Equilibrium
Dr. Davinder Bhachu
Teacher

Chemical Equilibria
Reversible reactions are reactions in which the products can reform the reactants.
The forward reaction describes reactants being converted to products
The reverse reaction describes products being converted back to reactants
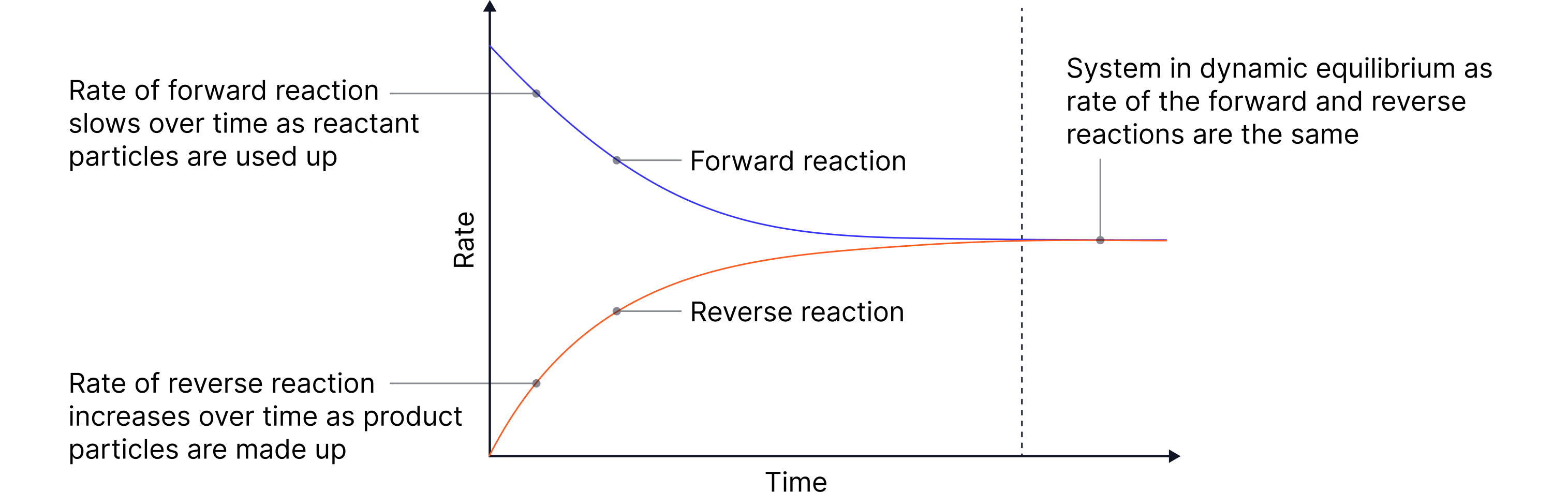
A dynamic equilibrium is established in a closed system when:
The forward and reverse reactions occur at the same rate
The concentrations of reactants and products remain constant
Features of Dynamic Equilibrium
The rate of the forward reaction is equal to the rate of the reverse reaction
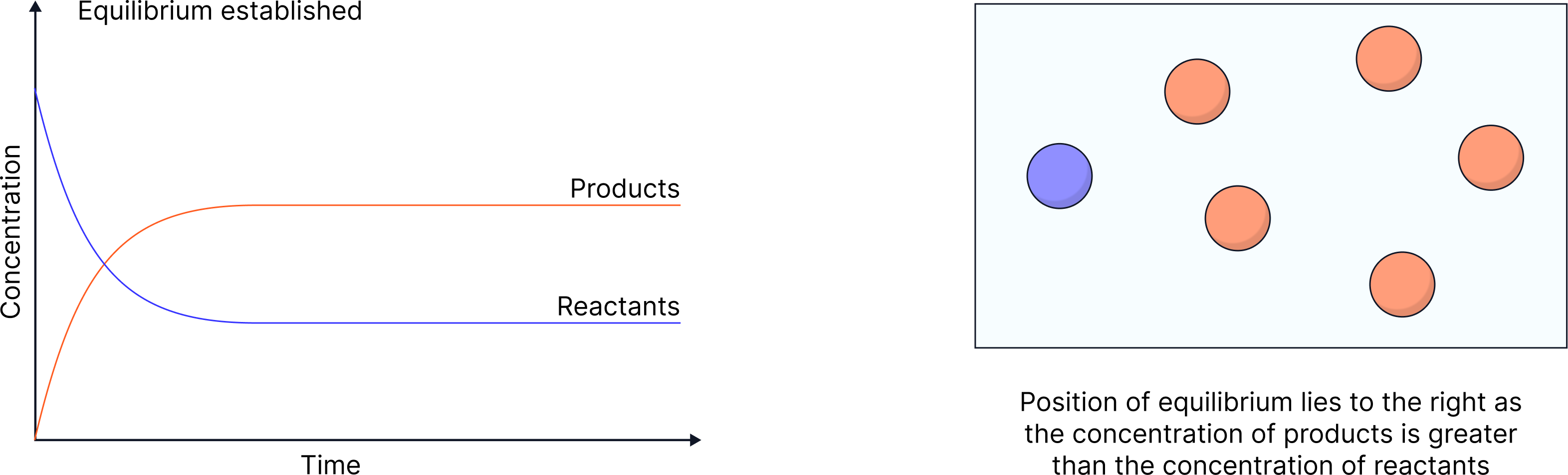
So, the concentration of products/reactants remains constant once equilibrium is reached
