Group 1 (alkali metals)
Lajoy Tucker
Teacher

Contents
Introduction
The elements in Group 1 of the periodic table are called the alkali metals.
They include lithium (Li), sodium (Na) and potassium (K).
They are all soft metals, can be cut with a knife, and have low melting points compared to most metals.
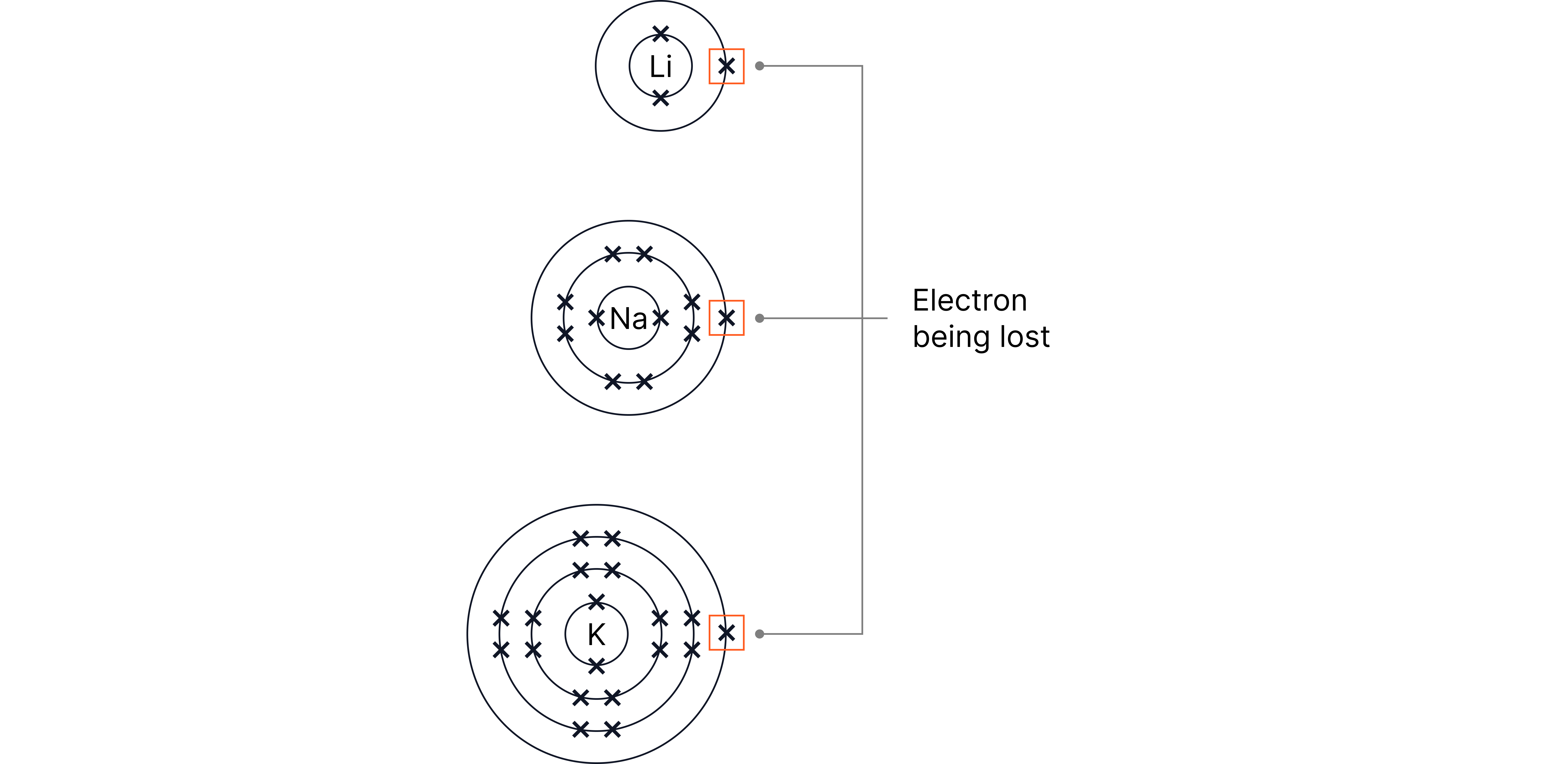
Each atom has one electron in its outer shell, which gives these elements their similar chemical properties.
