Metals and non-metals
Lajoy Tucker
Teacher

Contents
Introduction
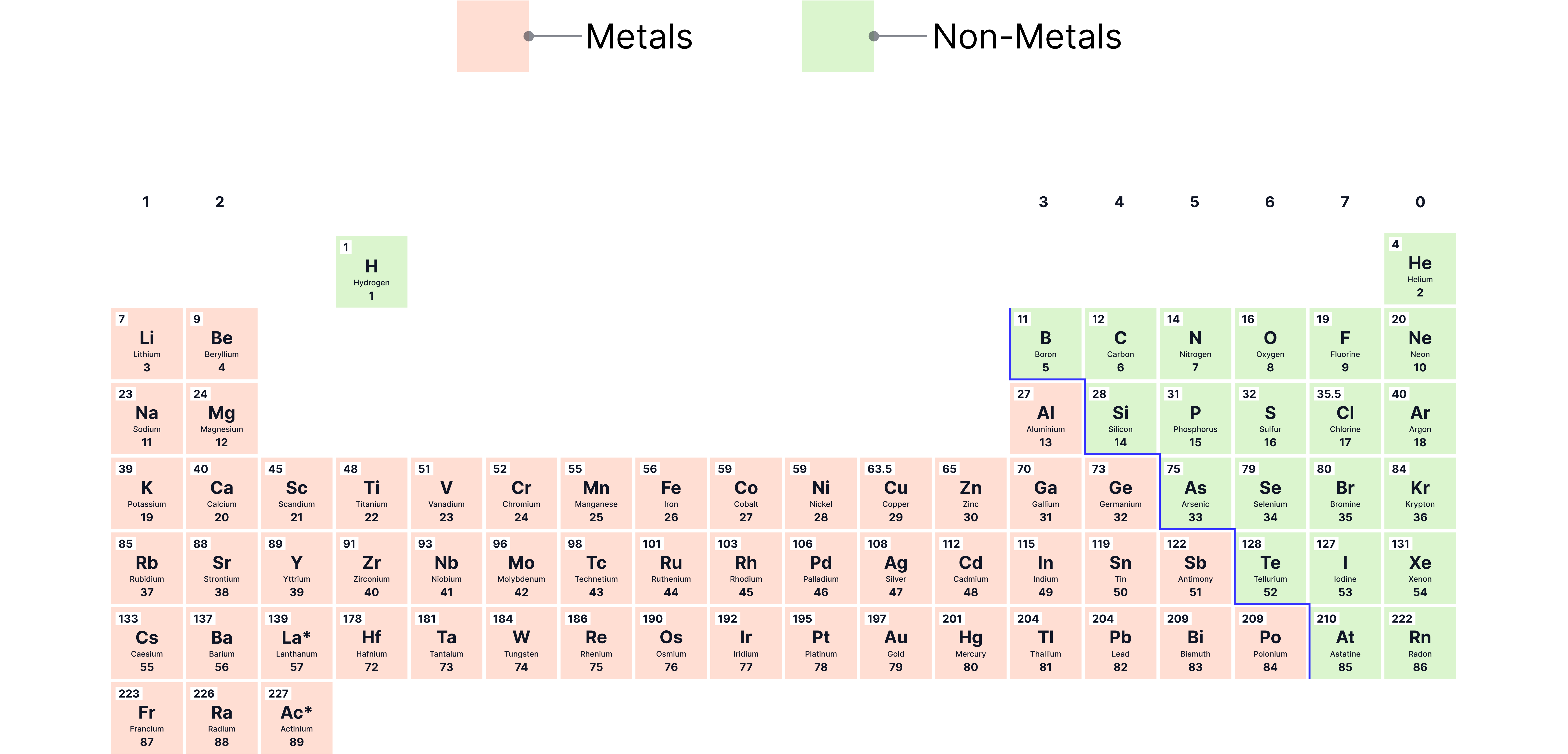
The majority of elements are metals, and they dominate the left and bottom of the periodic table.
Non-metals are found towards the right and top of the periodic table.