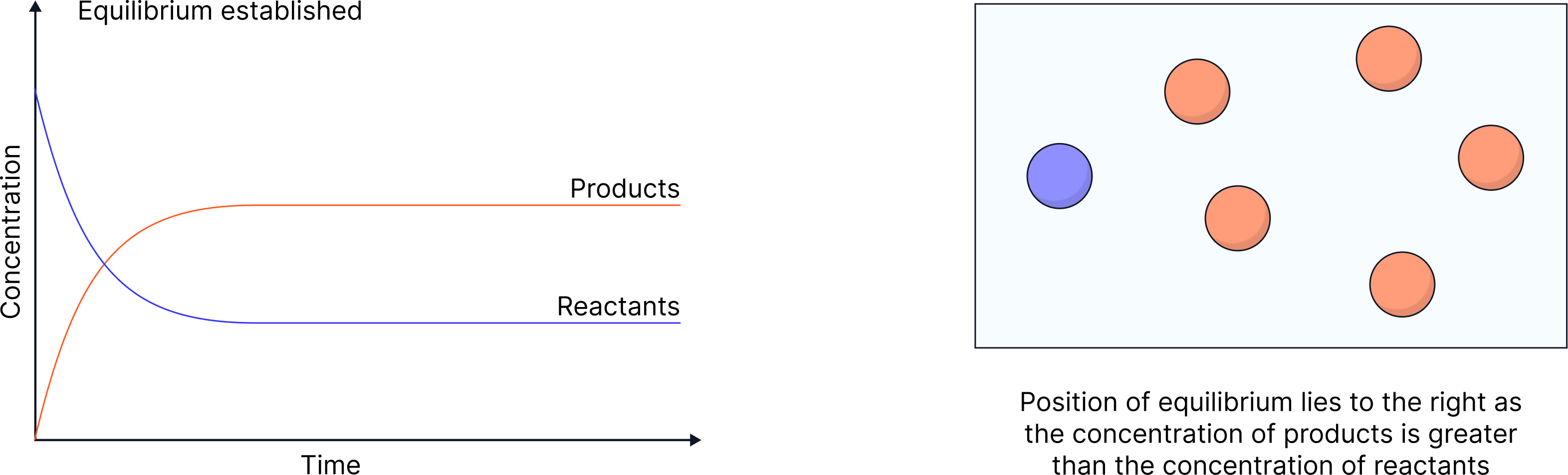
The concentration of products/reactants once equilibrium is reached is NOT necessarily equal
The position of equilibrium indicates which side is favoured:
More products → "lies to the right"
More reactants → "lies to the left"

Energy Transfer in Reversible Reactions
Opposite Energy Changes in Each Direction
Every reversible reaction has two paired energy changes:
If the forward direction is endothermic (absorbs energy), the reverse direction is exothermic (releases energy).
If the forward direction is exothermic, the reverse direction is endothermic.
Energy balance
The energy absorbed going one way equals the energy released going the other way.
