The pH scale (including strong and weak acids)
Emmanuel Opoku
Teacher

Contents
What Are Acids and Alkalis?
Acids are substances that produce hydrogen ions when dissolved in water.
Alkalis are soluble bases that produce hydroxide ions in water.
Type | Ions Produced | Examples |
|---|---|---|
Acid | ions | |
Alkali | ions |
An acid and an alkali react in a neutralisation reaction:
General Word equation: Acid + Alkali → Salt + Water
HT ONLY - Ionic Equation:
The pH Scale
The pH scale measures how acidic or alkaline a solution is (0–14).
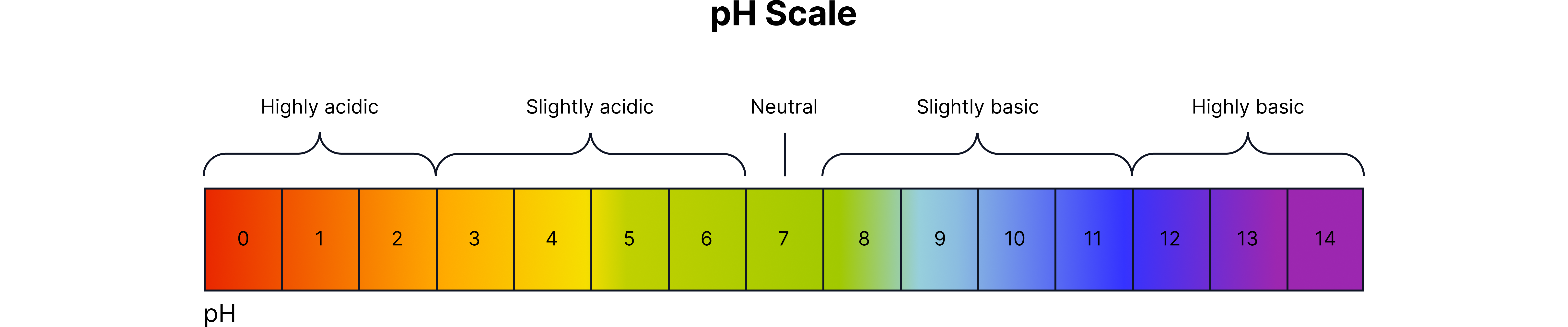
pH Range | Type of Solution | Indicator Colour |
|---|---|---|
1–3 | Strong acid | Red |
4–6 | Weak acid | Orange / Yellow |
7 | Neutral | Green |
8–11 | Weak alkali | Blue |
12–14 | Strong alkali | Purple |
pH 7 = neutral (pure water)
As pH decreases, acidity increases.
As pH increases, alkalinity increases.
The pH Equation (HT Only)
The pH scale is logarithmic - based on the hydrogen ion concentration.
Where:
= hydrogen ion concentration
pH decreases as increases
Example Calculations (HT Only)
Substitution into equation | Calculation | pH | |
pH = -log(1.0) | =0 | 0 | |
=1 | 1 | ||
| =2 | 2 | |
| =3 | 3 | |
| =4 | 4 | |
| =5 | 5 | |
| =6 | 6 | |
| =7 | 7 |
Each time increases by a factor of 10 (×10) → pH decreases by 1
Each time decreases by a factor of 10 (÷10) → pH increases by 1
pH and Concentration Changes Examples (HT Only)
Change in [H⁺] | Effect on pH | Example |
|---|---|---|
more concentrated | pH ↓ by 1 | pH 3 → 2 |
more concentrated | pH ↓ by 3 | pH 4 → 1 |
dilution | pH ↑ by 1 | pH 2 → 3 |
dilution | pH ↑ by 2 | pH 2 → 4 |
Each power of 10 change in concentration = 1 pH unit change. Increase in concentration leads to decrease in pH and vice versa.
Acid Strength vs Concentration
It’s common for students to mix up these two - but they are different concepts!
