The composition And Evolution Of The Earth’s Atmosphere
Dr. Davinder Bhachu
Teacher

Contents
Evolution Of The Earth’s Atmosphere
The Atmosphere Today
Key Idea
Earth’s modern atmosphere has stayed fairly stable for millions of years. It is mostly nitrogen and oxygen, with small quantities of other gases.
Current Composition (Approximate)
- Nitrogen (~78%) – the dominant gas
- Oxygen (~21%) – essential for aerobic life
- Trace gases (<1%) – carbon dioxide, water vapour, noble gases (argon, neon, etc.)
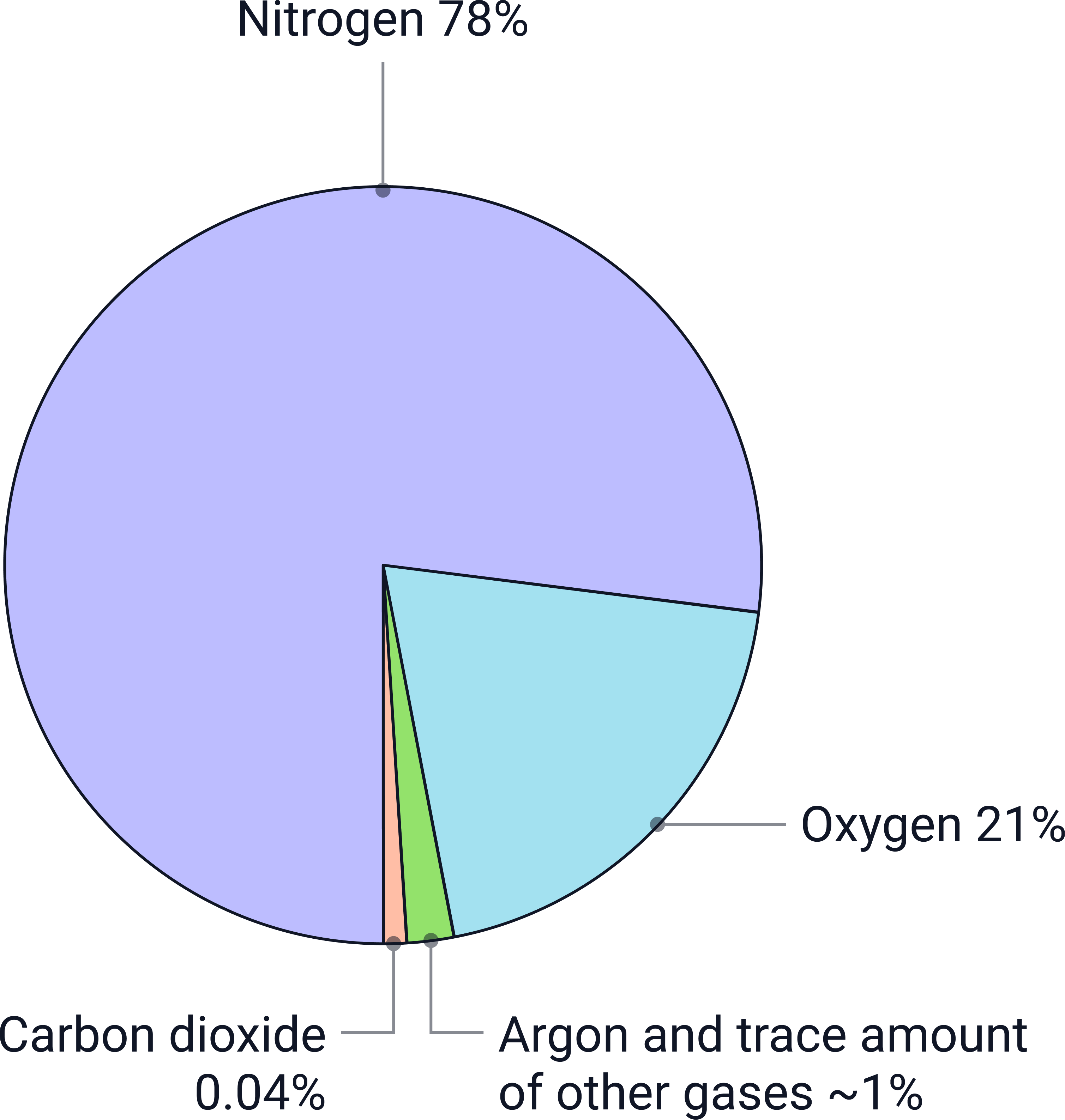
A simple visual comparison showing how nitrogen overwhelmingly dominates the modern atmosphere, followed by oxygen, with all other gases making up only a tiny fraction.
