Crude oil and fractional distillation
Lajoy Tucker
Teacher

What is Crude Oil?
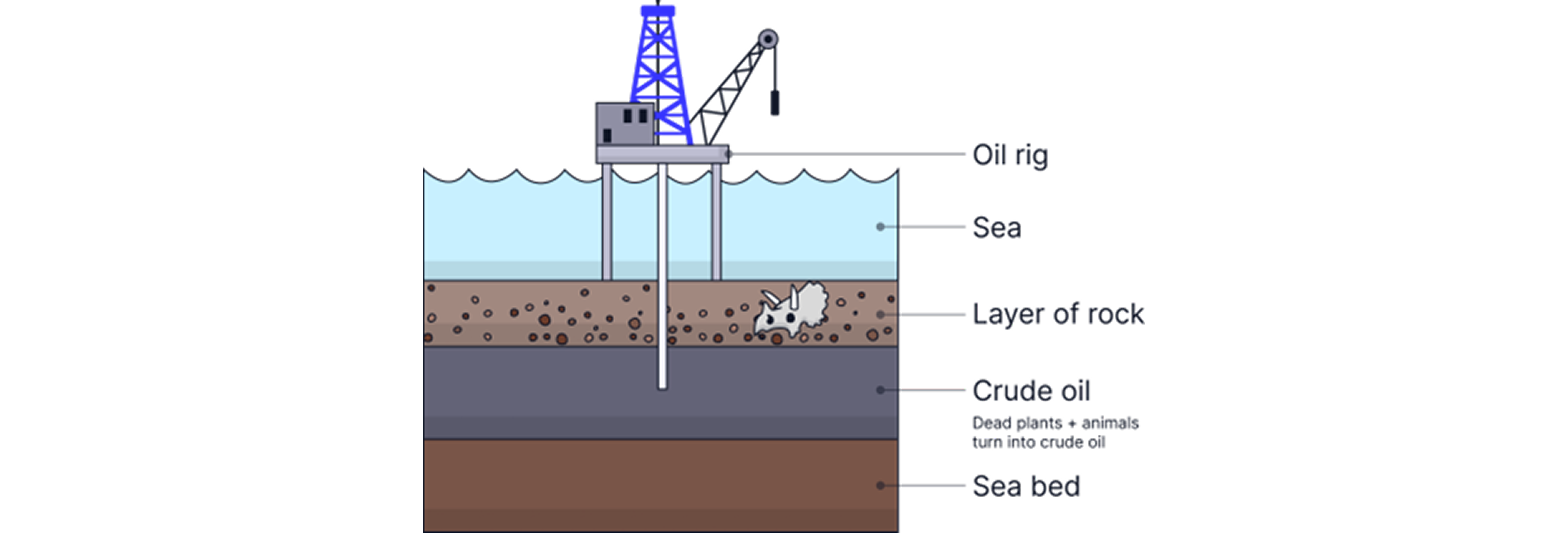
Crude oil is a finite resource found in rocks beneath the Earth’s surface.
It formed millions of years ago from the remains of ancient plankton that were buried in mud and subjected to heat and pressure over long periods in the absence of oxygen.
Because it takes so long to form, crude oil is considered a non-renewable (finite) resource
Composition of Crude Oil
Crude oil is a mixture of a very large number of compounds.
Most of these compounds are hydrocarbons – molecules made up only of hydrogen and carbon atoms.
The hydrocarbons vary in chain length and structure, which gives them different physical properties such as boiling points and viscosity.
Hydrocarbons and Alkanes
The simplest type of hydrocarbon is an alkane.
Alkanes are saturated hydrocarbons, meaning all carbon–carbon bonds are single covalent bonds.
They follow the general formula:
Alkanes can be represented as molecular formulas , structural formulas , or displayed formulas (showing all bonds).
Students should be able to recognise alkanes from these formula types.