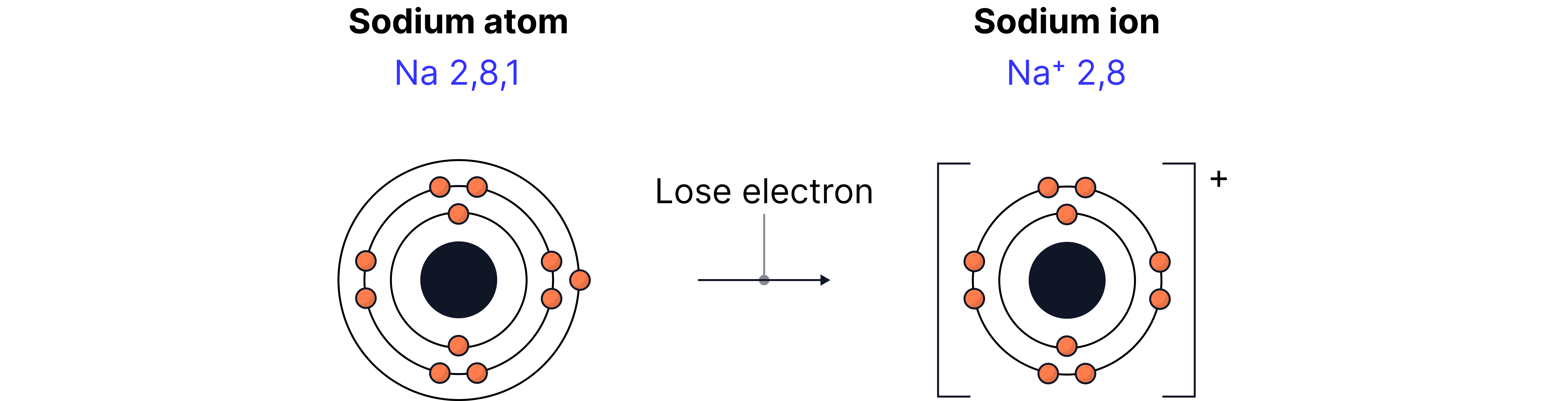
Metals form positive ions when they react
Physical Properties of Metals and Non-metals
Property | Metals | Non-metals | Explanation / Example |
Appearance | Shiny (metallic lustre) | Dull | Metals like copper are shiny; sulfur is dull. |
Melting and boiling points | High | Low (many are gases) | Metals have strong metallic bonds; non-metals have weak forces between molecules. |
Density | High | Low | Metals are closely packed; non-metals have more space between atoms or molecules. |
Malleability | Can be hammered or bent into shape | Brittle (shatter easily) | Metallic bonds allow layers of atoms to slide. |
Electrical conductivity | Good conductors | Poor conductors (except graphite) | Metals have delocalised electrons that carry charge through the structure. |
Thermal conductivity | Good | Poor | Delocalised electrons transfer energy easily in metals. |
Further detail on bonding and structure can be found in Topic 2
Metals have few outer electrons → they lose electrons, forming positive ions.
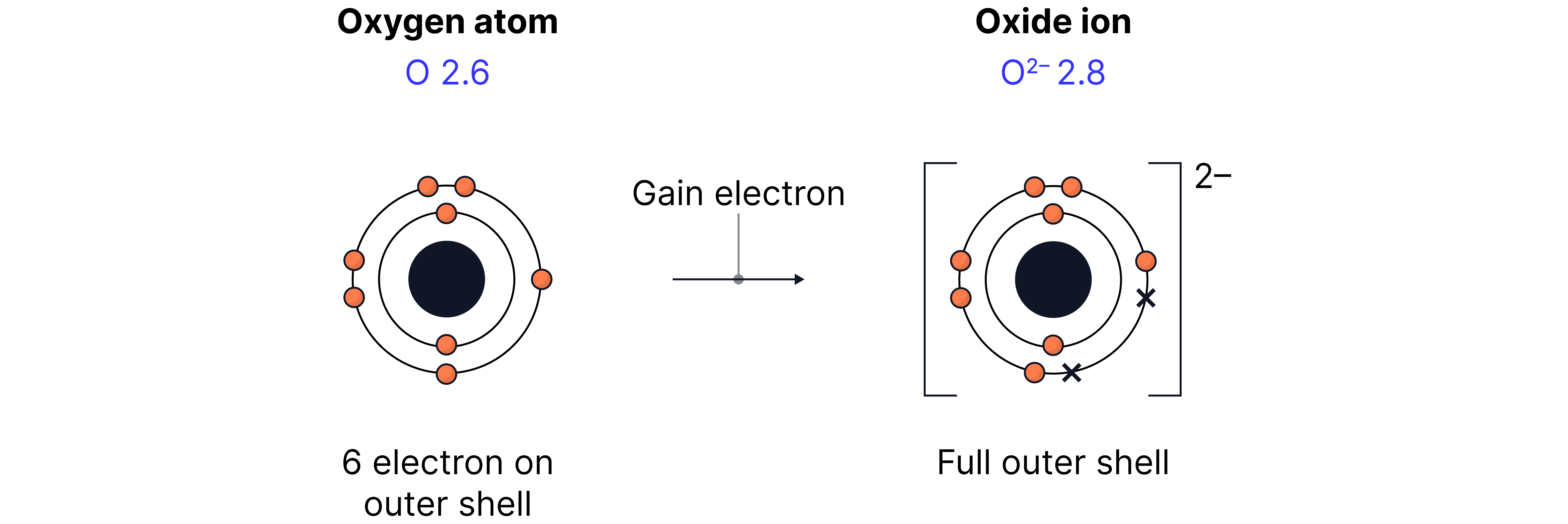
Non-metals have more outer electrons → they gain or share electrons to achieve a full shell (e.g. Group 7).
