Neutralisation of acids and salt production
Emmanuel Opoku
Teacher

Contents
Neutralisation Reactions
Definition
Neutralisation is the reaction between an acid and a base (such as a metal oxide, hydroxide, or carbonate) to form a salt and water.
Some reactions also produce carbon dioxide if a metal carbonate is used.
Ionic equation (HT only):
General Word Equations
Type of Base | Products | Example symbol equation |
Acid + Metal Oxide | → Salt + Water | |
Acid + Metal Hydroxide (alkali) | → Salt + Water | |
Acid + Metal Carbonate | → Salt + Water + Carbon dioxide |
Predicting the Salt Formed
The name of the salt depends on:
1. The acid used:
Hydrochloric acid → chlorides
Nitric acid → nitrates
Sulfuric acid → sulfates
2. The base, alkali, or carbonate used:
e.g. Sodium hydroxide → sodium salts
e.g. Copper oxide → copper salts
Examples of Neutralisation Reactions
Acid Used | Base / Alkali / Carbonate | Word Equation | Salt Formed |
Hydrochloric acid | Magnesium oxide | Hydrochloric acid + Magnesium oxide → Magnesium chloride + Water | Magnesium chloride |
Nitric acid | Potassium hydroxide | Nitric acid + Potassium hydroxide → Potassium nitrate + Water | Potassium nitrate |
Sulfuric acid | Copper(II) carbonate | Sulfuric acid + Copper carbonate → Copper sulfate + Water + Carbon dioxide | Copper(II) sulfate |
Citric acid | Calcium hydroxide | Citric acid Calcium hydroxide Calcium citrate Water | Calcium citrate |
Soluble Salts
Definition:
Soluble salts can be made by reacting acids with solid, insoluble substances such as metal oxides, hydroxides, or carbonates.
Key idea:
The solid is added to the acid until no more reacts (acid is neutralised).
The excess solid (insoluble base) is then filtered off, leaving a solution of the salt.
The solution can then be crystallised to form pure solid salt crystals.
Key Steps: Making a Soluble Salt
1. Add excess base:
Add excess base to warm dilute acid until no more reacts.
Ensures all acid reacts (acid fully neutralised).
2. Warm the mixture:
Gently heat to speed up reaction (do not boil).
3. Filter the mixture:
Remove unreacted solid base using filter paper, leaving an aqueous solution of the salt produced.
4. Evaporate some of the water:
Heat gently using a water bath or electric heater to concentrate the solution.
Avoid boiling to prevent decomposition (breaking down) of crystals.
5. Crystallise and dry:
Allow the solution to cool slowly to form large crystals.
Pat crystals dry with filter paper.
Required Practical 1 - Making a Soluble Salt
