Reacting masses
Lajoy Tucker
Teacher

Contents
Introduction & Definitions
Reacting Masses: Calculations used to determine how much of a reactant is required or how much product is formed in a chemical reaction.
or M (relative formula mass or molar mass) :The mass of 1 mole of a substance.
Basic Principles
Main Formulae:
Moles = Mass (g) /
Steps for Reacting Mass Calculations:
1. Write a balanced equation.
2. Find the moles of the known substance using
3. Use mole ratios from the equation to find the moles of the unknown substance.
4. Convert moles to mass using
5. NB: May also have to use c = n/v and/or c = nv for reactions involving solutions.
Why Mole Ratios Matter:
The coefficients in a balanced chemical equation tell you the ratio of moles of reactants and products. These ratios must be used when converting between substances.
Key Assumptions:
Reactions go to completion.
All reagents are pure.
You are calculating the theoretical mass which is the maximum mass you could make (not actual yield).
Worked Examples
Example 1
Determine the maximum mass of aluminum that can be formed from 1024 g of aluminum oxide.
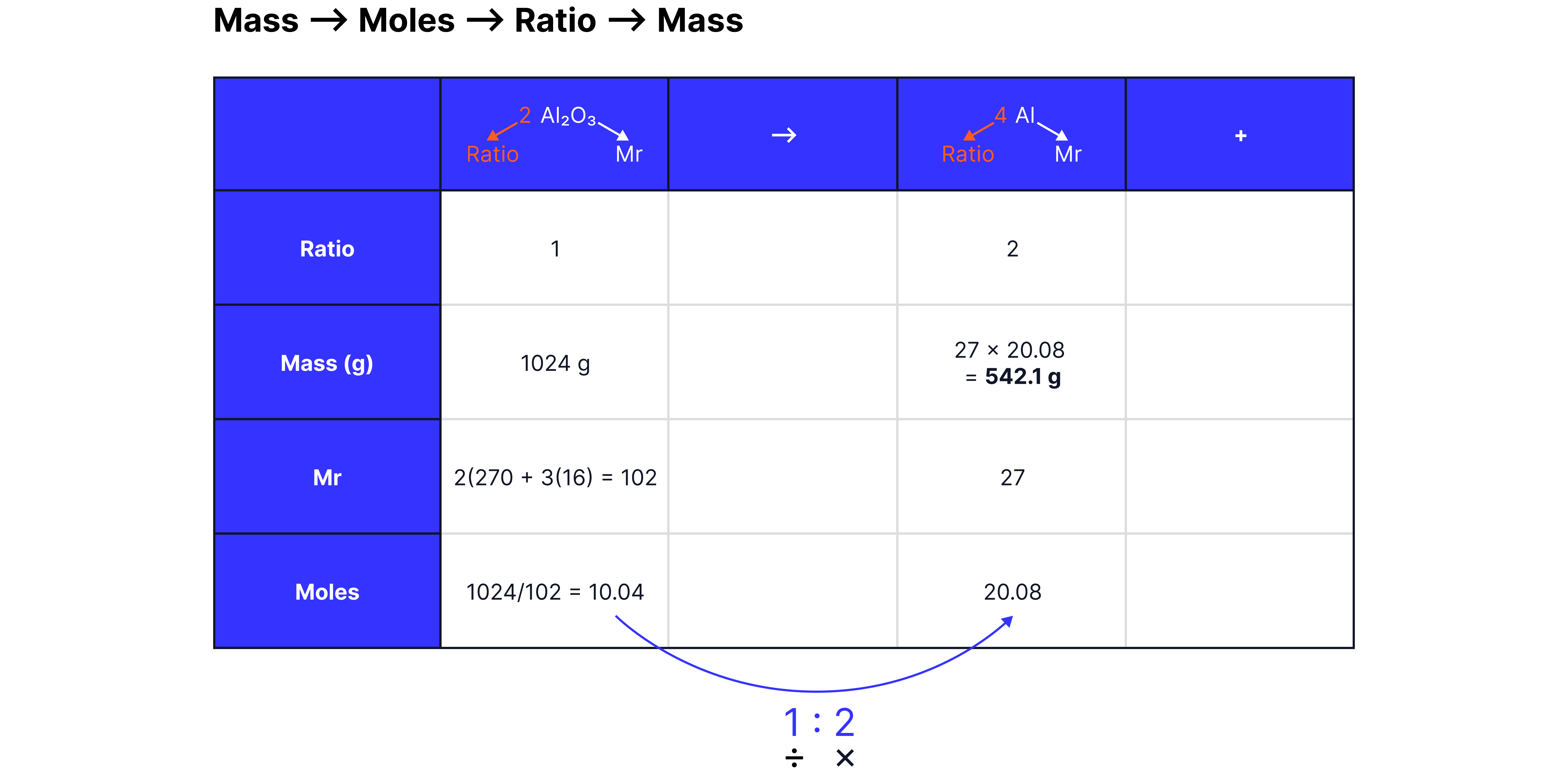
Example 2:
Question: What is the maximum mass of water that can be produced when 6.00 g of hydrogen reacts with oxygen?
