Pure substances
Lajoy Tucker
Teacher

Contents
What Is a Pure Substance?
In chemistry:
A pure substance is one element or one compound,
not mixed with any other substance.
Examples:
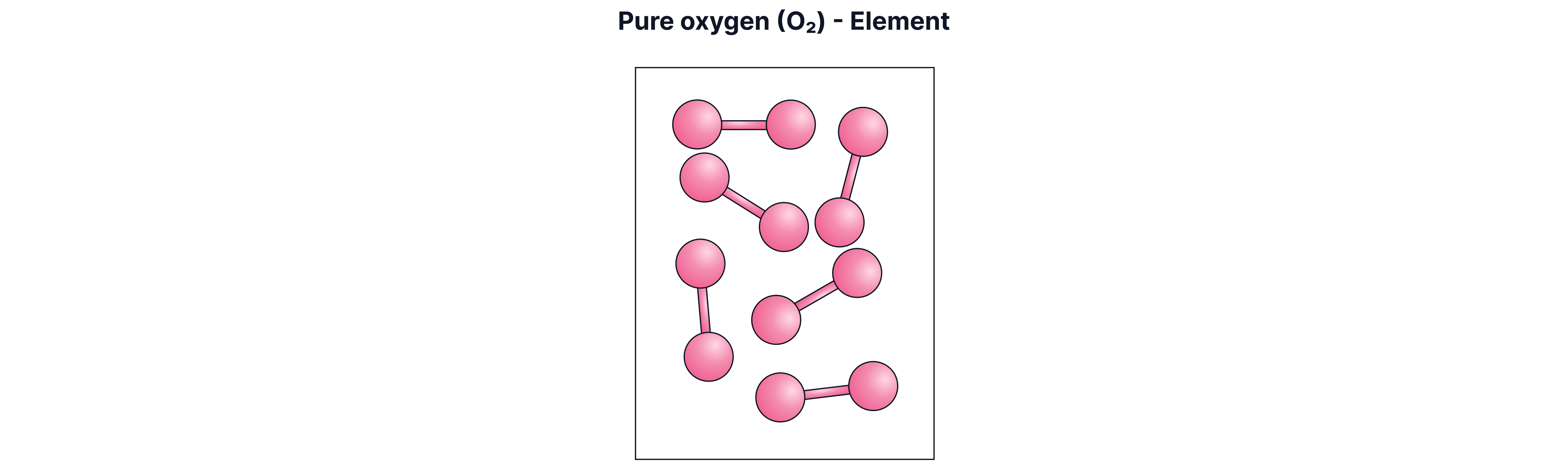
Pure oxygen - Element
Pure water - Compound
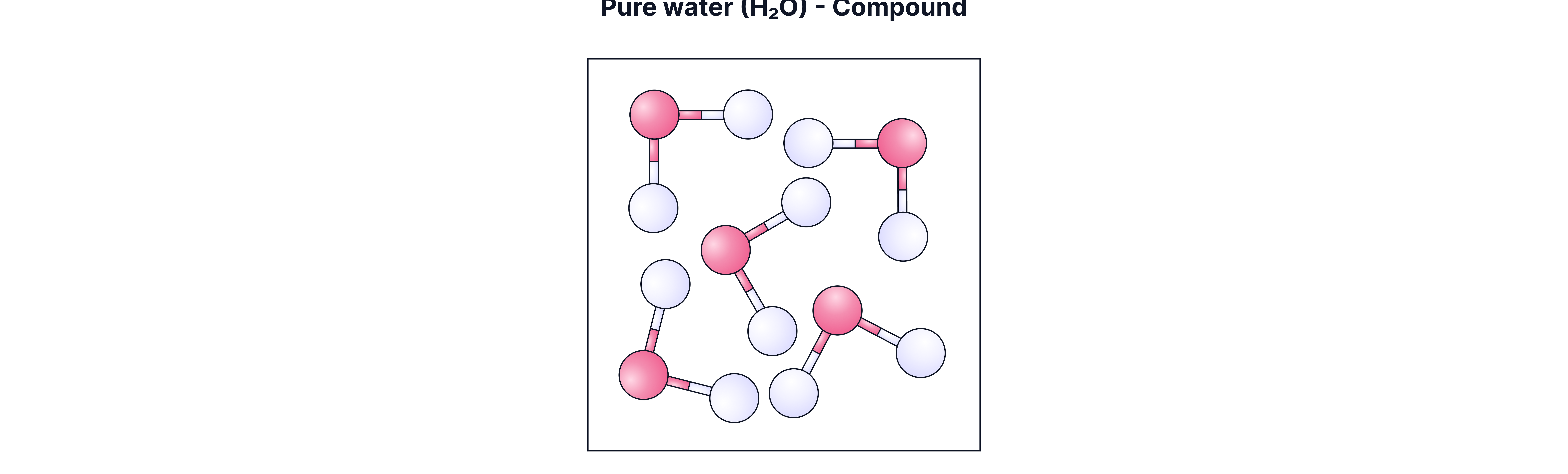
A pure substance has a fixed composition and exact, predictable properties.
In everyday language, pure can mean something that has had nothing added to it, such as “pure milk” or “pure honey", even though these are mixtures scientifically. Chemistry uses the word pure in a much stricter sense.
Melting and Boiling Points of Pure Substances
Pure substances melt and boil at specific, sharp temperatures.
Substance | Melting Point | Boiling Point |
|---|---|---|
Pure water | ||
Pure ethanol |
Distinguishing Pure Substances from Mixtures
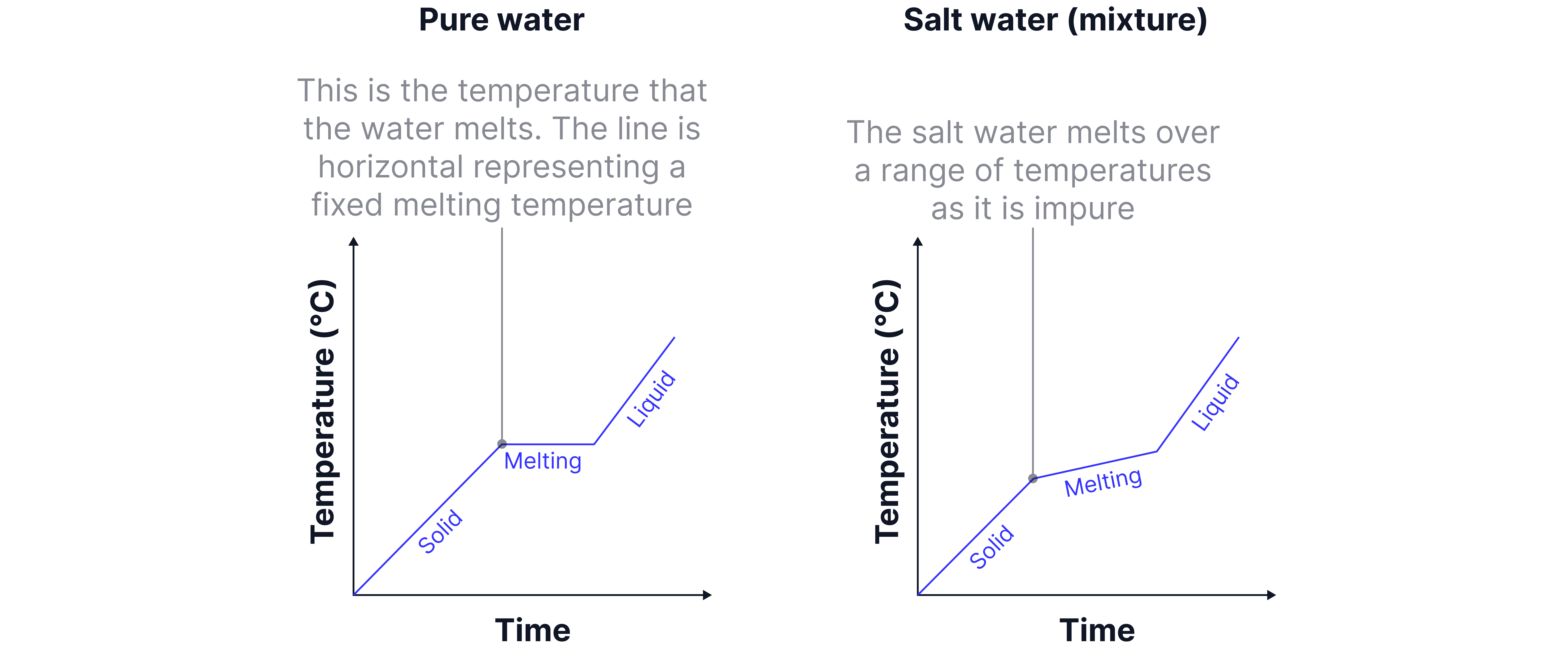
A pure substance has a single, exact melting point.
Mixtures do not have precise melting and boiling points.Instead, they:
Melt and boil over a range of temperatures
Change state gradually rather than sharply.
Substance | Melting point |
Pure water | 0 |
Salt water | -3 to -1 |
To decide whether a substance is pure:
1. Look up the known melting/boiling point of the pure substance.
2. Measure the sample’s melting/boiling point.
3. Compare the two sets of data.
If they match exactly, the substance is pure.
If there is a range or the temperature is higher or lower, the substance is impure.
Summary
A pure substance is a single element or compound with no other substances mixed in.
Pure substances have sharp melting and boiling points.
Mixtures melt and boil over ranges of temperatures.
Melting/boiling point data can be used to identify purity.
Everyday labels such as “pure orange juice” do not match the scientific definition.
Check for Understanding
Recall
a) Define a pure substance in chemistry.
Answer
One element or compound only.
b) How does a pure substance melt?
Answer
With a sharp melting point.
c) How does an impure substance melt?
Answer
Over a range of temperatures.
Application
d) A substance melts between 120 - 125°C. What does this indicate?
Answer
That the substance is impure.
e) A boiling point matches data exactly. What does this suggest?
Answer
Likely a pure substance.
Challenge (HT)
f) Explain why impurities lower and broaden melting point of an ionic compound.
Answer
Impurities disrupt lattice; sp requires less energy to break apart the lattice; causing it to melt over range.