Concentration of solutions
Lajoy Tucker
Teacher

Introduction
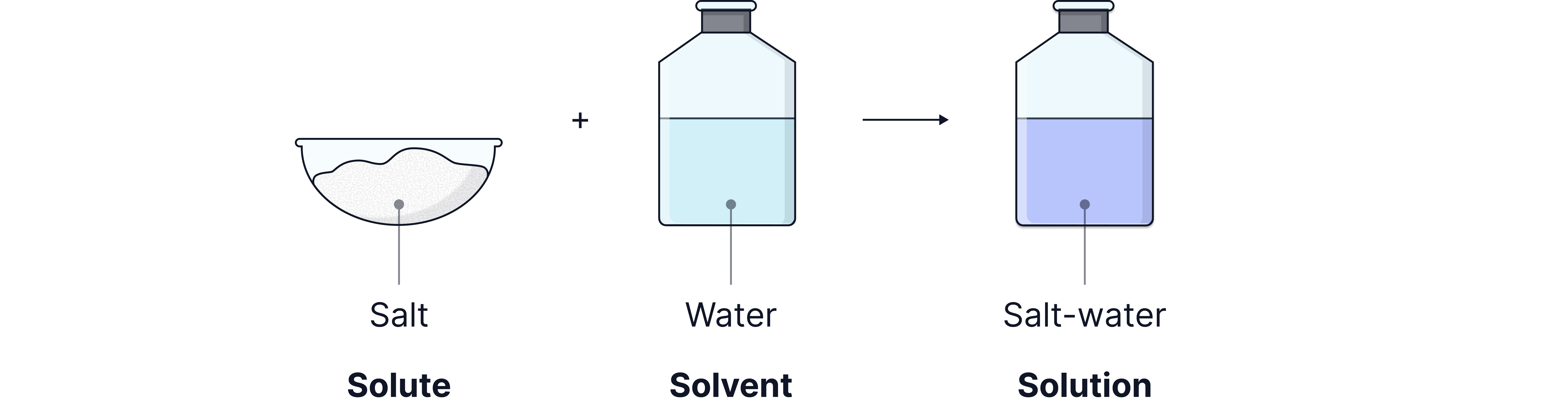
Many chemical reactions happen in solutions, where a solute (a solid substance) is dissolved in a solvent (often water) to form a solution.
For example, when salt (sodium chloride) dissolves in water, it forms a salt solution.
Key terms:
Solute – the substance that is dissolved.
Solvent – the liquid that dissolves the solute.
Solution – the mixture formed when the solute dissolves in the solvent.
Measuring Concentration
The concentration of a solution tells us how much solute is dissolved in a certain volume of solution.
It can be measured in grams per cubic decimetre ()
Formula:
Converting Units
= 1000 = 1 litre.
If the volume is given in , it must be converted to before using the formula.
Example
A solution contains 10 g of sodium chloride in 250 cm³ of solution.
Calculate the concentration in g/dm³.
Answer:
Volume in dm³ = 250 ÷ 1000 = 0.25
Concentration = 10 ÷ 0.25 = 40 g/dm³
Rearranging Formula
Mass and volume can be also calculated by rearranging the formula:
Mass (g) = Concentration (g/dm³) x Volume dm³
Example:
A solution has a concentration of 20 g/dm³.
If you have 0.5 dm³ of this solution, what mass of solute does it contain?
Answer:
Mass = concentration × volume
= 20 × 0.5 = 10 g
The mass of solute and volume of solution are directly proportional to concentration:
Increasing the mass of solute (in the same volume) → increases concentration.
Increasing the volume of solution (using the same mass of solute) → decreases concentration.
This means doubling the solute doubles the concentration, but doubling the volume halves it.
