Properties of hydrocarbons
Lajoy Tucker
Teacher

Contents
Properties of hydrocarbons
Hydrocarbons are compounds made of hydrogen and carbon atoms only.
Their properties depend on the length of their carbon chains – that is, the size of their molecules.
Property | Definition | Trend with Increasing Molecular Size (Chain Length) |
|---|---|---|
Boiling point | The temperature at which a substance changes from liquid to gas | Increases – larger molecules have stronger intermolecular forces and need more energy to evaporate |
Viscosity | How thick or sticky a liquid is | Increases – longer chains make the liquid flow more slowly |
Flammability | How easily a substance burns | Decreases – larger molecules are harder to ignite and burn less cleanly |
How These Properties Affect Fuel Use
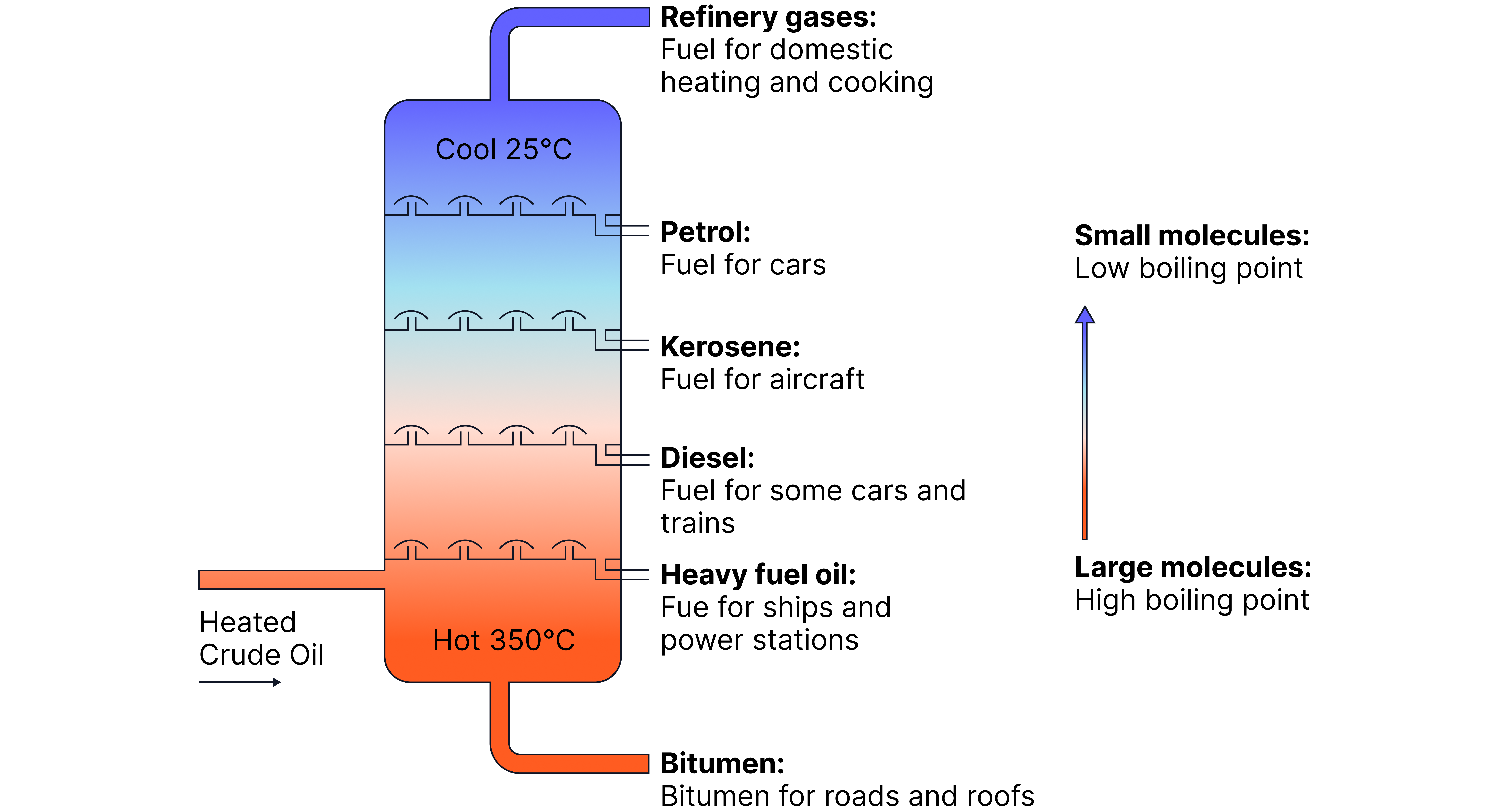
Typical Use | Reason | |
|---|---|---|
Short-chain hydrocarbons (e.g. refinery gases, petrol) | Fuels for cooking, heating, and vehicles | Burn easily and cleanly, gases at room temperature |
Long-chain hydrocarbons (e.g. heavy oils, bitumen) | Lubricants, ship fuels, road surfacing | Viscous, hard to burn, high boiling points |
Check for Understanding
Recall
a) What elements are in hydrocarbons?
Answer
Carbon and hydrogen only.
b) How does boiling point change with chain length?
Answer
Increases.
c) How does viscosity change with chain length?
Answer
Increases.
d. What happens to volatility as chain length increases?
Answer
Decreases.
Application
e) Explain why short-chain hydrocarbons are more flammable.
Answer
Smaller molecules with relatively weak intermolecular forces; Lower boiling point; ignite easily.
f) Explain why long-chain hydrocarbons are less useful as fuels.
Answer
Larger molecules with relatively stronger weak intermolecular forces (compared to short cains); Harder to ignite; high boiling point; viscous.
g) Explain why boiling point increases as molecular size increases.
Answer
Larger molecules; stronger intermolecular forces; more energy needed to overcome them.
Summary
Hydrocarbon properties depend on molecular size.
Larger molecules → higher boiling points, more viscous, less flammable.
Short-chain hydrocarbons are best as fuels