Group 0 (noble gases)
Lajoy Tucker
Teacher

Contents
Introduction
The elements in Group 0 of the periodic table are called the noble gases.
They include helium , neon , argon , krypton , xenon and radon .
All are non-metals and exist as single atoms (monatomic gases).

Electron Structure and Stability
Noble gases have full outer electron shells, which makes them very stable and therefore chemically inert (unreactive).
This stability means they do not easily gain, lose or share electrons.
Because of this, they rarely form molecules or compounds.
Element | Electron Structure |
Helium | 2 |
Neon | 2,8 |
Argon | 2,8,8 |
Trends Down the Group
As you go down Group 0 the boiling points increase, this is because the atoms get larger and the intermolecular forces between them become stronger, so more energy is needed to separate them.
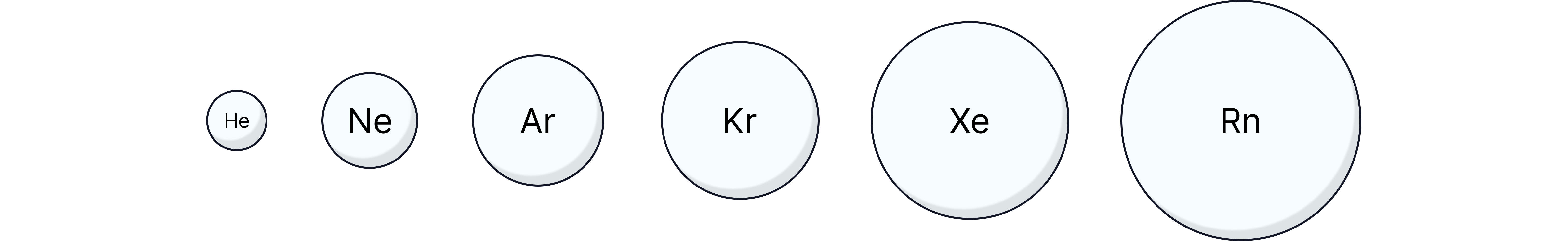
Element | Relative Atomic Mass | Boiling Point (°C) | Trend |
Helium | 4 | -269 | Lowest |
Neon | 20 | -246 | |
Argon | 40 | -186 | |
Krypton | 84 | -152 | |
Xenon | 131 | -108 | Highest |
Check for Understanding
Recall
a) What name is given to the elements in Group 0 of the periodic table?
Answer
Noble gases.
b) What type of particles do noble gases exist as?
Answer
Single atoms (monatomic gases).
c) Why are noble gases very unreactive?
Answer
They have full outer electron shells, making them very stable and unreactive.
Application
d) Explain why boiling points increase down Group 0.
Answer
Atoms become larger down the group and intermolecular forces become stronger, so more energy is needed to separate the atoms.
e) Argon is below neon in the periodic table. Predict how its boiling point compares with neon.
Answer
Argon has a higher boiling point than neon.
Challenge
f) A new element is discovered below xenon in Group 0. Predict its relative atomic mass, boiling point and reactivity compared with xenon.
Answer
It would have a greater relative atomic mass and a higher boiling point than xenon, but it would still be very unreactive because it would have a full outer electron shell.
Summary
Group 0 elements are unreactive (chemically inert) gases with full outer shells.
Boiling points increase down the group because atomic size and intermolecular forces increase.
You can predict trends down the group using their position in the periodic table.