Density RP
Brook Edgar & Hannah Shuter
Teachers


Explainer Video
Solids, Liquids and Gases
Solids, liquids, and gases are the three main states of matter. We can describe them by looking at how their particles are arranged and how they behave.
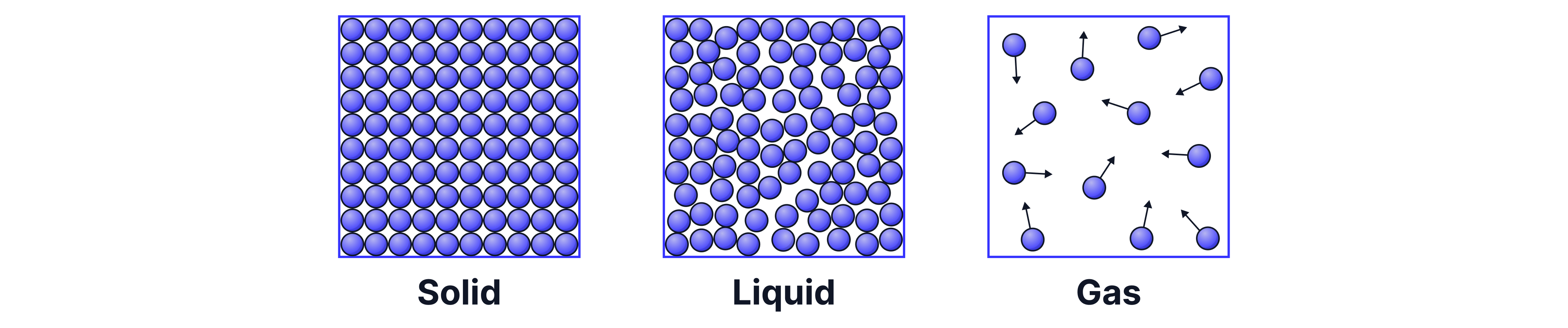
Solids have particles that are packed very closely together; they are all touching in a fixed, regular pattern. These particles can only vibrate. Because of this tight arrangement, solids have a fixed shape and take up the least amount of space (volume) -> they have the greatest density (more on this below).
Liquids have particles that are close together; they are touching but not in a fixed position - they are in an irregular arrangement. The particles can move and slide over each other, which allows liquids to flow and take the shape of their container.
Gases have particles that are far apart, not touching, and move freely and randomly in all directions. This means gases spread out to fill all the space available to them. Because of the large gaps between particles, gases take up the most volume of the three states -> they have the least density.
Density
Density is the mass per unit volume of a substance. A more dense substance has more particles packed together in a given volume -> a solid is more dense than a liquid.
Water has a density of about -> one meter cubed of water has a mass of Lead has a density of around -> one meter cubed of lead has a mass of . Lead is more dense than water, it has more particles in a given volume.
Formula:
For example if I wanted to calculate the density of a steel rod, knowing the mass is , and its volume is , I use the equation:
The density of our steel rod is greator than water so if the steel rod was placed in water it would sink.
If an object has a greater density than the fluid it is placed in, it will sink; if it has the same or a lower density, then it will float. Think about your rubber duck in your bath, as the duck floats, the rubber duck must have a lower density than water.
Take Note: At GCSE, density can either be given in terms of , or . To calculate density in you must use the density equation with mass in and volume in . To calculate the density in you must use the density equation with mass in and volume in .
Worked Example:
A large wooden log has a volume of . Its density is . Calculate its mass.
Answer:
Teacher Tip: The mass is in kilograms, as the density is given in .
Worked Example:
The density of water is . A ship has a mass of .
What is the minimum volume the ship must have to float?
is equivalent to .
Answer:
To float, the ship must have at least the same density as the water it is floating in, . So to find the minimum volume, we use the maximum density the ship can have ->.
Density RP
To calculate the density of any material you need to determine both its mass and its volume, as . The mass is measured using a balance/scale, but the method used to find the volume depends on the type of object you are measuring.
Finding the Density of a Regular Shape:
Measure the mass of the object using a balance.
Find the volume by measuring the length, width, and height, using a ruler, and multiplying these three values together.
Use the equation to calculate the density.