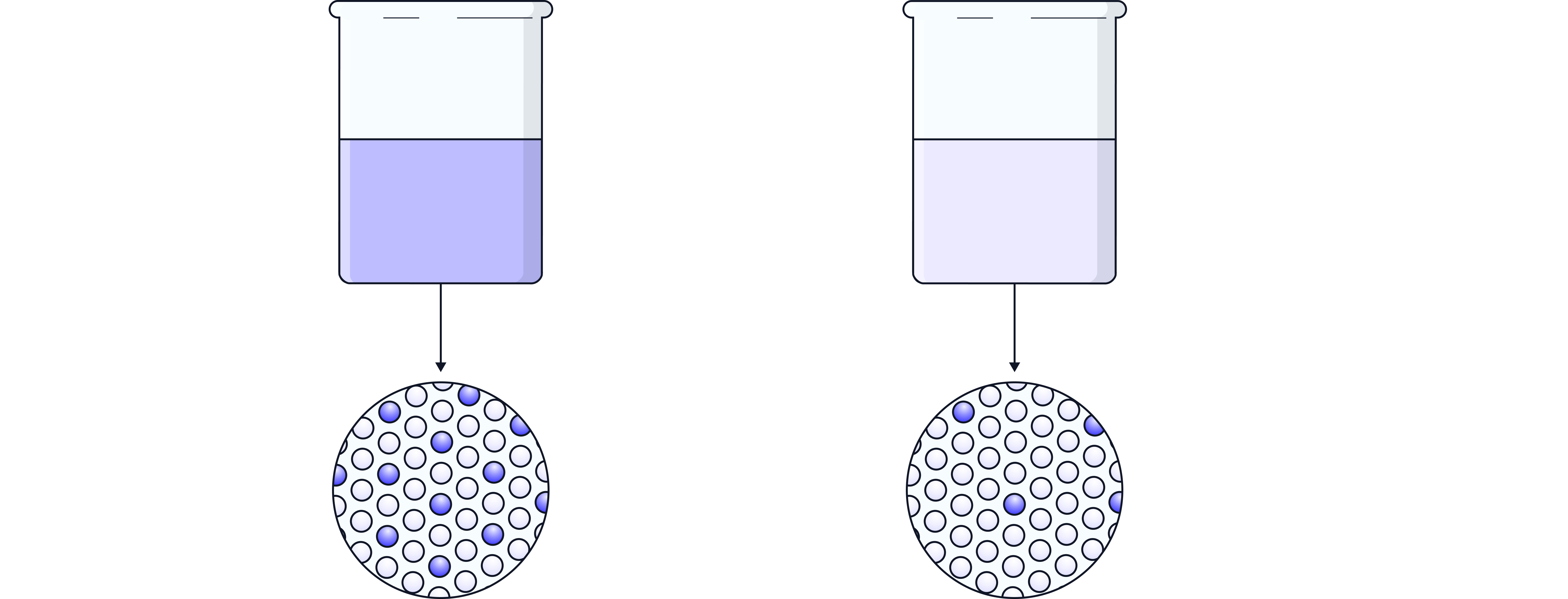
Example:
Explain what happens to the concentration if the same 10 g of solute is dissolved in 2 dm³ instead of 1 dm³ of water.
Answer:
The concentration halves because the solute is spread through twice the volume.
Original: 10 ÷ 1 = 10 g/dm³
New: 10 ÷ 2 = 5 g/dm³
Check for Understanding
Recall
a) What is meant by a solute?
Answer:
a) A solute is the substance that is dissolved in a solvent.
b) What is meant by a solution?
Answer:
b) A solution is the mixture formed when a solute dissolves in a solvent.
c) State the formula used to calculate concentration in g/dm³.
Answer:
c) Concentration = mass ÷ volume (c = m / V).
Application
d) A solution contains 12 g of solute dissolved in 0.40 dm³ of solution.
Calculate the concentration in g/dm³.
Answer:
d)
c = m / V
c = 12 ÷ 0.40
c = 30 g/dm³
e) A solution has a concentration of 25 g/dm³ and a volume of 0.20 dm³.
Calculate the mass of solute present.
Answer:
e)
Mass = concentration × volume
Mass = 25 × 0.20
Mass = 5 g
f) A solution contains 8 g of solute dissolved in 200 cm³ of solution.
Calculate the concentration in g/dm³.
Answer:
f)
Convert volume: 200 cm³ = 0.200 dm³
c = m / V
c = 8 ÷ 0.200
c = 40 g/dm³
Summary
Concentration tells us how much solute is dissolved per dm³ of solution.
Formula: c = m / V (Concentration = Mass ÷ Volume).
Always convert cm³ → dm³ (÷1000).
Increasing mass of solute raises concentration; increasing volume lowers it.
Be confident rearranging the formula to find mass, volume or concentration.