Group 7 (halogens)
Lajoy Tucker
Teacher

Contents
Introduction & Definitions
Group 7 elements (halogens): Fluorine, chlorine, bromine, iodine, and astatine – non-metals with similar chemical properties due to having seven outer electrons.
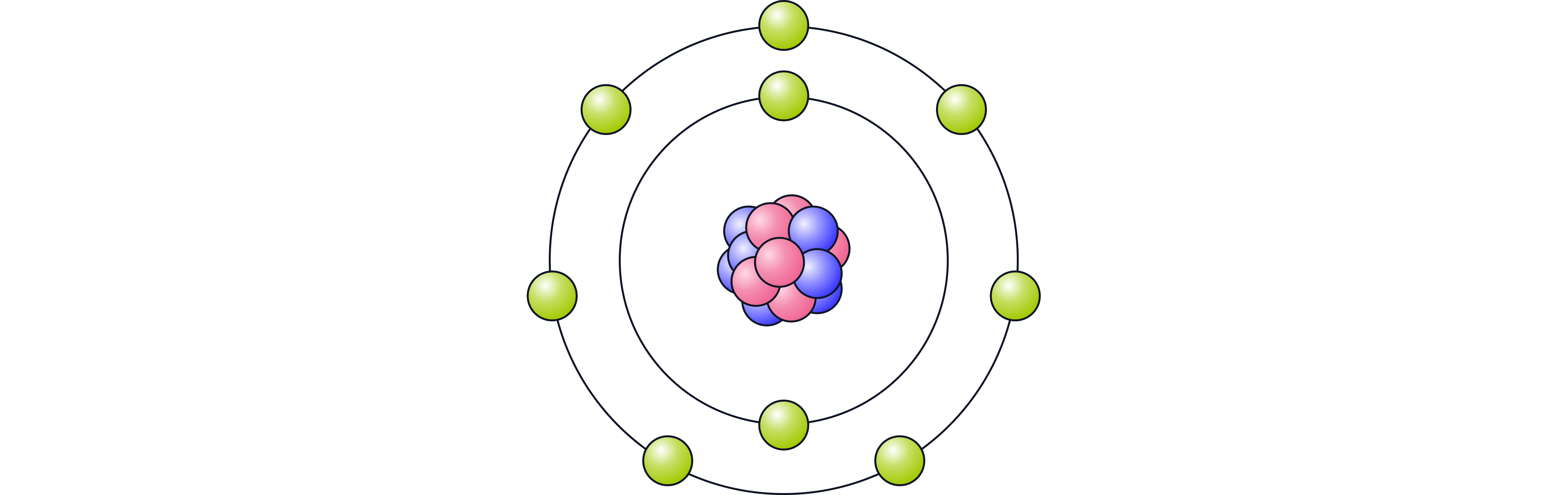
Halide ions: Negatively charged ions formed when halogens gain an electron (e.g., ).
Principles
Halogens exist as diatomic molecules . This means two halogen atoms are covalently bonded together.
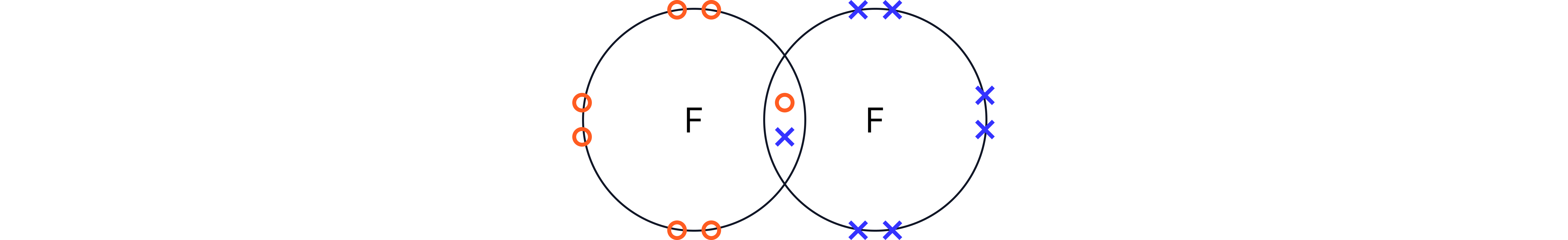
Trends are observed down the group in:
Boiling points
Reactivity
Physical Properties of Halogens
Boiling points increase down the group due to:
Larger molecules, leading to…
Stronger intermolecular forces, therefore…
More energy required to separate molecules.