Title: Preparation of a pure, dry sample of a copper sulphate salt from copper oxide and sulphuric acid.
Aim:
To produce a pure, dry, soluble salt (copper sulfate) using an acid and an insoluble base.
Apparatus:
Beaker
Bunsen burner or electric heater
Tripod and gauze
Filter paper and funnel
Evaporating basin
Water bath
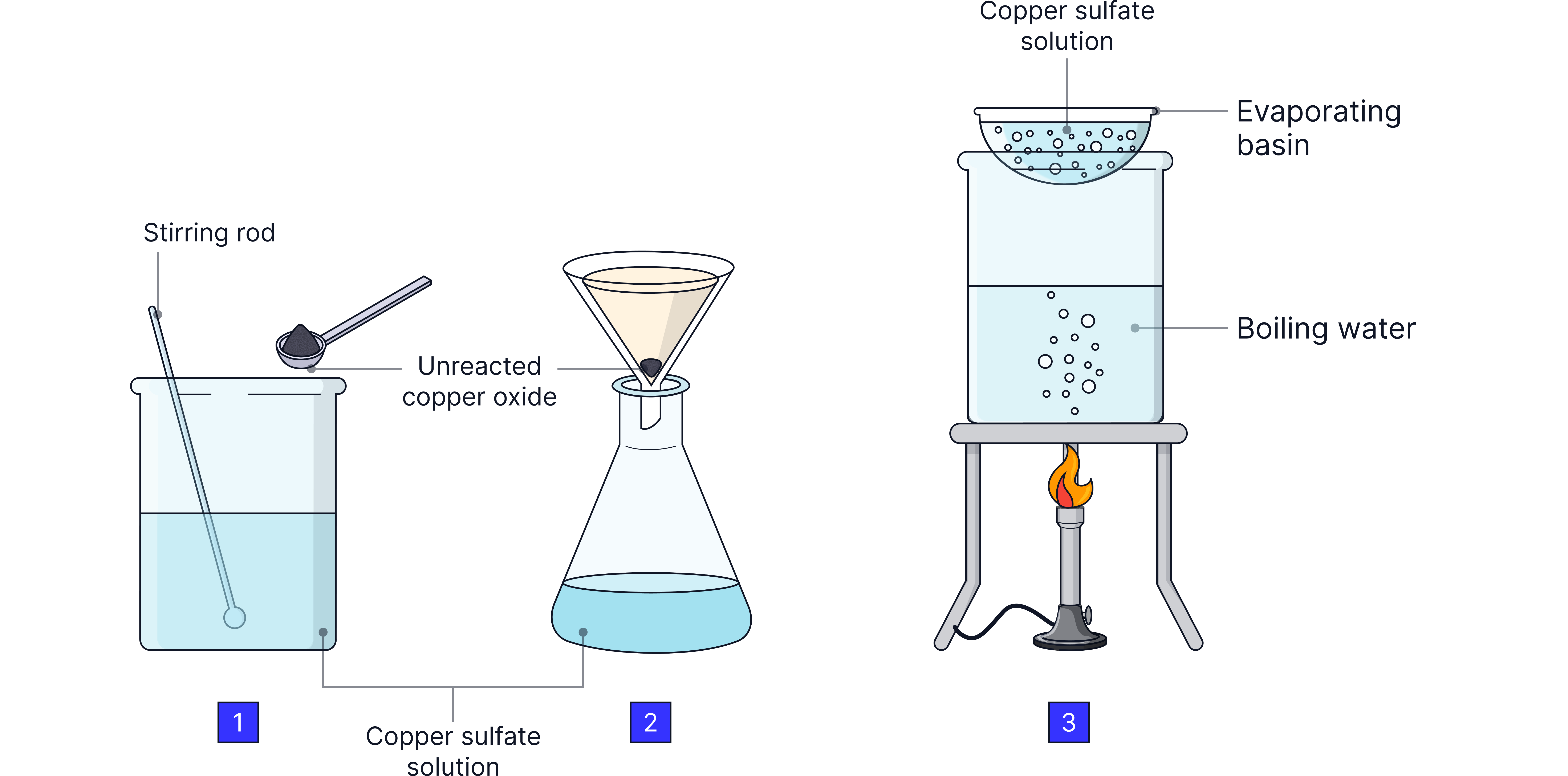
Method Summary:
Measure acid (e.g. sulfuric acid) into a beaker.
Warm acid gently.
Add excess of base (copper oxide) while stirring.
Continue until excess solid remains (acid fully reacted).
Filter to remove unreacted solid.
Evaporate part of the filtrate using a water bath.
Cool to crystallise, then dry crystals by patting with filter paper.
Summary Table
Concept | Key Points |
Neutralisation | Acid Base Salt Water |
Metal Carbonate Reaction | Acid Carbonate Salt Water CO₂ |
Salt Naming | Acid determines salt ending; metal in base determines first part |
Soluble Salt Formation | Acid Excess Insoluble Base Filter Evaporate Crystallise |
Unsuitable Metals | Sodium (too reactive), Copper (too unreactive) |
Equation (HT) |
Practice Questions
Recall
a) Write the general word equation for neutralisation.
Answer
a) Acid Base Salt Water
b) What type of reaction is acid carbonate?
Answer
b) Acid Carbonate Salt Water +
c) Which acid produces nitrate salts?
Answer
c) Nitric acid nitrates
d) Why is sodium not used to make salts in the lab?
Answer
d) Sodium is too reactive and reacts violently with acid.
e) Define a soluble salt.
Answer
e) A salt that dissolves in water.
f) Which ions react in the ionic equation for neutralisation? (HT only)
Answer
f) and ions.
Apply Your Knowledge
g) Describe how to make copper sulfate crystals from copper oxide and sulfuric acid.
Answer
g) Add excess filter evaporate crystallise dry.
h) Predict the products of nitric acid reacting with calcium hydroxide. water.
Answer
h) Calcium nitrate water.
i) Explain why an excess of base is used in this experiment.
Answer
i) Ensures all acid is neutralised.
j) Write the balanced symbol equation for zinc oxide reacting with hydrochloric acid.
Answer
j)
k) (HT only) Identify the spectator ions in .
Answer
k) and .
Challenge
l) Compare neutralisation using a metal oxide and using a metal carbonate in terms of observations and products.
Answer
l) Carbonate reactions produce fizzing (). Oxides do not.
m) Suggest how crystal size could be increased during salt preparation and explain why.
Answer
m) Slow cooling allows larger crystals to form.