Why This Happens
Chemical bonds break and form in both directions:
Endothermic direction: More energy must be supplied to break bonds.
Exothermic direction: More energy is released when bonds form.
Because the reverse process simply undoes the forward one, the energy change is symmetric but opposite.
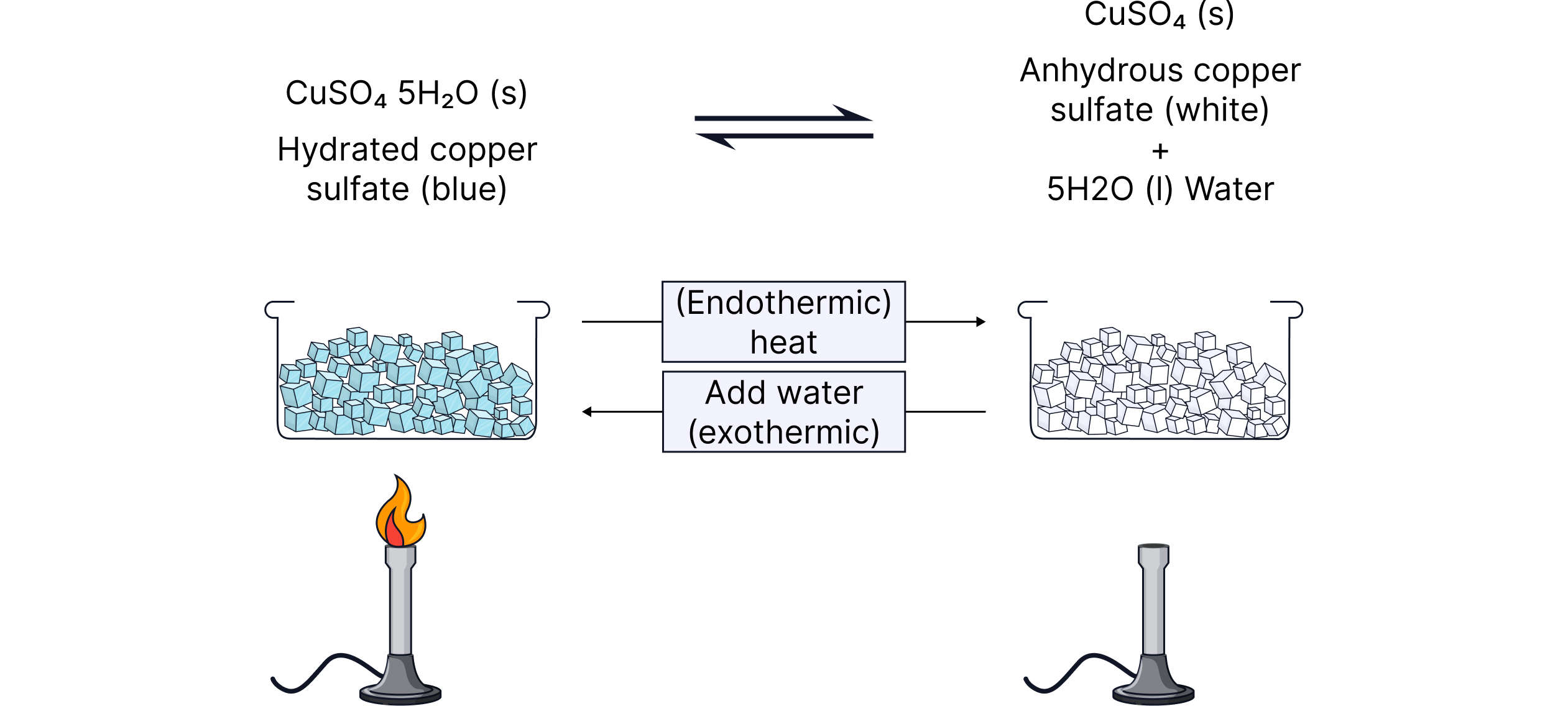
Example: Hydrated ⇌ Anhydrous Copper(II) Sulfate
Forward direction (heating)
Blue hydrated crystals lose water
Become white anhydrous copper sulfate
Endothermic — you must supply heat
Reverse direction (adding water)
White powder regains water
Blue crystals reform
Exothermic — heat is released