| Number of carbon atoms | Number of hydrogen atoms | Displayed formula | Structural formula | Molecular formula | Name of alkane |
| 1 | 4 |  |
Methane | ||
| 2 | 6 |  |
Ethane | ||
| 3 | 8 |  |
Propane | ||
| 4 | 10 |  |
Butane |
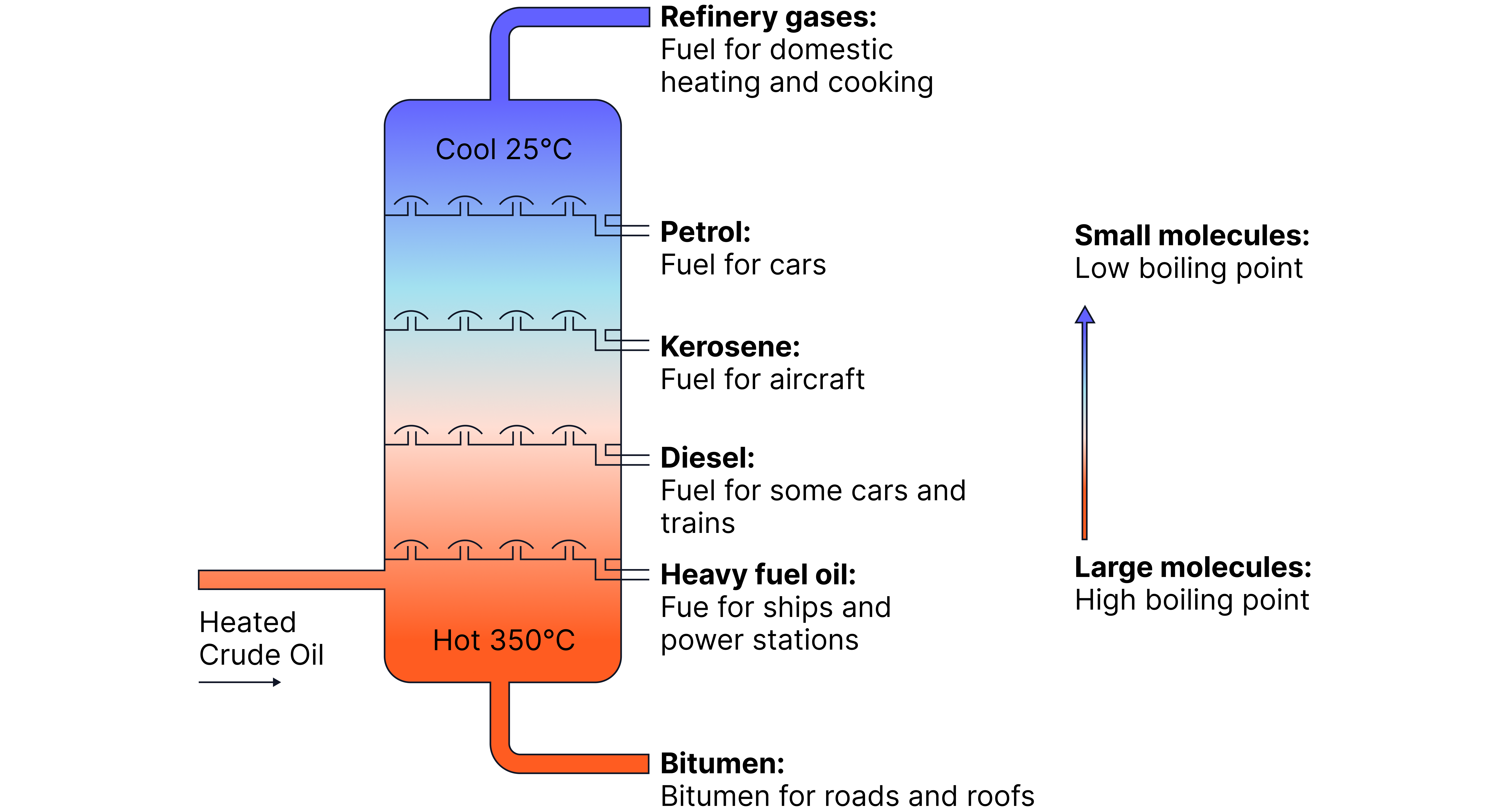
Fractional distillation
Petroleum is a term used for crude oil along with its refined products such as gasoline, diesel, and kerosene
These products can be separated by fractional distillation
Fractional distillation separates crude oil based on the boiling points of components.
Stage | Process Description |
1. Heating | Crude oil is heated until most of it evaporates (boils) into vapour. |
2. Entry to Column | The mixture enters a fractionating column, which is hot at the bottom and cooler at the top. |
3. Condensation | Vapours rise through the column and cool. Each fraction condenses when it reaches a level where the temperature is below its boiling point. |
4. Separation | Longer-chain hydrocarbons (with high boiling points) condense lower down, while shorter-chain hydrocarbons (with low boiling points) condense higher up. |
5. Collection | The condensed liquids are collected from trays at different levels — each is a fraction containing hydrocarbons of similar chain lengths. |
Check for Understanding
Recall
a) What is crude oil?
Answer
Mixture of hydrocarbon
b) State the general formula for alkanes.
Answer
.
c) What property allows separation in fractional distillation?
Answer
Different boiling points.
d) Which fraction has the lowest boiling point?
Answer
Refinery gases.
Application
e) Describe how crude oil is separated in a fractionating column.
Answer
Crude oil heated until vapourised; vapours rise up the column; column has a temperature gradient (hot at bottom, cooler at top); condense at different temperatures.
f) Predict the formula of an alkane with 12 carbon atoms.
Answer
.
g) Explain why refinery gases are collected at the top of the column.
Answer
Lowest boiling points; remain gases; rises highest in the column.
Challenge
h) Why do long-chain alkanes have higher boiling points than short-chain alkanes?
Answer
Long-chain alkanes are larger; this means they have relatively stronger weak intermolecular forces compared to short-chain alkanes; more energy is needed to overcome their intermolecular forces.
Summary
Crude oil is a finite, natural mixture of hydrocarbons formed from ancient plankton.
Most hydrocarbons are alkanes with the formula .
The first four alkanes are methane, ethane, propane, and butane.
Crude oil is separated into fractions by fractional distillation, based on boiling point differences.