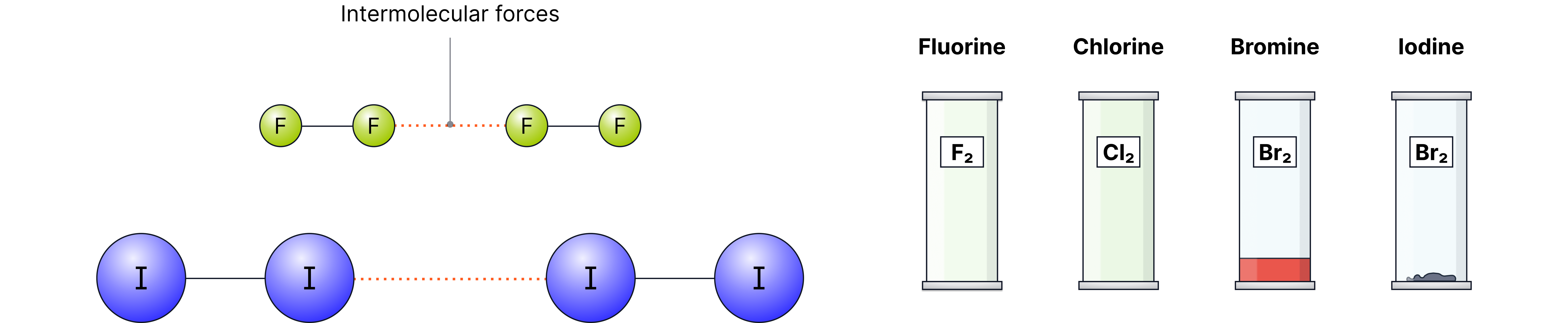
Halogen | Boiling point | Standard state | Colour | Colour in aqueous solution |
Fluorine | -188 | Gas | Pale yellow | |
Chlorine | -35 | Gas | Pale green | Pale green |
Bromine | 59 | Liquid | Red-brown | Orange |
Iodine | 184 | Solid | Grey | Brown |
Reactivity of halogens
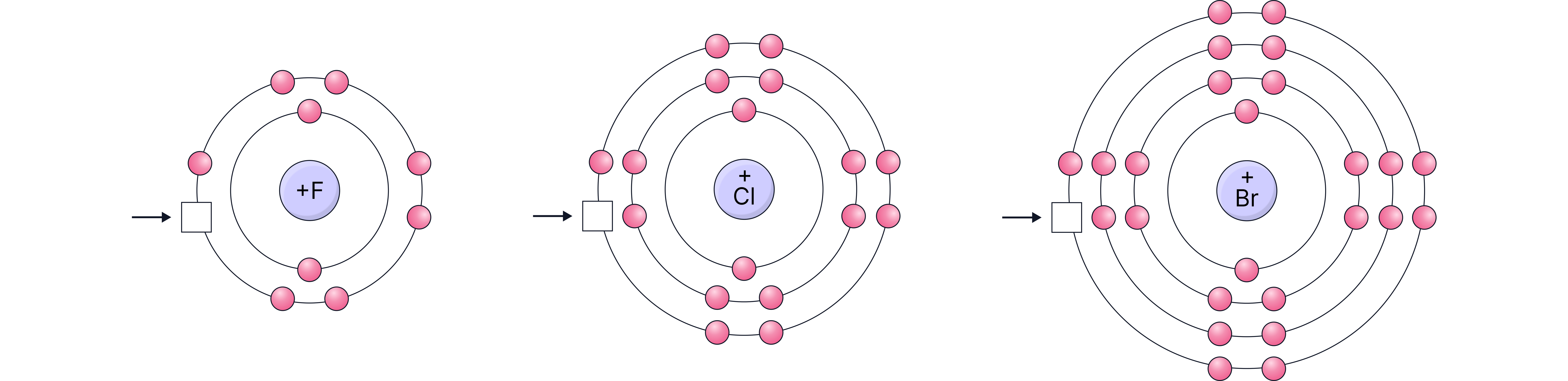
Group 7 elements are very reactive because they have 7 electrons in their outer shell.
They need to gain 1 electron during chemical reactions.
The reactivity of group 7 elements decreases as you go down the group.
This is because the outer shell is further from the nucleus so there is a weaker/less attraction between the nucleus and the electron being gained.
The atom can therefore gain an electron less easily down the group.
Reactions with Metals
When halogens react with metals, they form ionic compounds.
Each halogen atom gains one electron to form a halide ion .
The compound formed is a metal halide.