Before The Atmosphere Stabilised
A Hot Young Planet
- Early Earth was extremely hot with constant volcanic eruptions.
- No stable atmosphere existed yet — gases released by volcanoes gradually built one up.
Sources of Early Atmospheric Gases
Volcanoes released large amounts of:
- Carbon dioxide
- Water vapour
- Nitrogen
- Small amounts of methane and ammonia
This mix resembled the atmospheres of Mars and Venus today (high CO₂, almost no oxygen).
Cooling and Oceans Form
As Earth cooled:
- Water vapour condensed → oceans formed
- Dissolved volcanic gases entered the oceans
- A thin crust solidified, though volcanic activity continued
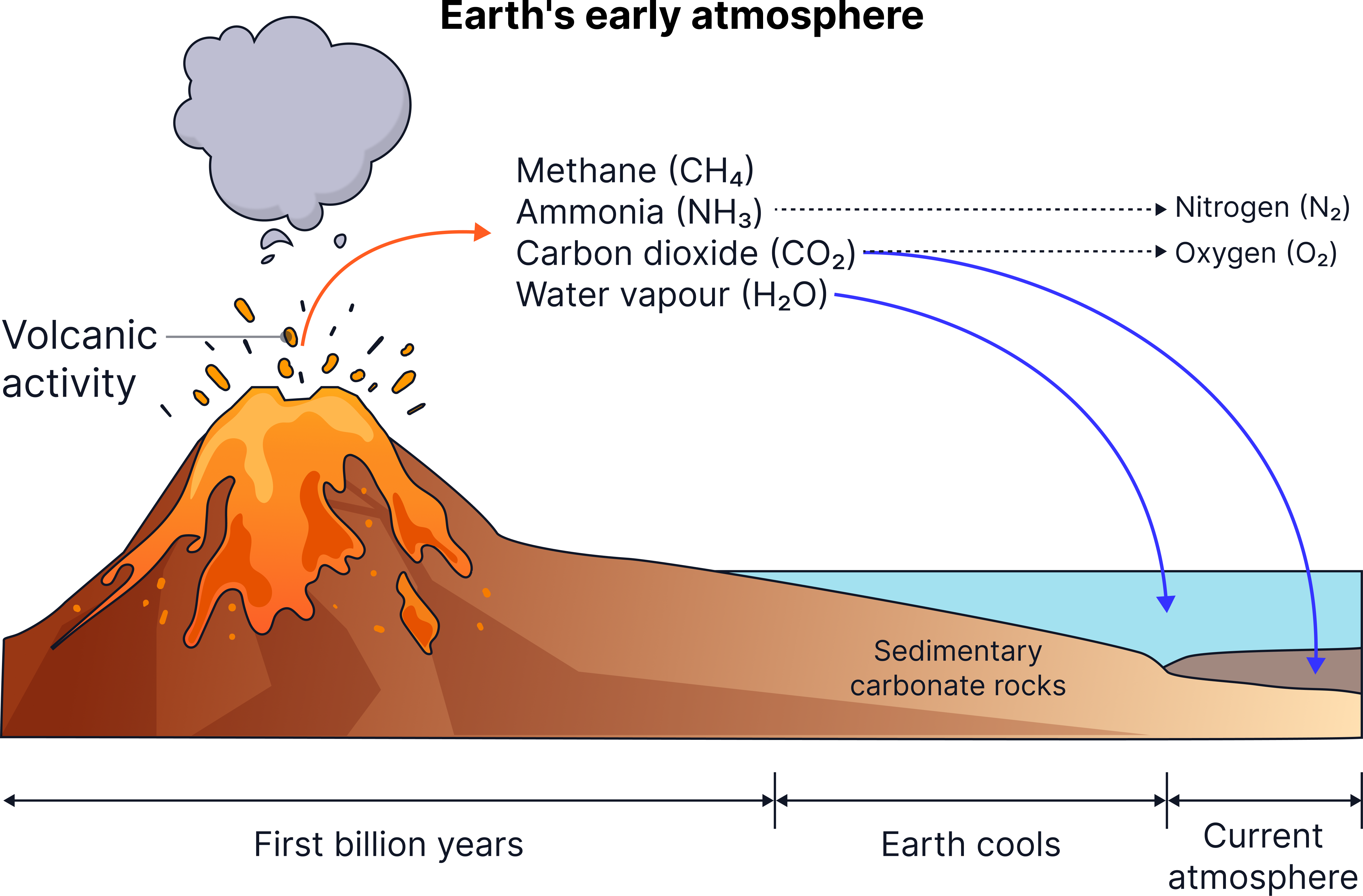
Illustration showing intense volcanic activity supplying gases that gradually accumulated to form an early, oxygen-poor atmosphere.