Term | Definition | Example / Notes |
|---|---|---|
Strength | The degree to which an acid dissociates/ionises in water (fully/partially) | = strong (fully dissociates/ionises), = weak (partially dissociates/ionises) |
Concentration | The amount of acid per of solution | acid is more concentrated than acid |
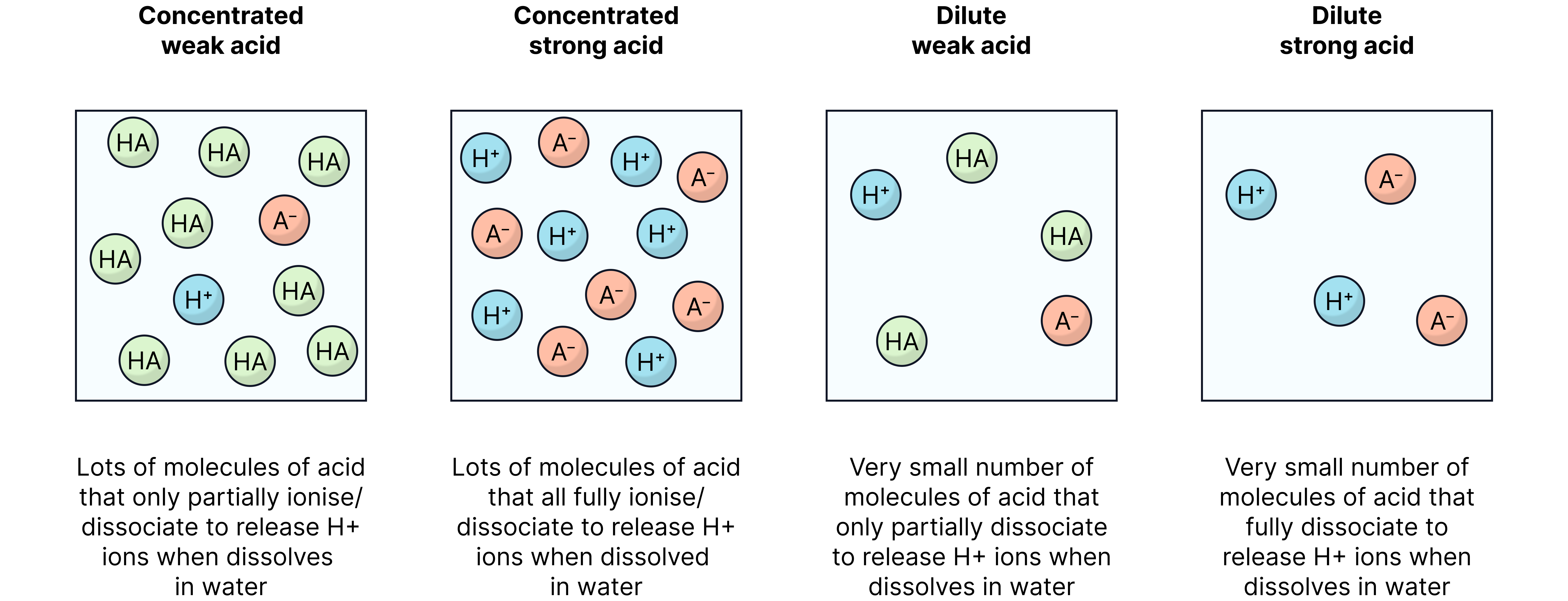
You can have a dilute strong acid (few particles but all dissociated/ionised) or a concentrated weak acid (many molecules but only partly dissociated/ionised). Same concept applies to bases.
Dissociate and ionise mean the same thing: breaking up into ions.
Strong and Weak Acids
Strong Acids
Completely dissociate/ionise in water.
Examples:
High → low pH (1–3)
Weak Acids
Partially dissociate/ionise in water.
Examples: Ethanoic acid , Citric acid, Carbonic acid
Lower → higher pH (4–6)
Comparing Strong and Weak Acids (HT Only)
Property | Strong Acid | Strong Acid |
|---|---|---|
Dissociation/Ionisation | Complete | Partial |
pH (same concentration of acid molecules) | Lower | Higher |
Reaction rate with metals | Faster | Slower |
Electrical Conductivity | Higher | Lower |
High | Low |
HT Key Idea:
For the same concentration, strong acids have more ions, so pH is lower.
Check Your Understanding
Recall
a) What ions do acids and alkalis produce?
Answer
a) Acids ; Alkalis .
b) What is the pH of a neutral solution?
Answer
b) pH 7.
c) State the pH ranges for strong acids, weak acids, weak alkalis, and strong alkalis.
Answer
c)
Strong acids: 1–3
Weak acids: 4–6
Weak alkalis: 8–11
Strong alkalis: 12–14
d) Define acid strength.
Answer
d) Strength = how completely an acid dissociates/ionises in water.
e) What is meant by acid concentration?
Answer
e) Concentration = the number of moles of acid per of solution.
f) Write the ionic equation for neutralisation.
Answer
f)
Apply
g) Explain the difference between acid strength and acid concentration. (HT only)
Answer
g) Strength = degree of dissociation/ionisation; concentration = amount of acid per .
h) Calculate the pH of a solution with . (HT only)
Answer
h)
i) Calculate the pH of a solution with . (HT only)
Answer
i) .
j) If an acid becomes more concentrated, how does its pH change? (HT only)
Answer
j) pH decreases by 2 units .
k) Why does a strong acid at have a lower pH than a weak acid at the same concentration? (HT only)
Answer
k) Strong acids fully dissociate/ionise, releasing more ions, lowering pH.
Challenge (HT only)
l) A solution has a pH of 2. What happens to its pH if it becomes more dilute?
Answer
l) pH increases by 3 (dilution).
m) Compare the rate of reaction of and with magnesium.
Answer
m) Hydrochloric acid reacts faster - more ions available for collisions per second as it is a stronger acid and fully dissociates.