Electron Structure and Reactivity
Because each atom has one outer electron, they are very reactive – they easily lose this electron to form positive ions () with a full outer shell.
As you go down the group, the outer electron is further from the nucleus and less strongly attracted, so it is lost more easily.
Therefore, reactivity increases down the group.
Element | Electronic Structure | Trend in Reactivity |
|---|---|---|
Lithium | 2,1 | Least reactive |
Sodium | 2,8,1 | More reactive |
Potassium | 2,8,8,1 | Most reactive (of the three |
Reactions of Group 1 Metals
Reaction with Oxygen
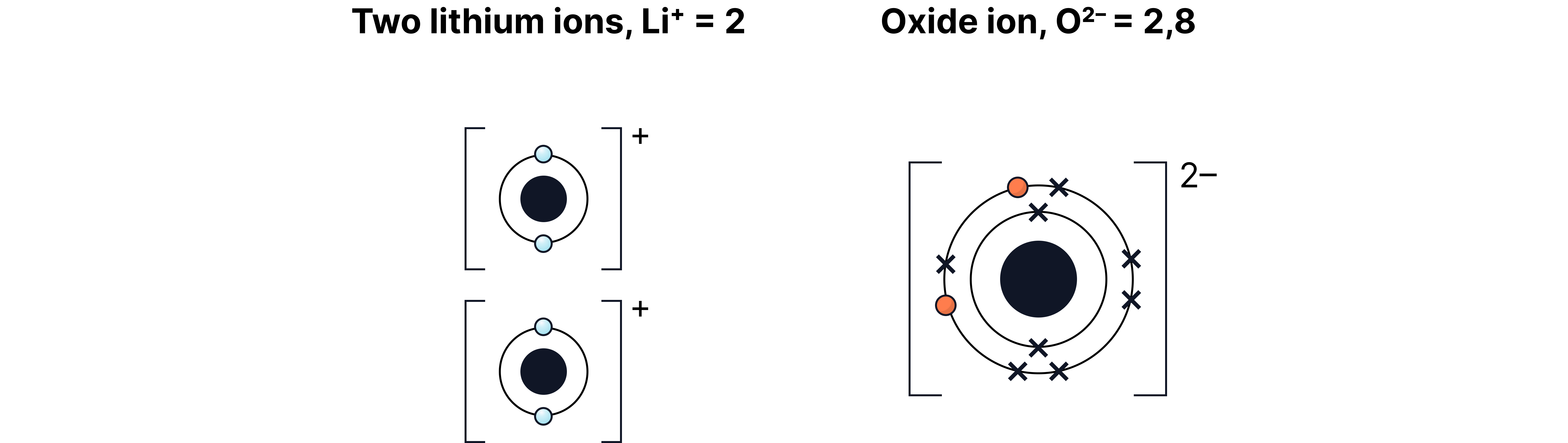
The alkali metals react quickly with oxygen in air to form metal oxides.
They tarnish (go dull) as a thin oxide layer forms.
Examples:
Lithium oxygen lithium oxide
Sodium oxygen sodium oxide
Potassium oxygen potassium oxide
Type of compound: Ionic
Reaction with Chlorine
The alkali metals react vigorously with chlorine gas to form white crystalline metal chlorides (salts).