How Carbon Dioxide Levels Decreased Over Time
A. The Oceans Absorb CO₂
- CO₂ dissolves readily in water.
- Once dissolved, it reacts to form carbonates.
- Carbonates settle on the seafloor and become part of sediments.
Simplified Sequence
- CO₂ dissolves in seawater
- Carbonate compounds form
- These settle as sediments
- Over time these form sedimentary rocks
B. Life Begins to Help Remove CO₂
- Early algae appeared around 2.7 billion years ago.
- Later, simple plants evolved.
- Both used photosynthesis, which consumes CO₂.
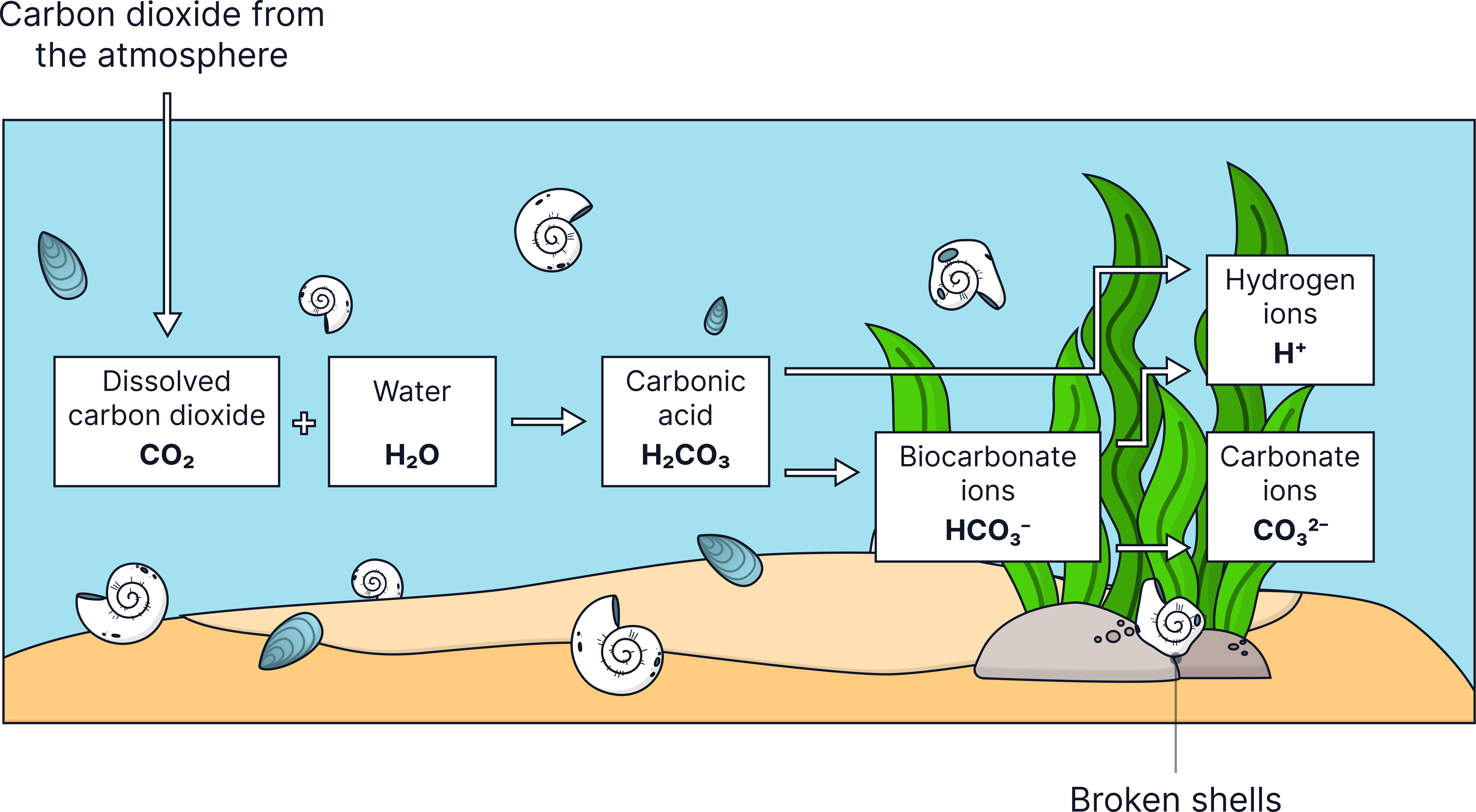
A sequence showing how carbon dioxide from the atmosphere ends up dissolved in seawater, reacts to form solid carbonates, and eventually becomes part of seabed sediments.
