Chromatography
Lajoy Tucker
Teacher

Contents
Chromatography
Chromatography is a technique used to separate mixtures and help identify substances within them.
It works because different substances move through a system at different speeds.
Chromatography involves:
- A stationary phase – does not move (e.g. chromatography paper)
- A mobile phase – moves (e.g. solvent such as water or ethanol)
The substances in the mixture separate because they distribute differently between the two phases.
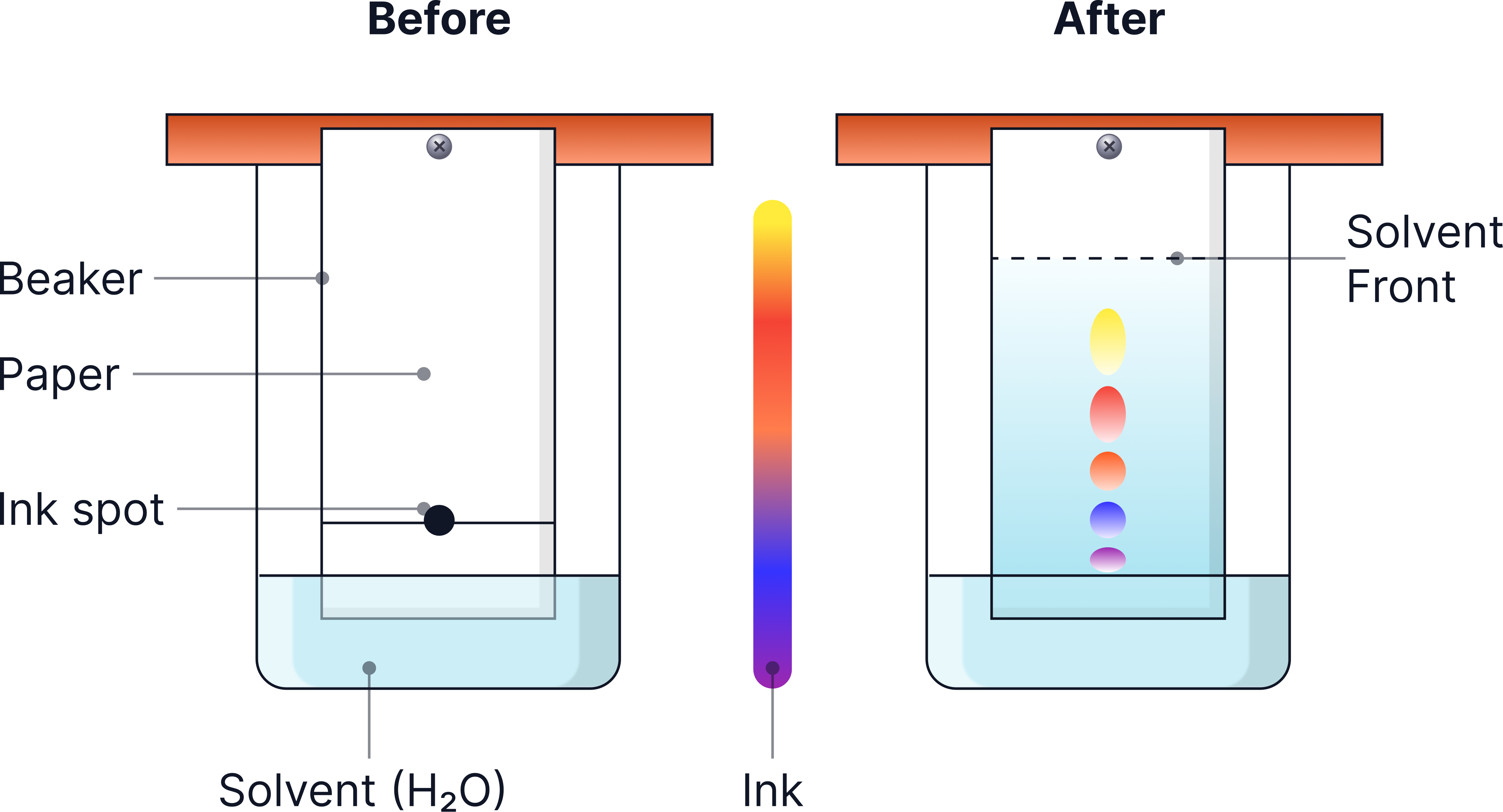
How Paper Chromatography Works
- A small spot of the mixture is placed on the baseline of chromatography paper.
- The paper is placed in a solvent, with the spot above the solvent level.
- The solvent moves up the paper, carrying the substances with it.
- Substances that are more soluble in the solvent (attracted to the mobile phase) travel further.
- Substances that are less soluble in the solvent travel shorter distances.
As a result, the mixture separates into spots at different heights.