Example – Sodium reacting with chlorine
Word equation:
sodium chlorine sodium chloride
Symbol equation:
Type of bonding: Ionic
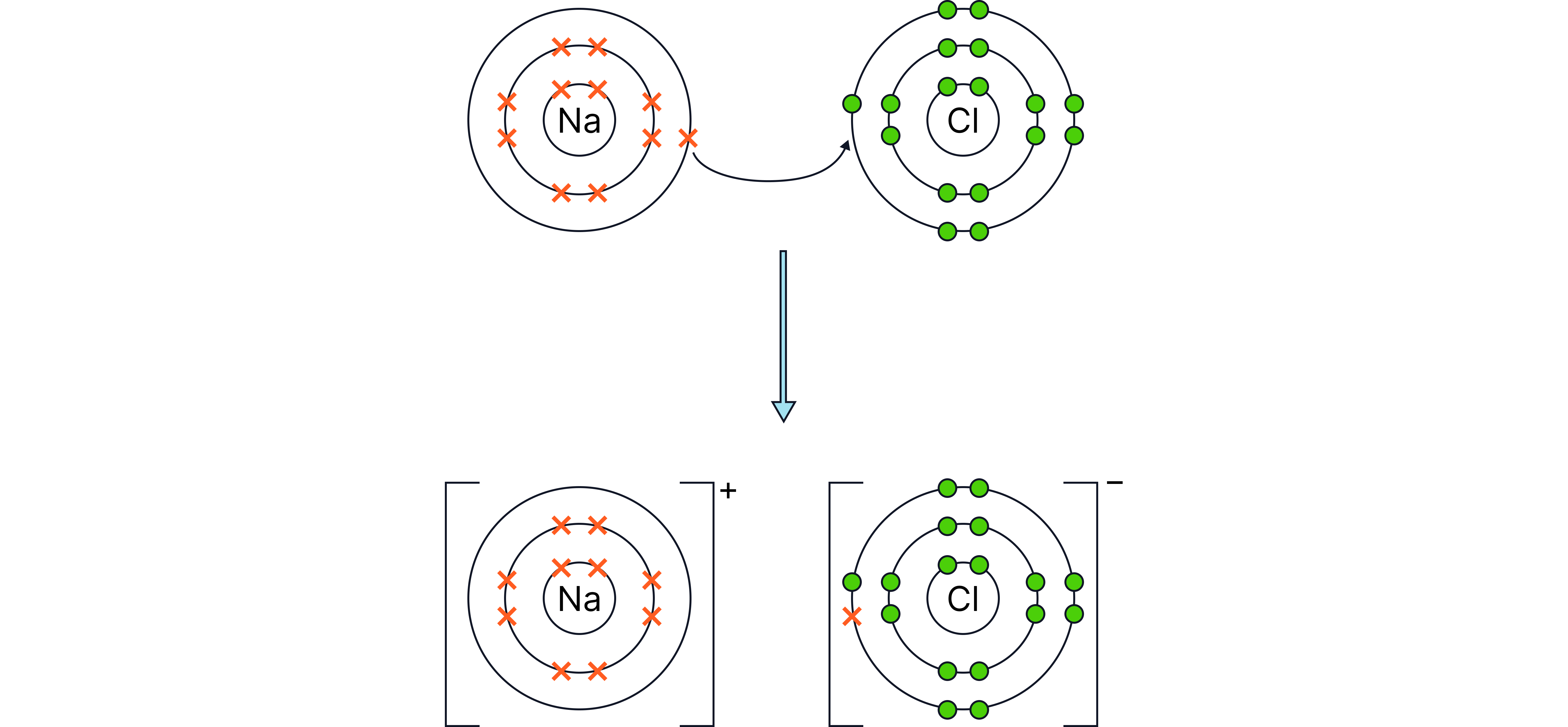
Explanation: Each sodium atom loses one electron, each chlorine atom gains one electron.
Reactions with Non-metals
When halogens react with other non-metals, they form covalent compounds.
Atoms share pairs of electrons so that each achieves a full outer shell.
