Flame emission spectroscopy
Lajoy Tucker
Teacher

Contents
What Is Flame Emission Spectroscopy?
Flame emission spectroscopy is an instrumental method used to detect and identify metal ions in a solution.
When a sample is heated in a flame, the metal ions emit light.
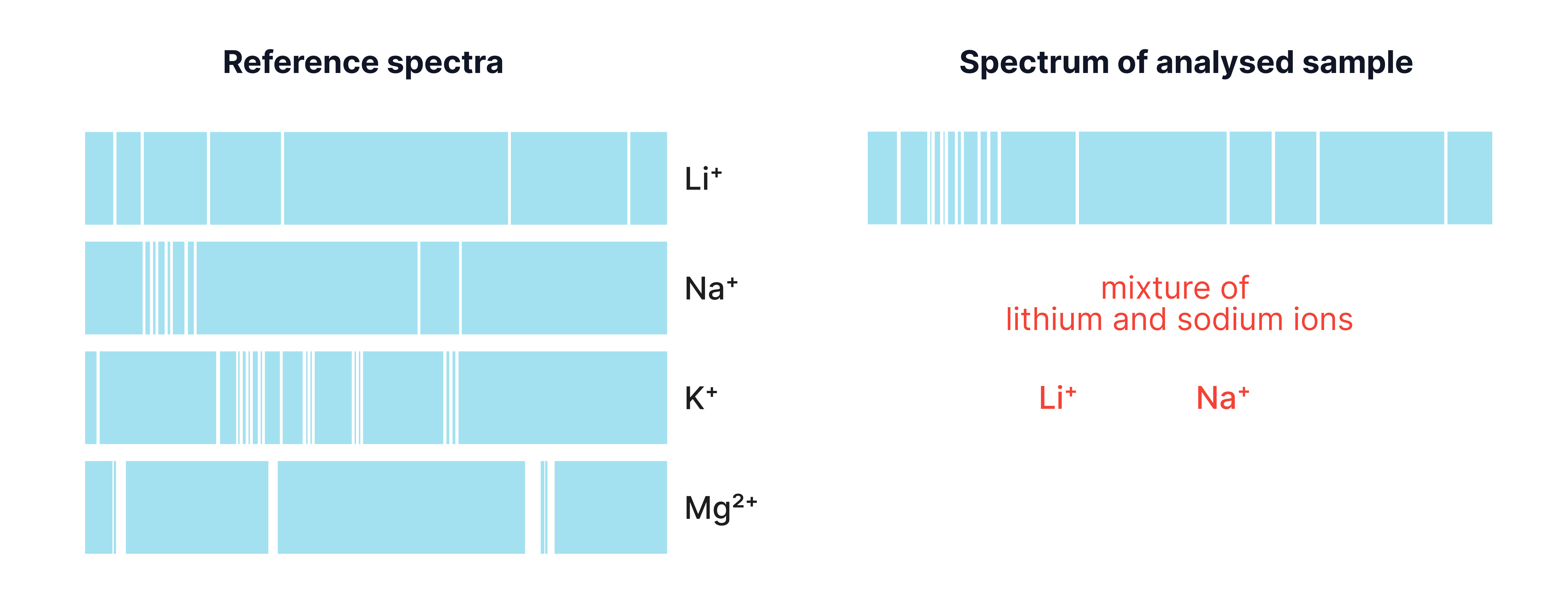
This light is passed through a spectroscope, which produces a line spectrum – a pattern of coloured lines.
Each metal ion produces a unique line spectrum, which can be used to:
Identify which metal ions are present
Measure their concentration
Using Line Spectra to Identify Metal Ions
Each metal ion has a unique pattern of lines at specific wavelengths.

To identify a metal ion:
Compare the positions of the lines with a reference spectrum.
If they match exactly, the metal ion is present.
Advantages Over Flame Tests
Flame Tests | Flame Emission Spectroscopy |
Mixtures give unclear results (flame colours are masked) | Can analyse mixtures of ions |
Qualitative (colour only) | Quantitative (gives concentration) |
Subjective (colour judgement) | Objective (instrument-based) |
Low sensitivity | Detects very low amounts |
Compared with traditional chemical tests, instrumental methods such as flame emission spectroscopy are:
More accurate – give precise results
More sensitive – detect very small amounts of substances
More rapid – produce results quickly and can analyse multiple samples efficiently
These advantages make them widely used in industry, environmental testing and forensic science.
Check for Understanding
Recall
a. What is produced when light from heated metal ions is passed through a spectroscope?
Answer:
a. A line spectrum
b. Why are line spectra useful for distinguishing between metal ions?
Answer:
b. Each metal ion produces a unique pattern of lines at specific wavelengths
c. State two advantages of flame emission spectroscopy over traditional flame tests.
Answer:
c. More accurate; more sensitive; more rapid (any two)
Application
d. Explain how spectrum can be used to identify a metal ion in a sample.
Answer:
d. Compare positions of lines with a reference spectrum; exact match confirms identity.
e. Explain why flame emission spectroscopy is suitable for analysing mixture of metal ions.
Answer:
e. Each metal ion produces its own unique set of lines; lines from different ions appear together; can identify multiple ions in one sample.
f. Explain why flame emission spectroscopy is described as both sensitive and accurate.
Answer:
f. Sensitive: detects very small amounts; accurate: gives precise , reliable measurement.
Challenge (HT)
g. Explain how flame emission spectroscopy can be used not only to identify a metal ion but also to determine its concentration in a solution.
Answer:
g. Intensity (brightness) of spectral lines depends on concentration; higher concentration produces stronger emission; compare intensity to calibration data/known standards to determine concentration.
Summary
Flame emission spectroscopy is an instrumental method for identifying metal ions and measuring their concentration.
It produces a line spectrum, unique to each metal.
It is accurate, sensitive and rapid, and works well for mixtures.
Students must be able to interpret simple spectra using provided data.