Here we used a ruler to measure the lengths, but if the object is very small, we need to use different equipment. For measurements shorter than , a micrometer or Vernier calliper should be used as they can measure much smaller distances.

Finding the Density of an Irregular Shape:
The mass of an irregular object can be measured using a balance, but its volume cannot be found with a ruler as it can with a regular solid. As the sides are all different shapes, they are not straight. Imagine a rock; you could not measure its length using a ruler. Instead, to find the volume, we use the principle of displacement -> an object submerged in a liquid causes the liquid to rise by an amount equal to the object's volume. We can see this when you get into a bath: the water level rises. The water level will rise more for an adult (who has a bigger volume) than for a child (who has a smaller volume).
There are two main methods for determining volume by displacement:
-> Displacement Can (also known as the Eureka can).
Measure the mass of the object using a balance.
Fill the displacement can with water until it begins to flow out (the water is now level with the spout).
Gently lower the object into the can; the water displaced by the object flows out of the spout and can be collected in a measuring cylinder. This is the object's volume.
Use the equation to calculate the density
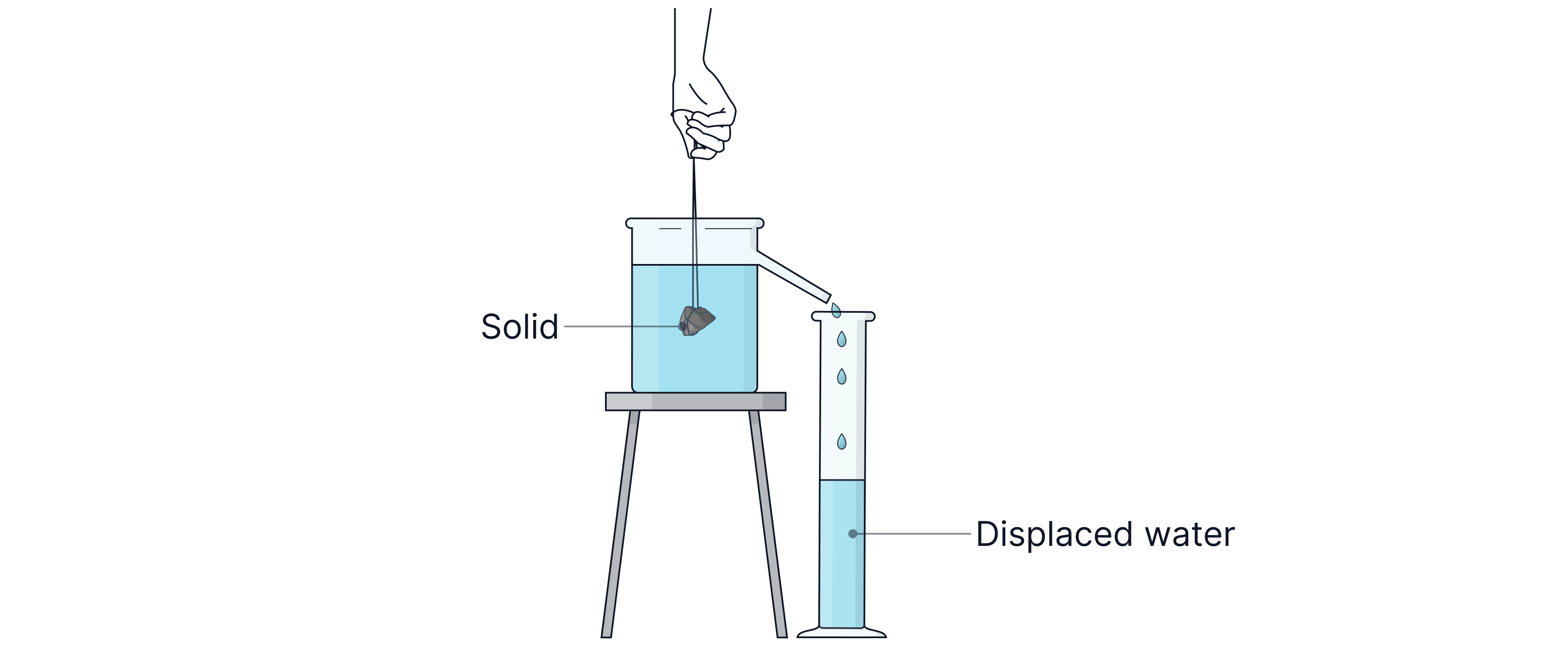
-> Measuring Cylinder
Measure the object's mass using a balance.
Measure the initial volume of the water in the measuring cylinder.
Place the object into the measuring cylinder and measure the final volume of the water.
Use the equation to calculate the density