Step 1: Moles of H₂:
of = 2
Step 2: Mole ratio H₂: H₂O = 2:2 → 3.00 mol H₂ makes 3.00 mol H₂O
Step 3: Mass of H₂O:
of H₂O = 18
Example 3:
Question: What is the minimum mass of iron required to react completely with 7.0 moles of oxygen gas.
Moles of O2 = 7.0 mol
Mole ratio: : Fe = 3:4 → moles of Fe = 7.0 x 4/3 = 9.33
of Fe = 56
Mass of Fe = 9.33 x 56 = 522.7g = 520g (2 s.f.)
Note - The appropriate number of significant figures for an answer is determined by the least number of significant figures given in the question.
e.g. What is the minimum mass of iron required to react completely with 7.50 moles of oxygen gas.
Check for Understanding
Recall
a) Write the formula used to calculate moles from mass and relative formula mass.
Answer:
a) Moles = Mass ÷ Mr
b) What do the coefficients in a balanced chemical equation tell you?
Answer:
b) They show the ratio of moles of reactants and products in the reaction.
c) What is meant by the theoretical mass of a product?
Answer:
c) The maximum mass of product that can be formed from the reactants.
Application
d) Hydrogen reacts with oxygen according to the equation:
2H₂ + O₂ → 2H₂O
Calculate the maximum mass of water that can be produced when 6.00 g of hydrogen reacts with excess oxygen. (Mr H₂ = 2, Mr H₂O = 18)
Answer:
d)
Moles of H₂ = 6.00 ÷ 2 = 3.00 mol
Ratio H₂ : H₂O = 2 : 2 → 1 : 1
Moles of H₂O = 3.00 mol
Mass = 3.00 × 18
= 54.0 g
e) Iron reacts with oxygen according to the equation:
4Fe + 3O₂ → 2Fe₂O₃
Calculate the minimum mass of iron required to react completely with 7.0 moles of oxygen gas. (Mr Fe = 56)
Answer:
e)
Ratio O₂ : Fe = 3 : 4
Moles of Fe = (7.0 × \frac{4}{3}) = 9.33 mol
Mass = 9.33 × 56
= 522.7 g
= 520 g (2 s.f.)
Challenge (HT)
f) Propane burns in oxygen according to the equation:
C₃H₈ + 5O₂ → 3CO₂ + 4H₂O
Calculate the mass of CO₂ produced when 8.4 g of propane (C₃H₈) burns in excess oxygen. (Mr C₃H₈ = 44, Mr CO₂ = 44)
Answer:
f)
Moles of C₃H₈ = 8.4 ÷ 44 = 0.191 mol
Ratio C₃H₈ : CO₂ = 1 : 3
Moles of CO₂ = 0.191 × 3 = 0.573 mol
Mass of CO₂ = 0.573 × 44
= 25.2 g
≈ 25 g (2 s.f.)
g) What mass of iron(III) oxide is needed to react with 12.0 g of aluminium?
g)
Answer: