Flame tests
Lajoy Tucker
Teacher

Contents
What Are Flame Tests?
Flame tests are a simple method used to identify metal ions (cations) in compounds.
Many metal ions produce distinctive flame colours when heated strongly.
These colours can be used as a quick, qualitative test.
How a Flame Test Works
1. A nichrome wire loop is cleaned in hydrochloric acid and then placed in a blue Bunsen flame until the flame does not change colour
2. The loop is dipped into the sample (solid or solution).
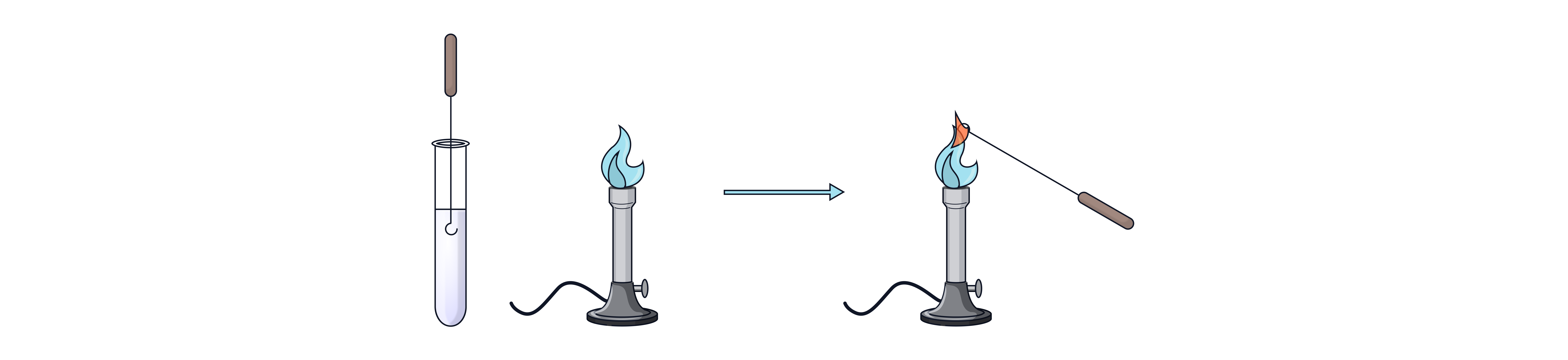
3. The loop is held in the flame again.
4. The colour observed indicates which metal ion is present.
The flame colour is produced when electrons in the metal ions absorb heat, jump to higher energy levels, and then release light when they fall back again.
Flame Colours for Common Metal Ions

Metal Ion (Cation) | Flame Colour |
Lithium (Li⁺) | Crimson red |
Sodium (Na⁺) | Yellow |
Potassium (K⁺) | Lilac |
Calcium (Ca²⁺) | Orange-red |
Copper (Cu²⁺) | Green |
Mixed Samples
Cleaning the wire with hydrochloric acid is important to remove any other ions that may be present which may make the flame colour difficult to interpret.
If a sample contains more than one metal ion, some flame colours may be hidden (masked) or unclear.
Further tests may be needed (e.g. flame emission spectroscopy or precipitation reactions using sodium hydroxide solution).
Check your Understanding
Recall
a. What are flame tests used to identify?
Answer:
a. Metal ions
b. Why is the nichrome wire cleaned with hydrochloric acid?
Answer:
b. Remove contamination / avoid false colours.
c. State the flame colour for sodium ions.
Answer:
c. Yellow
Application
d. A sample produces a crimson flame. Identify the metal ion present.
Answer:
d. Lithium ion
e. A mixture contains sodium and calcium ions but only a yellow flame is seen, Explain why.
Answer:
e. Sodium produces intense yellow; mask calcium's colour.
Challenge (HT)
f. Explain why flame tests are not reliable for mixtures and suggest a more accurate technique.
Answer:
f. Colours overlap/masked; this is subjective; flame emission spectroscopy would be a more accurate technique.