Take Note: If the object is less dense than water and will not sink, meaning we cannot find its volume, we need to attach a dense object with a known volume to our object to make it sink, then subtract its volume from the final volume.
Finding the Density of a Liquid:
Find the liquid's volume by pouring it into a measuring cylinder.
Measure its mass using a balance (place the empty measuring cylinder on the balance and zero it, so that when you fill the measuring cylinder with liquid, you only get the liquid's mass, not the mass of the liquid and the measuring cylinder).
Use the equation to calculate the density
Worked Example:
A teacher wants to find the density of a ring pull.
She collects ring pulls and measures them on a balance as shown below.

Calculate the mass of a single ring pull.
To find the volume of a ring-pull the teacher uses a measuring cylinder as shown in the pictures. The measuring cylinder is marked in .
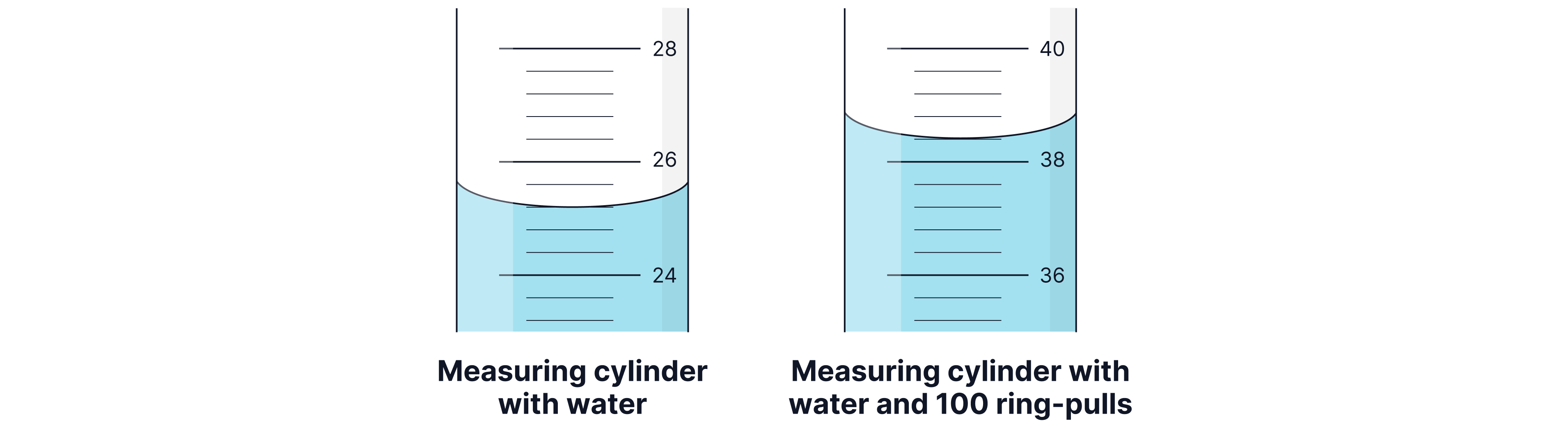
Determine the volume of a single ring-pull. