Reversible Reactions
Dr. Davinder Bhachu
Teacher

Contents
The Big Idea
In many reactions you meet at school, the change feels one-way:
Reactants → Products (and that’s it)
But some reactions can go in both directions. These are called reversible reactions.
Definition
A reversible reaction is one where the substances produced can change back into the original substances.
We show this using a double arrow:
That symbol (⇌) tells you that:
The forward reaction (left → right) and
The reverse reaction (right → left)
can both happen.
How Reversible Reactions Are Written
We normally write two separate equations to show the two directions:
Forward reaction:
Reactants → Products
Reverse reaction:
Products → Reactants
Then we combine them into one line with a double arrow:
This reminds you that neither direction is “more real” than the other — they’re both valid changes.
Closed Systems
A closed system is essential for studying reversible reactions properly.
A closed system is a situation where:
No chemicals can enter or leave the container
Nothing is added or removed once the reaction starts
In practice, this usually means:
A sealed tube
A stoppered flask
A reaction vessel that doesn’t let gas escape
If particles can escape, the reverse reaction might not be able to happen fully, and the reaction will behave more like a one-way change.
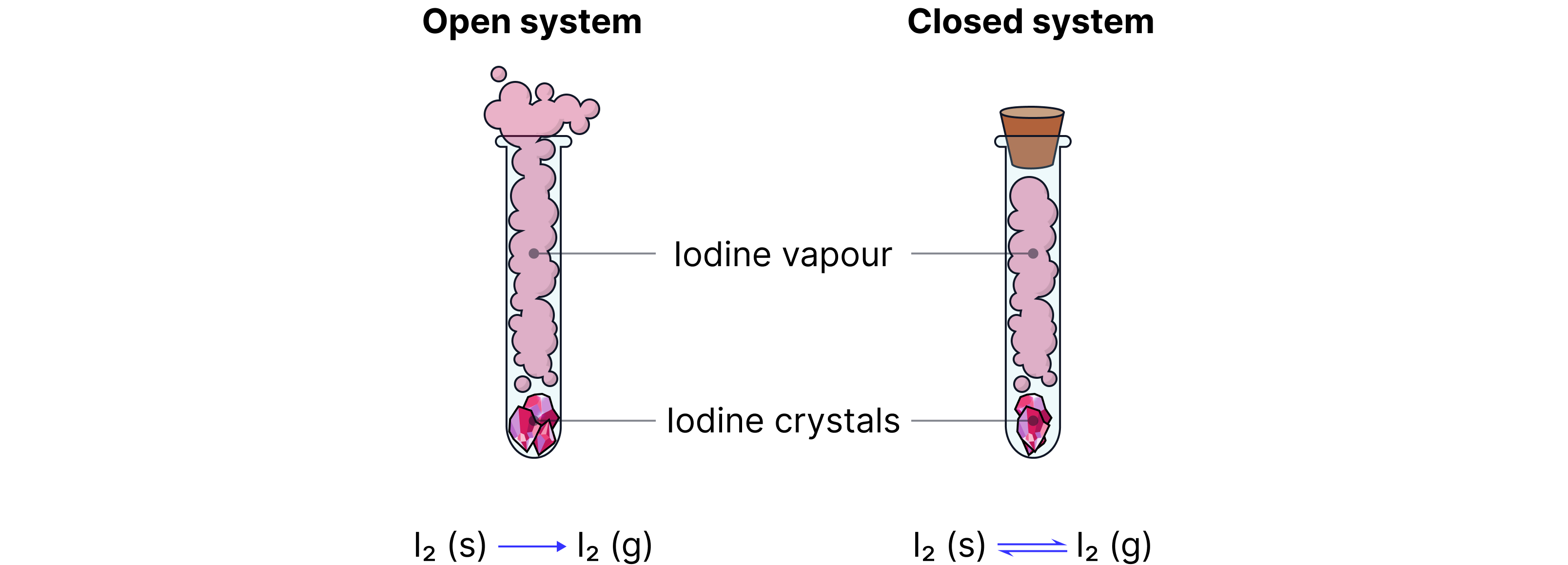
Why reversible reactions need a closed system
In a closed system:
Reactants and products stay together
Both the forward and reverse processes can keep happening
The system can reach a state where both directions occur at the same rate
(this idea leads into dynamic equilibrium, which you usually study next)
If the system is not closed:
A gas might escape
A solid might be removed
One of the substances might be lost to the surroundings
Then the change becomes effectively irreversible, because the particles needed to go backwards are no longer all there.