Locking Carbon Away (Long-Term Storage)
Formation of Sedimentary Rocks and Fossil Fuels
Over millions of years:
- Dead marine organisms and plant matter were buried under sediments.
- Pressure and heat converted them into:
- Coal – from ancient, compressed plant matter
- Crude oil and natural gas – from marine microorganisms trapped in sediments
These fuels store carbon for geological timescales, reducing CO₂ in the atmosphere.
Limestone Formation
- Marine creatures with shells made of calcium carbonate died and sank.
- Their remains accumulated and compressed into limestone.
- This effectively removed significant amounts of carbon from the atmosphere.

A piece of limestone showing embedded remains of ancient sea organisms, illustrating how biological material becomes locked away as rock.
How Oxygen Increased
Photosynthesis Changes Everything
Once plants and algae evolved, they used sunlight to convert CO₂ and water into glucose and oxygen:
Word equation:
carbon dioxide + water → glucose + oxygen
Symbol equation:
6 CO₂ + 6 H₂O → C₆H₁₂O₆ + 6 O₂
Impact on the Atmosphere
- Early oxygen first dissolved into oceans.
- Once saturated, oxygen began building up in the air.
- Higher oxygen eventually allowed the evolution of animals and more complex life forms.
- About 200 million years ago, the atmosphere settled into its modern composition.
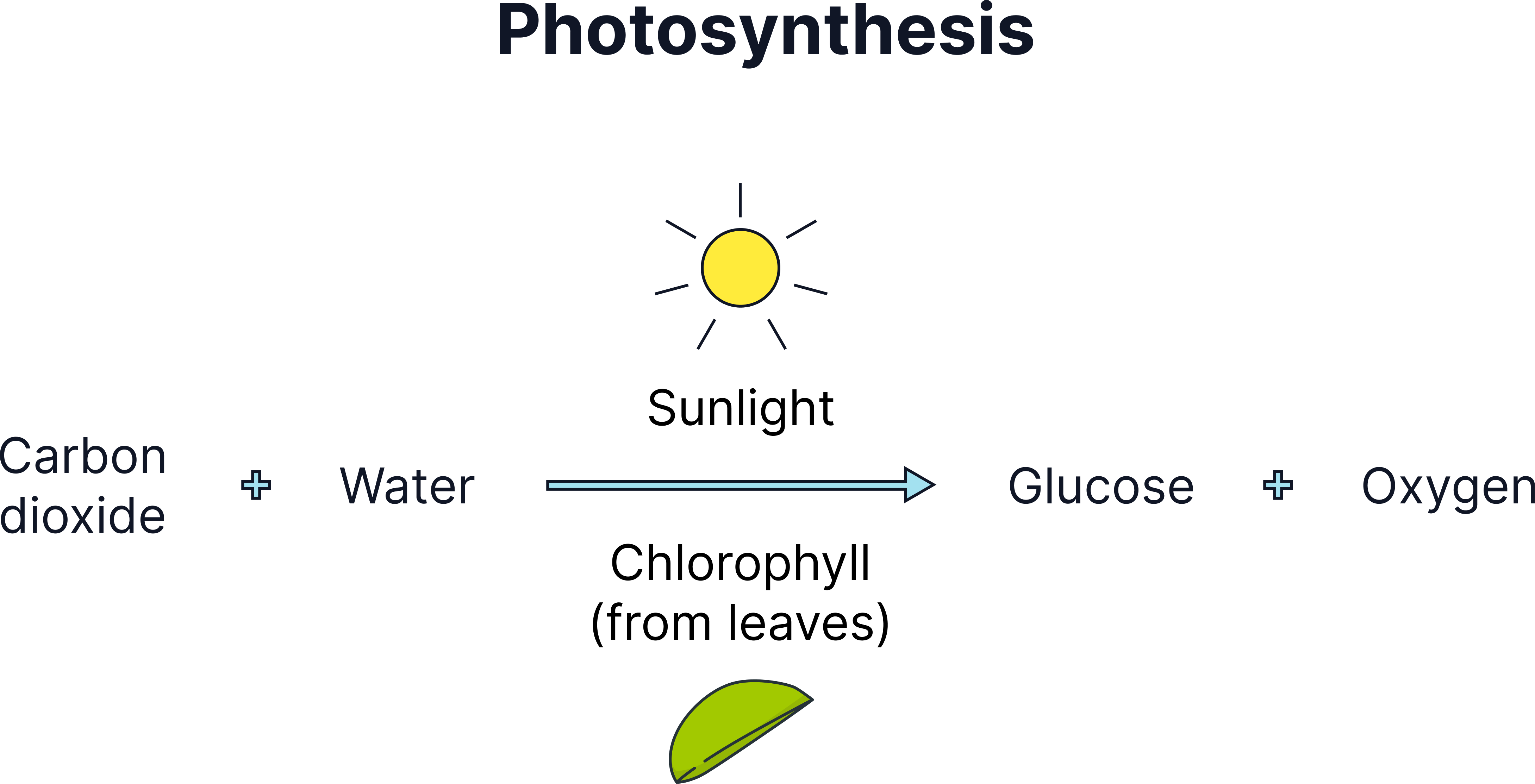
A process diagram showing how photosynthetic organisms use sunlight to create glucose while releasing oxygen into the atmosphere.