Examples:
Lithium chlorine lithium chloride
Sodium chlorine sodium chloride
Potassium chlorine potassium chloride
Type of compound: Ionic
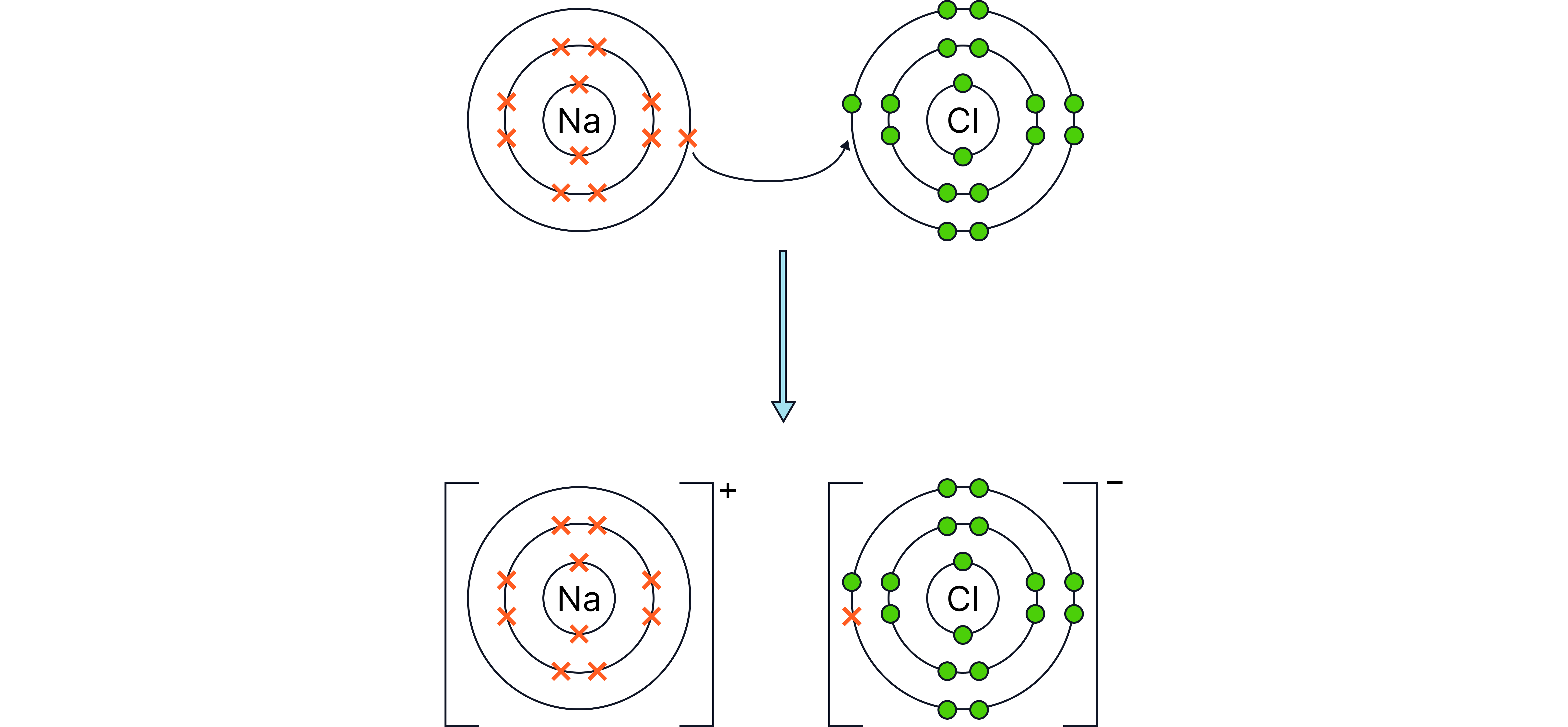
Explanation: Each metal atom loses one electron; each chlorine atom gains one.
Reaction with Water
The alkali metals react vigorously with water to form hydrogen gas and a metal hydroxide (alkaline solution).
The reaction gets more vigorous down the group.