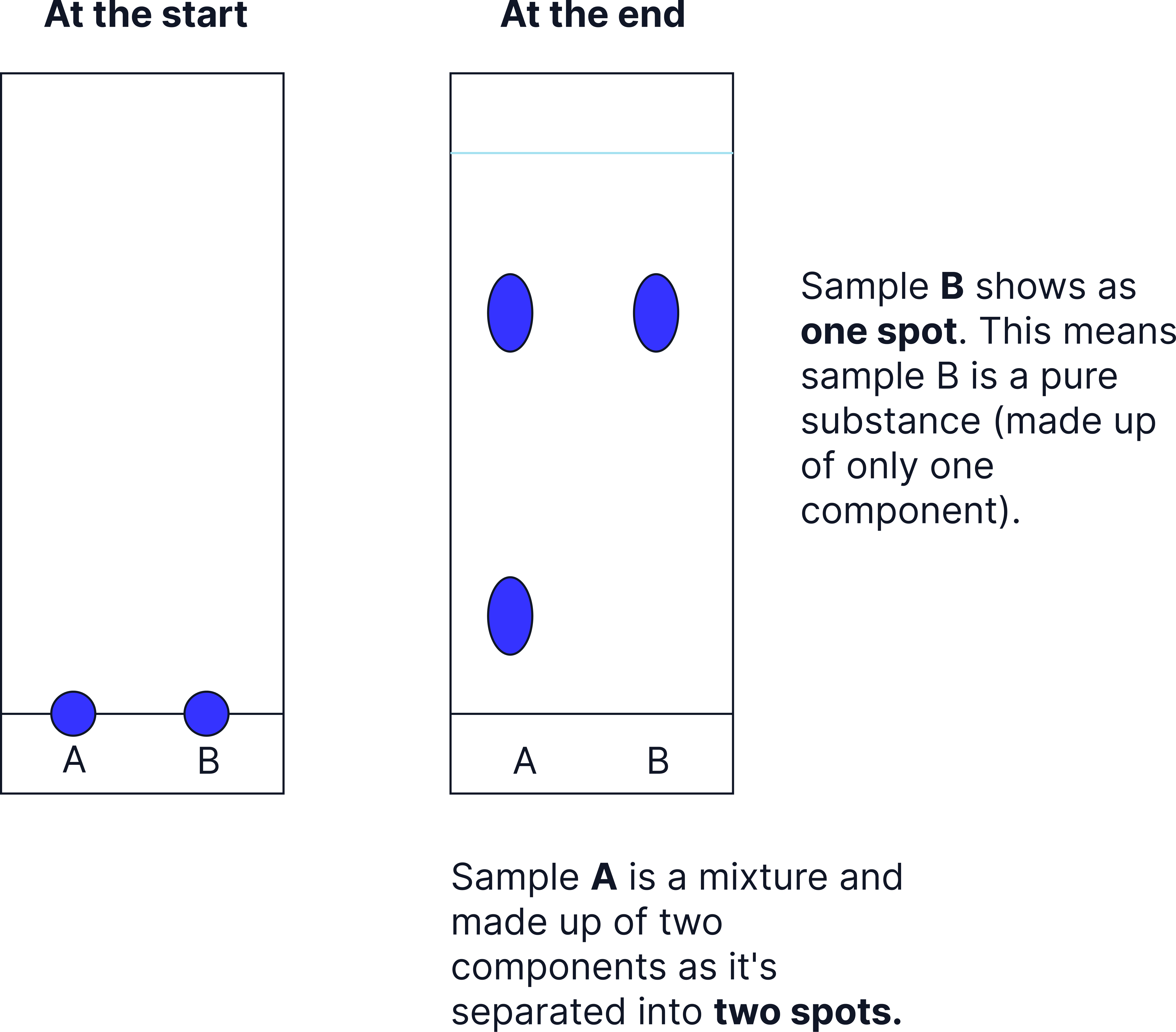
Identifying Pure vs Impure Substances
Chromatography is a helpful test of purity:
- A pure substance will produce one spot
- A mixture will produce two or more spots, because it contains multiple components
Retention Factor (Rf)
Rf values allow a calculation of how far a component travels compared to the solvent front.
Rf = Distance travelled by substance / Distance travelled by solvent.
The distance is always measured from the baseline where the sample was spotted.