How Scientists Study The Atmosphere’s Evolution
- Nearly all direct evidence from early Earth is gone.
- Rocks from billions of years ago are rare because Earth’s crust is constantly recycled.
Therefore, scientists rely n:
- Indirect clues
- Comparisons with other planets
- Chemical signatures in ancient rocks
- Fossil evidence of early life
- Computer models simulating atmospheric change
Theories are updated as new evidence emerges, so there is no single “final” explanation.
Examples Of Evidence Used
1. Volcanic Gases Today
Modern volcanic emissions help scientists infer what ancient eruptions may have released.
2. Atmospheres of Other Planets
Mars and Venus have CO₂-rich atmospheres with almost no oxygen; they provide clues about what Earth may have been like before life evolved.
3. Ancient Rocks
Some rocks contain minerals that only form when oxygen is present. Their age indicates when oxygen began increasing.
4. Fossils of Early Organisms
Ancient microbes that didn’t need oxygen show that life existed before oxygen was abundant.
Summary
- The Earth’s earliest atmosphere was produced mainly by intense volcanic activity, which released large amounts of carbon dioxide and virtually no oxygen. This created an environment very different from the modern one.
- As the planet slowly cooled, water vapour in the atmosphere condensed and formed the first oceans. The presence of large bodies of water allowed carbon dioxide to begin dissolving into them, reducing its level in the air.
- Over time, the dissolved carbon dioxide took part in reactions in the oceans that produced solid carbonates. These settled on the sea floor and built up as sediments, eventually becoming carbonate-rich rocks that held carbon for millions of years.
- With the development of early life, photosynthetic organisms such as algae began to capture carbon dioxide and release oxygen. This biological activity marked the start of a gradual rise in atmospheric oxygen.
- As plants and algae continued photosynthesising over vast spans of time, oxygen concentrations increased to the point where more complex life—including animals—could evolve and survive.
- After these long periods of chemical and biological change, the atmosphere eventually reached a stable state, dominated by nitrogen and oxygen, resembling the composition we see today.
- Scientists build their understanding of this long evolution by examining modern volcanic gases, comparing Earth with the atmospheres of nearby planets, analysing ancient rocks for chemical clues, and studying fossils of early organisms. Each type of evidence offers insight into what the atmosphere was like at different stages of Earth’s history.
Check Your Understanding
Recall
a) State the approximate percentage of nitrogen in the modern atmosphere.
Answer:
Approximately 78%
b) Name three gases released by early volcanic activity.
Answer:
Carbon dioxide; water vapour; nitrogen (methane and ammonia also acceptable)
c) What process carried out by algae and plants increased oxygen levels in the atmosphere?
Answer:
Photosynthesis
Application
d) Explain why the formation of oceans caused carbon dioxide levels to decrease.
Answer:
Water vapour condensed to form oceans; CO₂ dissolved in water; formed carbonates; carbonates settled as sediments; reduced atmospheric CO₂
e) Describe how limestone formation helped remove carbon dioxide from the atmosphere.
Answer:
Marine organisms formed shells of calcium carbonate; shells accumulated on seabed; compressed into limestone; carbon locked away long-term
f) Explain how the evolution of photosynthetic organisms changed the composition of the atmosphere over time.
Answer:
Photosynthesis used carbon dioxide and water; released oxygen; CO₂ decreased; oxygen built up in oceans then atmosphere; allowed complex life
Challenge (HT Only)
g) Explain why scientists rely on indirect evidence to study the early atmosphere and describe two types of evidence they use.
Answer:
Early rocks are rare due to recycling of crust; little direct evidence remains; use volcanic gas comparisons; atmospheres of Mars/Venus; chemical signatures in ancient rocks; fossil evidence; computer models
More Practice
Try to answer the practice question from the TikTok video on your own, then watch the video to see how well you did!