A Classic Example: NO₂ ⇌ N₂O₄
One of the best reversible reactions to visualise is the equilibrium between nitrogen dioxide and dinitrogen tetroxide.
NO₂(g) is brown
N₂O₄(g) is colourless or very pale
Because these gases have different colours, you can see the effect of the reaction shifting one way or the other.

The forward reaction (to the right)
Two NO₂ molecules join together
They form one N₂O₄ molecule
The gas becomes less brown (more colourless)
This direction is exothermic (releases energy to the surroundings)
The reverse reaction (to the left)
One N₂O₄ molecule splits into two NO₂ molecules
The gas becomes more brown
This direction is endothermic (absorbs energy from the surroundings)
Hydrated Copper(II) Sulfate
Another reversible reaction we should be familiar with involves hydrated copper sulfate and anhydrous copper sulfate.
CuSO₄ 5H₂O(s) = blue hydrated copper(II) sulfate
CuSO₄(s) = white anhydrous copper(II) sulfate
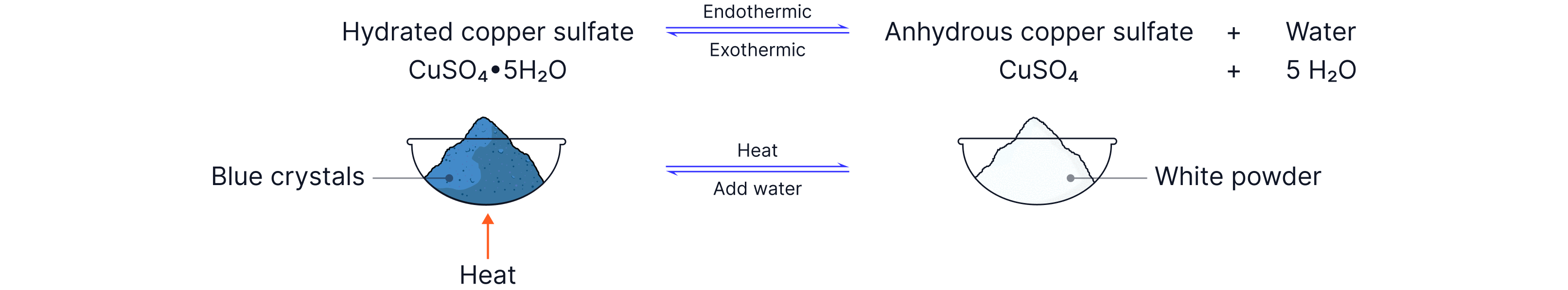
Forward direction (heating)
Heat drives out water
Blue crystals turn white
This is usually treated as endothermic
Reverse direction (adding water)
Adding water reforms the blue crystals
The change releases energy (feels warm)
This is exothermic
Check Your Understanding
Recall
What is a reversible reaction?
Answer:
A reaction in which products can change back into reactants
What does the double arrow (⇌) symbol show?
Answer:
The reaction can go in both forward and reverse directions
What is meant by a closed system?
Answer:
A system where no substances enter or leave
Application
Explain why reversible reactions must be carried out in a closed system.
Answer:
So reactants and products remain together; reverse reaction can occur; substances are not lost
In the reaction NO₂(g) ⇌ N₂O₄(g), identify which gas is brown and describe what happens to the colour when the reaction shifts to the right.
Answer:
NO₂ is brown; shifting right forms N₂O₄; mixture becomes less brown/more colourless
Describe what happens when hydrated copper(II) sulfate is heated and when water is added again.
Answer:
Heating drives off water; blue crystals turn white (anhydrous); adding water reforms blue hydrated crystals; heat released
Challenge (HT)
Explain why heating the NO₂ ⇌ N₂O₄ system makes the mixture darker brown, using ideas about energy and reversible reactions.
Answer:
Forward reaction is exothermic; reverse is endothermic; heating favours the endothermic direction; more NO₂ formed; mixture becomes darker brown
More Practice
Try to answer the practice question from the TikTok video on your own, then watch the video to see how well you did!