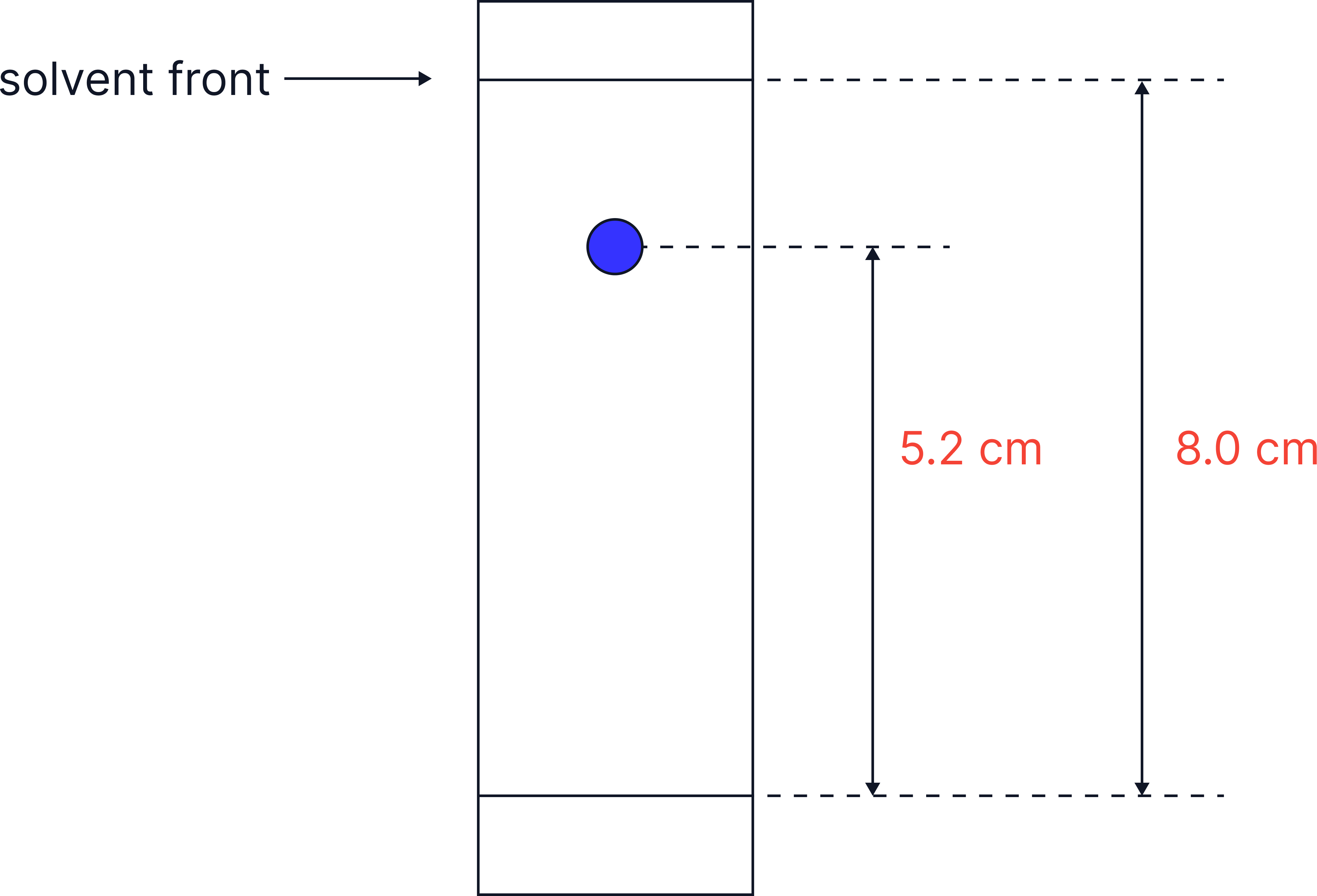
If the solvent front moved 8.0 cm and a spot moved 5.2 cm, then:
Rf = 5.2/8.0 = 0.60
Interpreting Rf values
- Substances that are less soluble in the solvent (mobile phase) travel shorter distances → lower Rf.
- Substances that are more soluble in the solvent (mobile phase) travel further → higher Rf.
As Rf values depend on solubility, the same substance will have different Rf values in different solvents.
Identifying a Substance in a Mixture
To identify whether a substance is present in a mixture, a reference sample (a known pure substance) is run on the same chromatogram.
How to compare a mixture to a reference:
- Place both the mixture and the reference sample on the same baseline.
- Run the chromatography under identical conditions.
A substance in the mixture is present if it produces a spot at exactly the same height as the reference/it has the same Rf value.
If the spots do not match, the reference substance is not present in the mixture.
