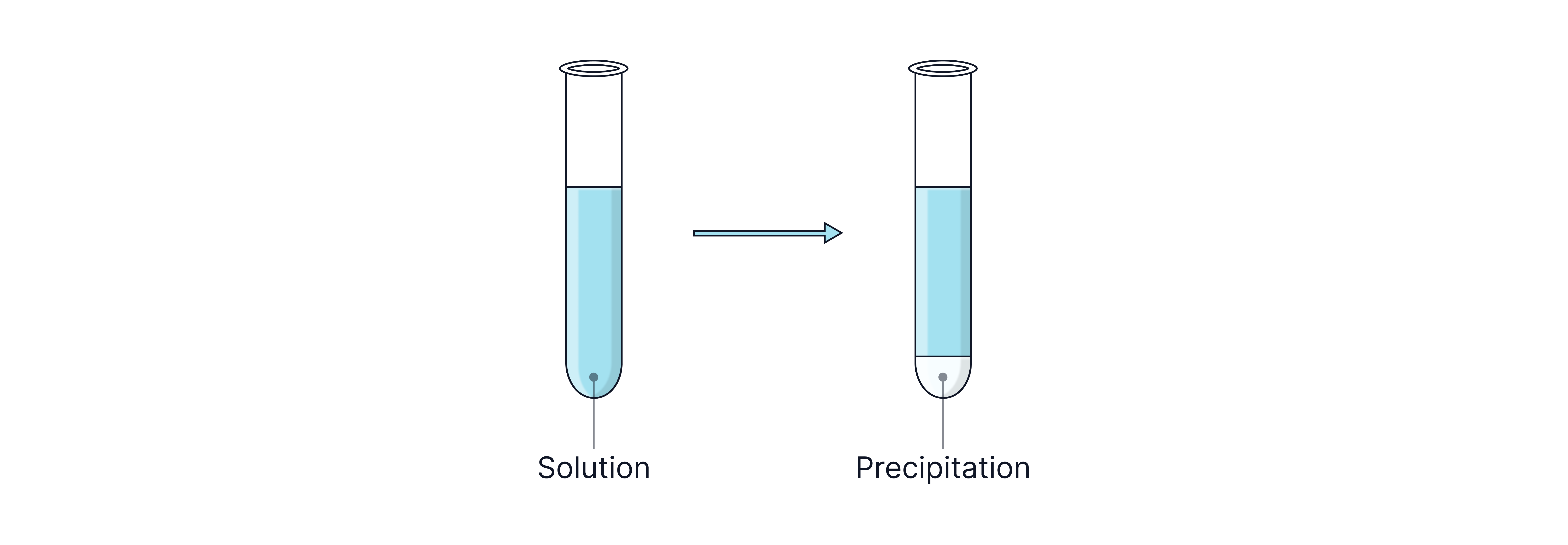
Metal Ions Forming White Precipitates
Ion | Observation with limited NaOH | Identity of precipitate | Observation with Excess NaOH |
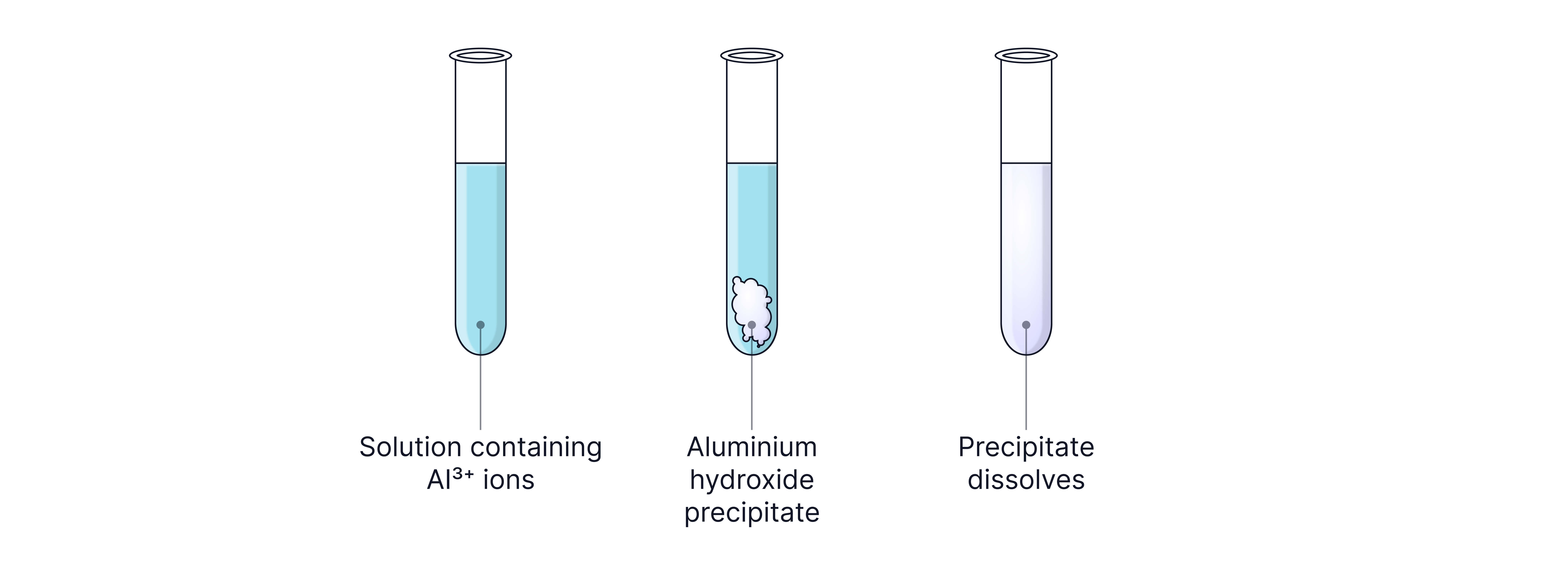
Al³⁺ | White precipitate | Aluminium hydroxide | Precipitate dissolves |
Ca²⁺ | White precipitate | Calcium hydroxide | Precipitate does not dissolve |
Mg²⁺ | White precipitate | Magnesium hydroxide | Precipitate does not dissolve |
All three ions above form white metal hydroxide precipitates with sodium hydroxide.
The precipitate will dissolve when excess sodium hydroxide is formed.
Metal Ions Forming Coloured Precipitates
Transition metal ions form coloured precipitates.

Ion | Observation with limited NaOH | Identity of precipitate | Observation with Excess NaOH |
Cu²⁺ | Blue precipitate | Copper (II) hydroxide | Precipitate does not dissolve |
Fe²⁺ | Green precipitate | Iron (II) hydroxide | Precipitate does not dissolve |
Fe³⁺ | Brown precipitate | Iron (III) hydroxide | Precipitate does not dissolve |
Ionic equations
Metal hydroxide formation can be shown using ionic equations. The number of hydroxide ions required for balancing is equal to the charge of the metal ion. The state symbols show that a solid precipitate (s) is formed from two aqueous solutions (aq)
Worked example
A white precipitate dissolves in excess NaOH. Which ion is present?
