Limiting reactants (HT only)
Lajoy Tucker
Teacher

Understanding Limiting and Excess Reactants
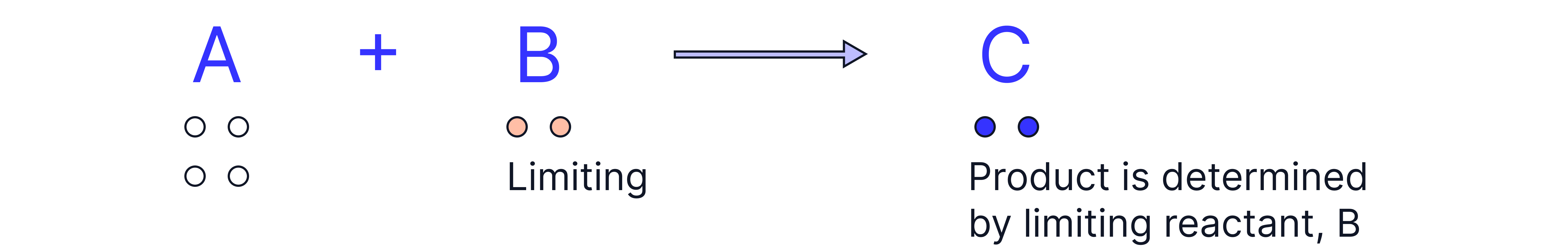
In many reactions, one reactant is deliberately added in excess to ensure the other is completely used up.
The reactant that is used up first is called the limiting reactant because it limits the amount of product that can be formed.
Once the limiting reactant is all used, the reaction stops – any other reactant left over is in excess.
Worked Examples
Worked Example 1
Hydrochloric acid reacts with sodium hydroxide:
HCl + NaOH → NaCl + H₂O
Given:
36.5 g of HCl
80.0 g of NaOH
Step 1 – Calculate moles = mass / Mᵣ
Mᵣ of HCl = 36.5 ⇒ Moles of HCl = 36.5 ÷ 36.5 = 1.00
Mᵣ of NaOH = 40.0 ⇒ Moles of NaOH = 80.0 ÷ 40.0 = 2.00
Step 2 – Compare ratio
Equation ratio HCl : NaOH = 1 : 1
→ HCl is limiting (fewer moles than needed)
→ NaOH is in excess
Step 3 – Effect on product yield (determined by limiting reactant)
Because 1 mol of HCl produces 1 mol of NaCl, only 1.00 mol of NaCl can form.
Mass of NaCl = 1.00 × 58.5 = 58.5 g
Result: The maximum yield is 58.5 g of sodium chloride – limited by HCl.
Worked example 2 (challenging)
When the reactants don’t react in a 1:1 ratio, the number of moles can not be directly compared. Instead, the molar ratios from the balanced symbol equation are used to determine how much of each reactant is needed.
Sodium fluoride is formed when 4.60 g of sodium reacts with 5.70 g of fluorine.
Use calculations to show which reactant is in excess (4).
Calculate the maximum mass of sodium fluoride that can be formed (2).