Example – Hydrogen reacting with chlorine
Word equation:
hydrogen chlorine hydrogen chloride
Symbol equation:
Type of bonding: Covalent
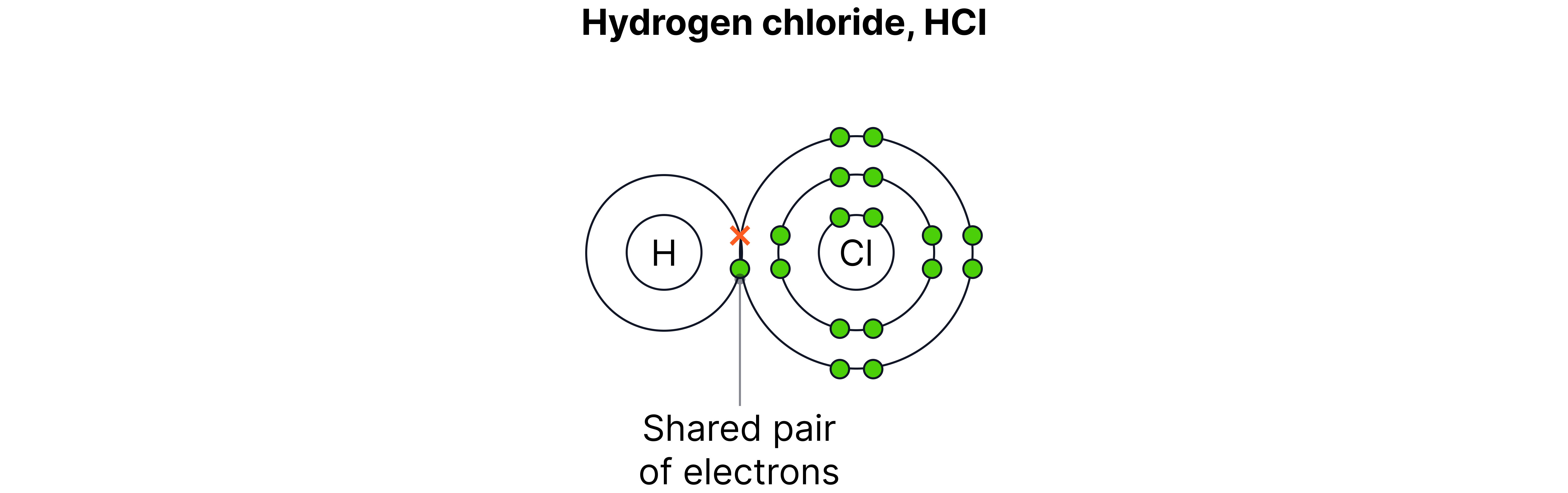
Explanation: Each hydrogen atom shares one electron with chlorine.
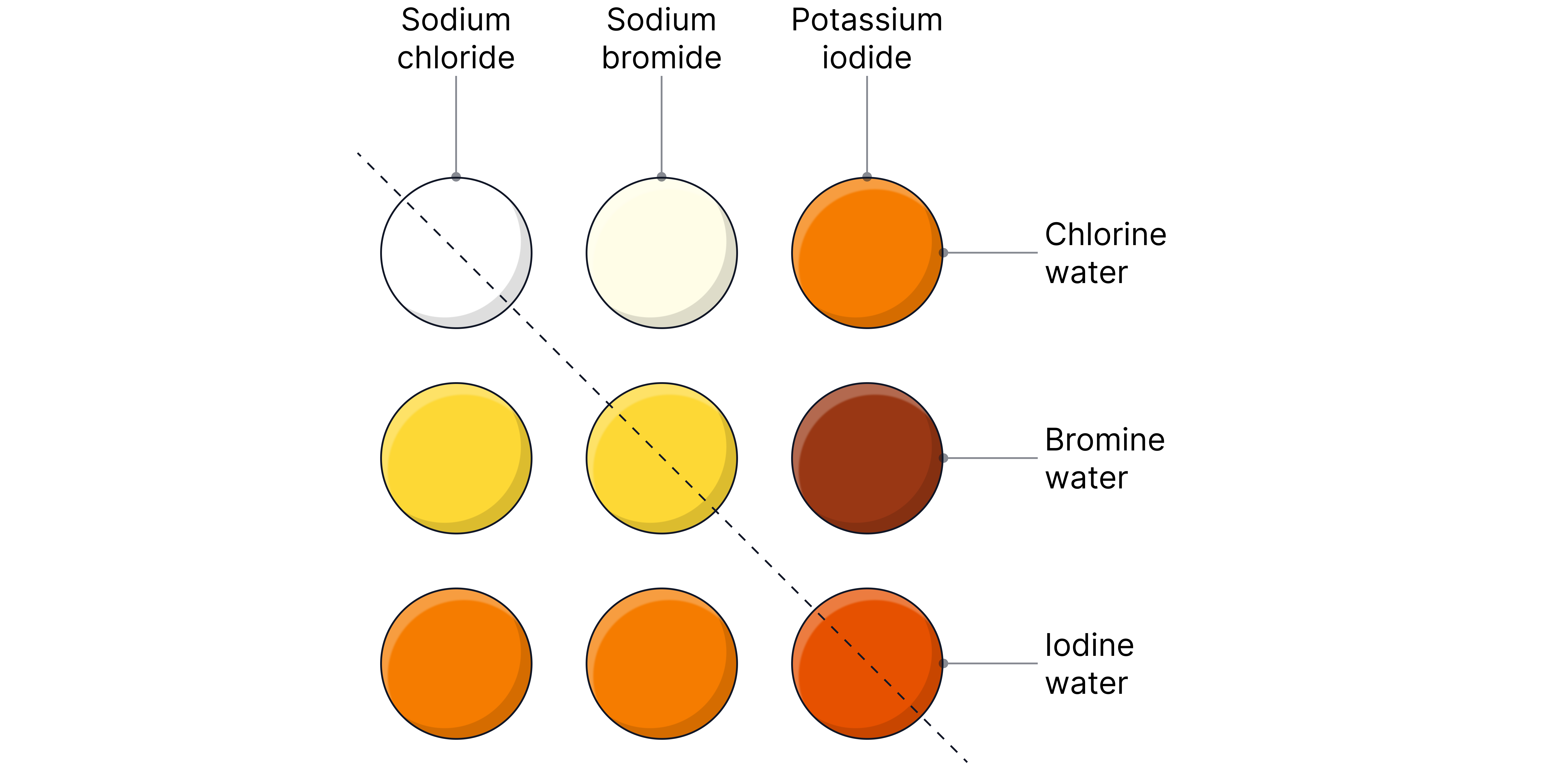
Displacement Reactions
The reactivity of the halogens can be demonstrated experimentally through displacement reactions.
A more reactive halogen displaces a less reactive halide from its compound.
For example:
Chlorine sodium bromide sodium chloride bromine
The bromine is displaced from its compound by chlorine
A colour change is observed if a reaction takes place as the halogen in solution determines its colour.
Example ionic equations (without the spectator ions):
Reaction | Observation | |
|---|---|---|
Pale green to orange solution | ||
Pale green to brown solution | ||
Orange to brown solution | ||
Solution stays brown is less oxidising than |
Check for Understanding
Recall
a) Name the four main halogens studied at GCSE.
Answer
Fluorine; chlorine; bromine; iodine.
b) What is meant by a halide ion?
Answer
A negatively charged ion formed when a halogen gains one electron (e.g. Cl⁻, Br⁻, I⁻).
c) What type of molecules do halogens exist as?
Answer
Diatomic molecules (X₂).
d) How does reactivity change down Group 7?
Answer
Reactivity decreases down the group.
Application
e) Explain why the boiling points of halogens increase down the group.
Answer
Molecules become larger; stronger intermolecular forces between molecules; more energy required to overcome force/separate molecules; boiling point increases.