Test for negative ions (anions)
Test for Carbonates (CO₃²⁻)
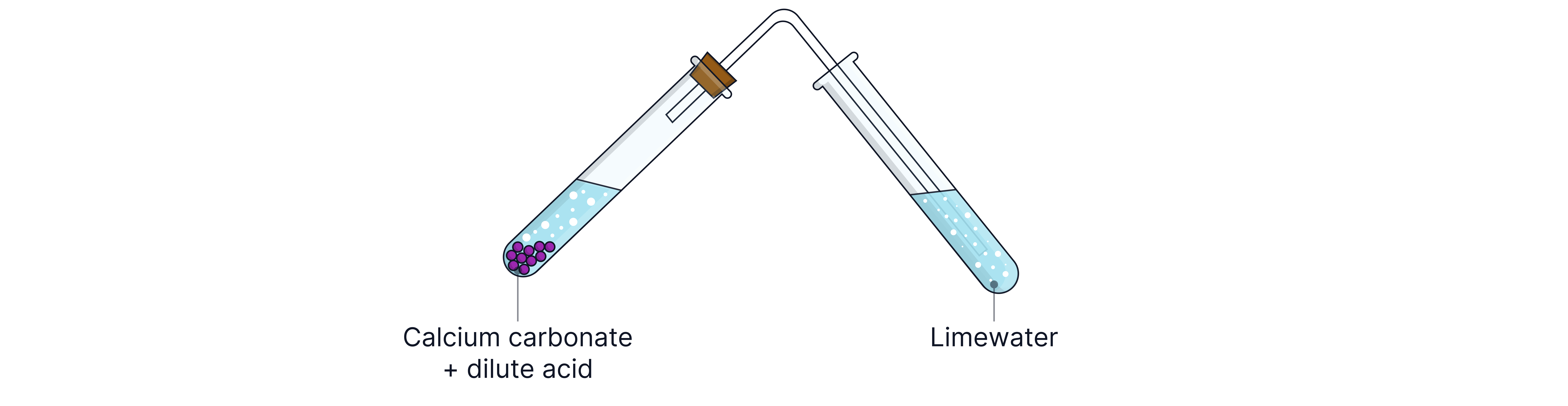
Carbonates react with dilute acids to produce carbon dioxide gas.
Test Method
Add dilute acid.
Collect gas and bubble through limewater.
Positive Result
Limewater turns milky/cloudy.
Example Equation
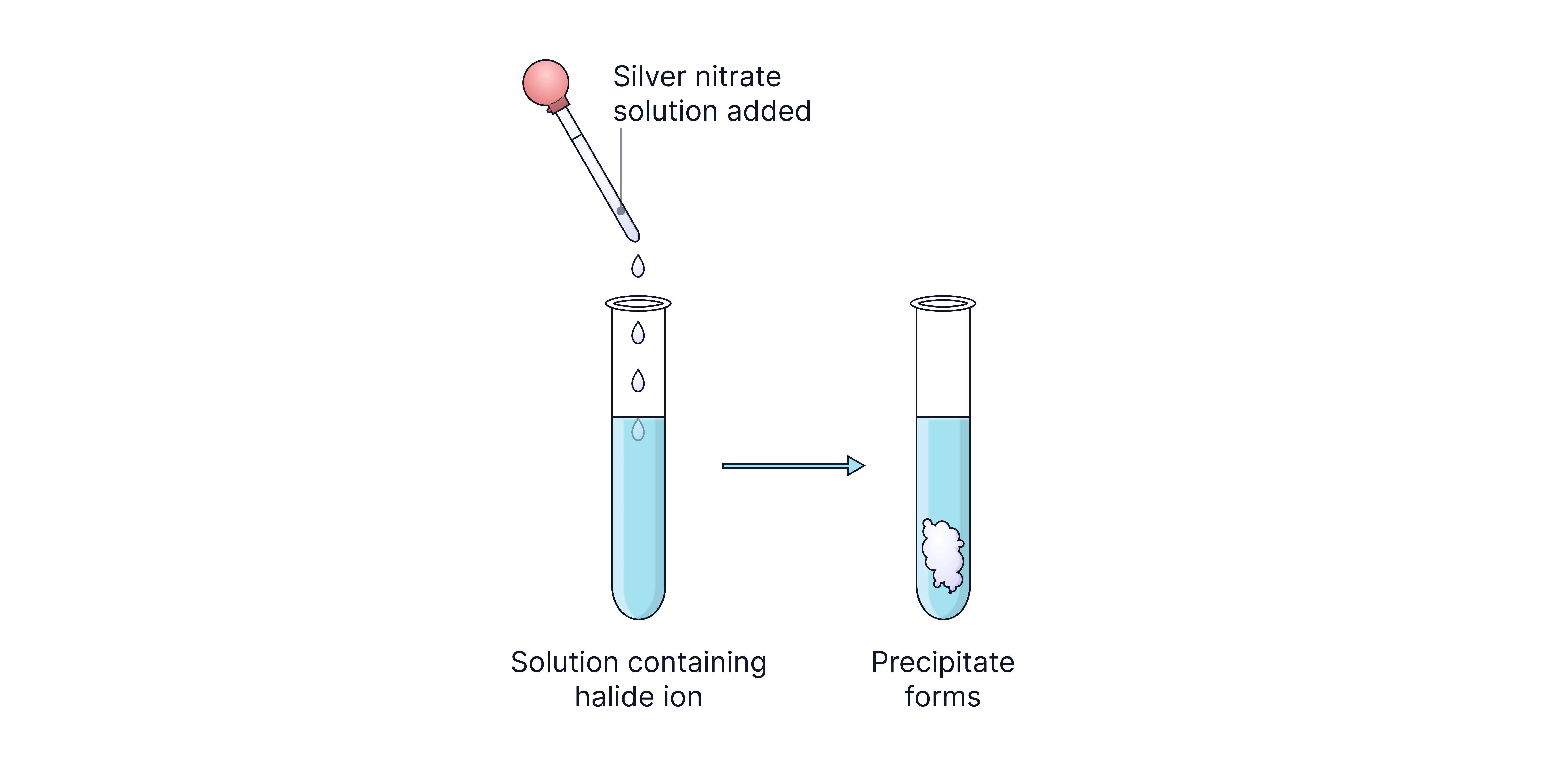
Test for Halide Ions (Cl⁻, Br⁻, I⁻)
Halides react with silver nitrate solution in the presence of dilute nitric acid. The colour of the precipitate formed identifies which ion was present in the sample.
Example equation
Ion | Precipitate Colour |
Cl⁻ | White |
Br⁻ | Cream |
I⁻ | Yellow |
Test for Sulfate Ions (SO₄²⁻)
Sulfate ions form a white precipitate with barium chloride solution in the presence of dilute hydrochloric acid.
Equation

Check for Understanding
Recall
a. What reagent is used to test for metal ions using the hydroxide test?
Answer:
a. Sodium hydroxide solution
b. What colour is the precipitate formed with ions when sodium hydroxide solution is added?
Answer:
b. Blue
c. What colour precipitate is formed when sulfate ions react with barium chloride solution (in the presence of dilute hydrochloric acid)?
Answer:
c. White
Application
d. A white precipitate forms when sodium hydroxide is added and dissolves in excess. Identify the metal ion present and explain reasoning.
Answer:
d. Aluminum ion ; forms white with ; characteristics of
e. Describe how you would test for halide ions and explain how you would identify chloride, bromide and iodide ions from the results.
Answer:
e. Add dilute nitric acid; then add silver nitrate solution; chloride gives white precipitate; bromide gives cream precipitate; iodide gives yellow precipitate.
f. A green precipitate forms when sodium hydroxide is added to a solution but nothing happens when hydrochloric acid is added. However, a cream precipitate forms with addition of silver nitrate. Identify the positive and negative ions present and hence, name the compound.
Answer:
f. Positive ion: Fe²⁺ (green precipitate with NaOH); negative ion: Br⁻ (cream precipitate with silver nitrate). The compound is iron (II) bromide.
Challenge
g. Explain why hydrochloric acid is unsuitable for use as an acid to remove other anions from a solution before the halide test?
Answer:
g. Hydrochloric acid would dissociate into H⁺ and Cl⁻ ions. The Cl⁻ ions from the acid would produce a false positive.