Non-metal atoms gain electrons when reacting with metals to form ionic bonds.
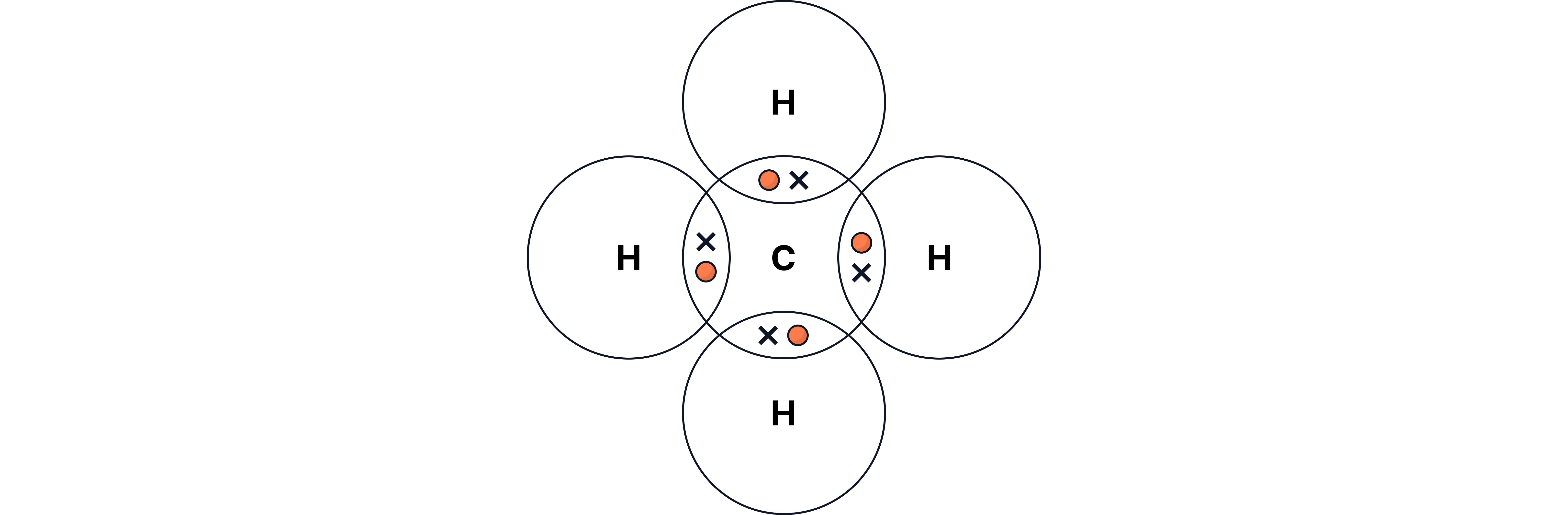
Non-metal atoms share pairs of electrons when reacting with non-metals to form covalent bonds.
Chemical Properties of Metals and Non-metals
Property | Metals | Non-metals |
|---|---|---|
Reaction with oxygen | Form metal oxides, which are basic (e.g. ) | Form non-metal oxides, which are acidic (e.g. ) |
Reaction with water | Some react to form metal hydroxides and hydrogen gas (e.g. ) | Generally, do not react with water directly |
Reaction with halogens | Form ionic compounds (e.g. ) | Form covalent compounds (e.g. ) |
Type of ions formed | Positive ions (cations) | Negative ions (anions) or neutral molecules |
Check for Understanding
Recall
a) Where are metals located in the periodic table?
Answer
Metals are found on the left and towards the bottom of the periodic table (left of steps starting between boron and auminium).
b) What type of ions do metals form when they react?
Answer
Positive ions.
c) What type of ions do non-metals form when they react?
Answer
Negative ions, or they share electrons.
d) Name one physical property of metals.
Answer
Acceptable answers include strong, dense, or good conductors of electricity or heat.
Application
e) Explain why metals tend to form positive ions when they react.
Answer
Metals have few electrons in their outer shell and lose electrons to achieve a full outer shell, forming positive ions.
f) Compare how a metal and a non-metal form compounds with chlorine.
Answer
Metals transfer electrons to chlorine to form ionic chlorides (for example NaCl), whereas non-metals share electrons with chlorine to form covalent chlorides (for example HCl).
Summary
Metals form positive ions, are strong, dense, and conductive, and react by losing electrons.
Non-metals form negative ions or covalent bonds, are brittle or gaseous, and react by gaining or sharing electrons.
The position in the periodic table reflects atomic structure and number of outer electrons.
These differences explain patterns in reactivity across Groups 0, 1, and 7, and how elements bond to form compounds.