f) Explain what happens when chlorine reacts with sodium and identify the type of bonding formed.
Answer
Sodium loses one electron; chlorine gains one electron; sodium chloride forms; ionic bonding formed.
g) Describe what is meant by a displacement reaction and explain why chlorine can displace bromine from sodium bromide.
Answer
A more reactive halogen displaces a less reactive halide from a compound; chlorine is more reactive than bromine; therefore chlorine displaces bromine from sodium bromide
h) Explain why fluorine is more reactive than bromine.
Answer
Fluorine is more reactive than bromine because fluorine has fewer electron shells, so its outer shell is closer to the nucleus.
This means the attraction between the nucleus and an incoming electron is stronger.
Therefore, fluorine can gain one electron more easily than bromine, making it more reactive.
Challenge
i) Write the ionic equation for the reaction between chlorine and iodide ions and explain why the reaction occurs.
Answer
Ionic equation:
Cl₂ + 2I⁻ → 2Cl⁻ + I₂
Explanation: chlorine is more reactive than iodine; chlorine gains electrons more readily; therefore it oxidises iodide ions to iodine.
Key Tips
Use the correct language. The halogens (fluorine, chlorine, bromine, iodine) vs the halide ions (fluoride, chloride, bromide, iodide)
Halogens react with metals to form ionic halides (e.g.)
Halogens react with non-metals to form covalent molecules (e.g. ).
Halogen atoms gain or share one electron to achieve a full outer shell.
Reactivity decreases down the group – chlorine reacts more vigorously than bromine or iodine.