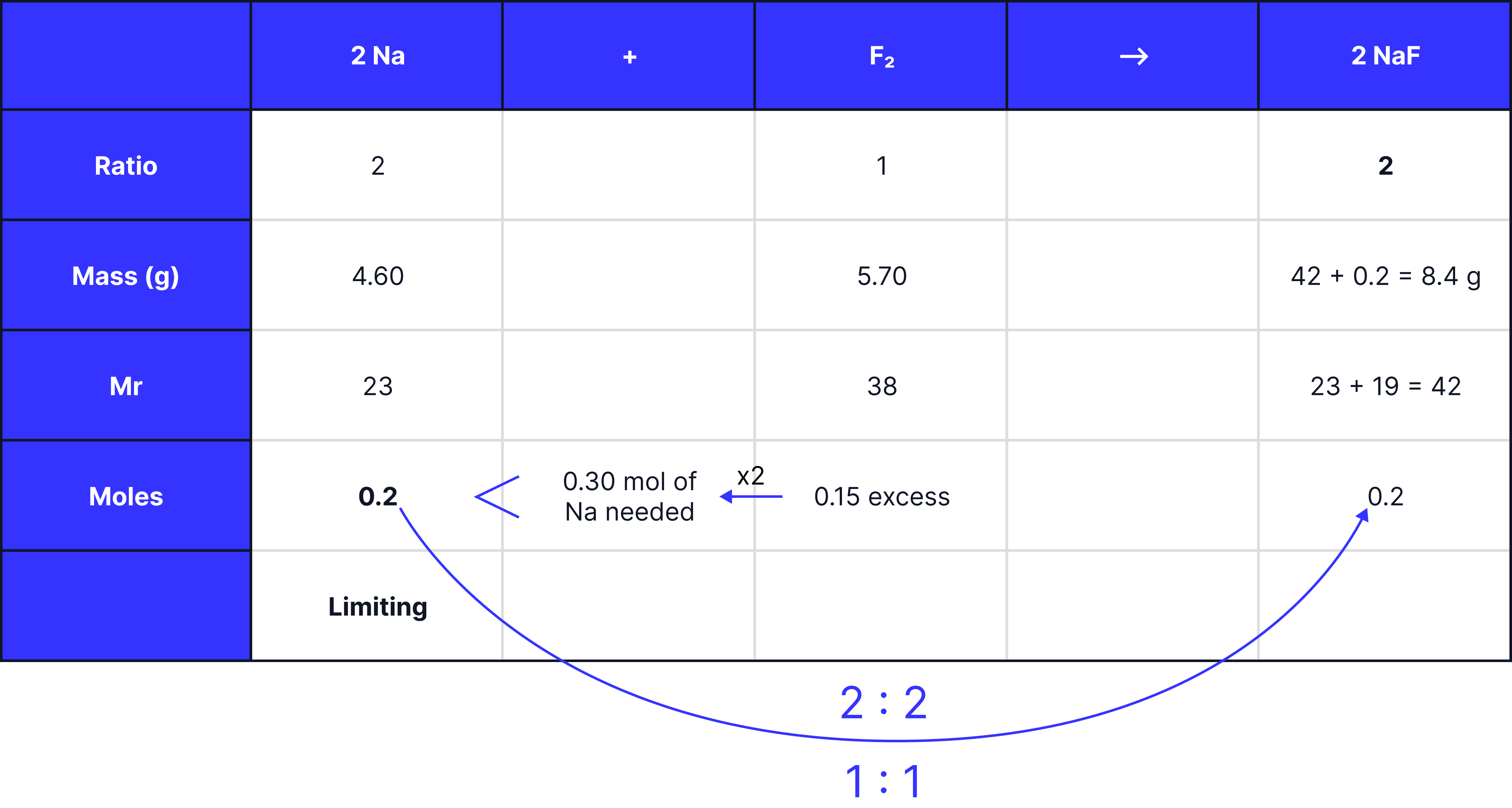
1 . Calculate the moles of each reactant using moles =
2. Multiply the moles of the reactant with the smaller coefficient () by the larger coefficient ().
3. Identify whether you have enough (excess) or not enough (limiting).
4. Use the moles of the limiting reactant to determine the moles and mass of the product.
Check for Understanding
Recall
a) What is meant by a limiting reactant?
Answer:
a) The reactant that is used up first / the reactant that limits the amount of product formed.
b) What is meant by an excess reactant?
Answer:
b) A reactant that remains after the reaction / not completely used up.
Application
c) Why might a chemist deliberately add one reactant in excess during a chemical reaction?
Answer:
c) To ensure the other reactant is completely used up / to make sure the limiting reactant fully reacts.
d) In a reaction between magnesium and oxygen, all the magnesium is used up but oxygen remains.
Which substance is the limiting reactant?
Answer:
d) Magnesium.
e) Before comparing the amounts of reactants in a balanced equation, what quantity must be calculated from the mass of each substance?
Answer:
e) The number of moles.
Challenge (HT)
f) Sodium reacts with fluorine according to the equation:
2Na + F₂ → 2NaF
A student reacts 23 g of sodium (Na) with 19 g of fluorine (F₂).
i) Calculate the number of moles of sodium.
ii) Calculate the number of moles of fluorine.
iii) Use the mole ratio to identify the limiting reactant.
Answer:
f)
i) 23 ÷ 23 = 1 mol Na.
ii) 19 ÷ 38 = 0.5 mol F₂.
iii) Ratio Na : F₂ = 2 : 1.
0.5 mol F₂ requires 1 mol Na, so the reactants are in the correct ratio and both react completely.