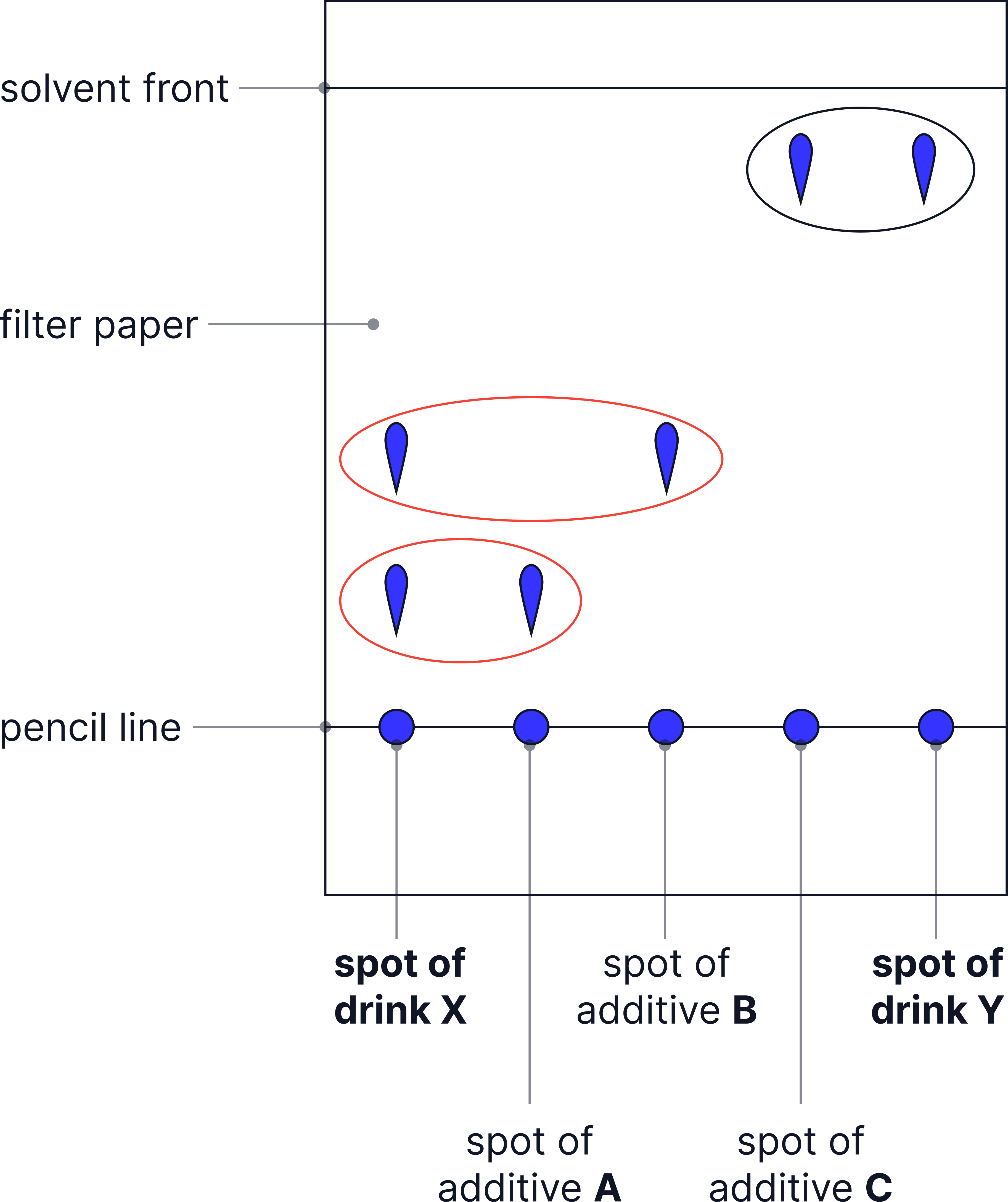
Example:
Below, two drinks X and Y are tested for different additives.
Drink X has spots that line up with A and B so contains those additives.
Drink Y has a spot that lines up with additive C
Check Your Understanding
Recall
a) What is the stationary phase in paper chromatography?
Answer:
Chromatography paper
b) What does a pure substance produce on a chromatogram?
Answer:
One spot
c) State the formula used to calculate the Rf value.
Answer:
Rf = distance travelled by substance ÷ distance travelled by solvent
Application
d) The solvent front moves 10.0 cm and a spot moves 6.5 cm. Calculate the Rf value.
Answer:
Rf = 6.5 ÷ 10.0 = 0.65
e) Explain why a substance may have different Rf values in different solvents.
Answer:
Rf depends on solubility; substances distribute differently between mobile and stationary phases; different solvents change solubility; therefore distance travelled changes
f) Describe how you would use a reference sample to identify a substance in a mixture.
Answer:
Place mixture and reference on same baseline; run under identical conditions; compare spot heights/Rf values; matching height or same Rf indicates reference is present in unknown sample
Summary
- Chromatography separates mixtures using a stationary phase and a mobile phase.
- Substances separate because they move at different speeds depending on how they distribute between the phases. Pure substances → one spot. Mixtures → multiple spots.
- Rf values help identify substances and compare results.
- Chromatograms can be interpreted by analysing spot positions and calculating Rf values.
More Practice
Try to answer the practice question from the TikTok video on your own, then watch the video to see how well you did!