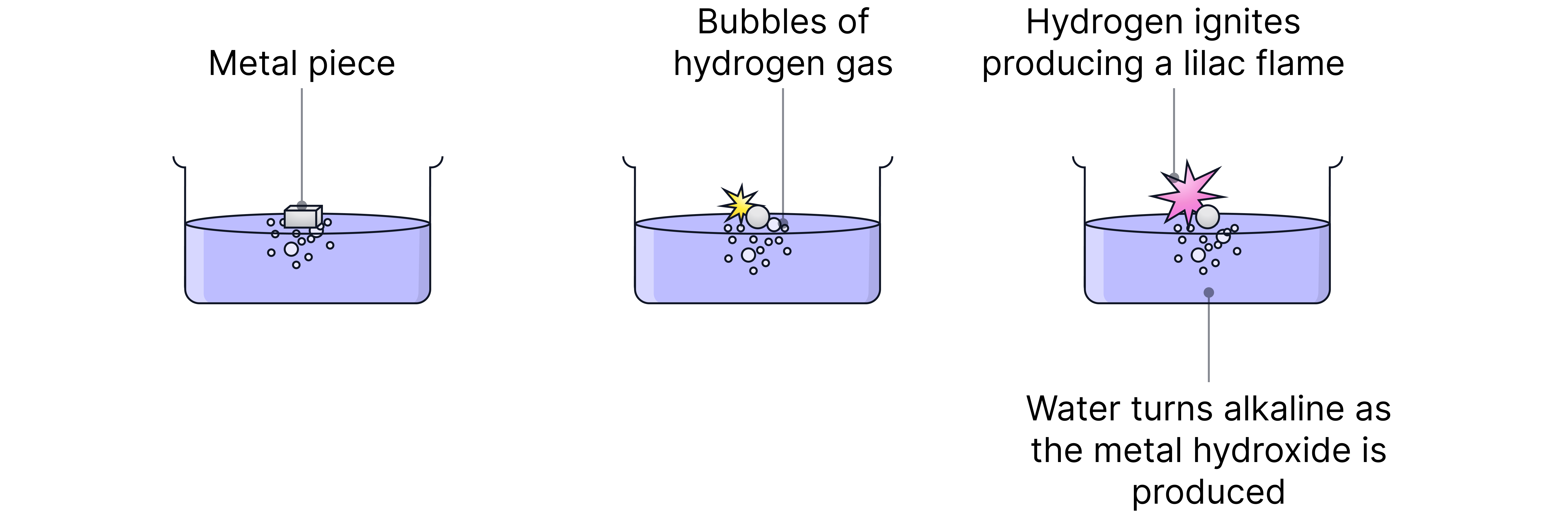
Observations with lithium | Observations with sodium | Observations with potassium |
Fizzing, Floats, Moves slowly | Melts into a ball, Rapid fizzing Moves faster, Possible orange flame | Melts into a ball, Vigorous fizzing, Moves rapidly, Lilac flame |
Examples:
Lithium water lithium hydroxide hydrogen
Sodium water sodium hydroxide hydrogen
Potassium water potassium hydroxide hydrogen
Check for Understanding
Recall
a) What name is given to the elements in Group 1 of the periodic table?
Answer
Alkali metals.
b) How many electrons are in the outer shell of Group 1 elements?
Answer
One electron.
c) What two products are formed when a Group 1 metal reacts with water?
Answer
Hydrogen gas and a metal hydroxide.
Application
d) Explain why reactivity increases down Group 1.
Answer
The outer electron is further from the nucleus down the group and is less strongly attracted, so it is lost more easily.
e) Write the word equation for the reaction between sodium and water.
Answer
Sodium + water → sodium hydroxide + hydrogen.
Challenge
f) A new element is discovered below potassium in Group 1. Predict how its reaction with water would compare to potassium and state the products formed.
Answer
It would react more violently than potassium because reactivity increases down the group. The reaction would produce hydrogen gas and a metal hydroxide.
Summary for Students
Group 1 elements = alkali metals with one outer electron.
They form ionic compounds when reacting with non-metals.
They react with:
Oxygen metal oxides
Chlorine metal chlorides
Water metal hydroxides hydrogen
Reactivity increases down the group because the outer electron is easier to lose.
Properties and trends can be predicted using their position in the group.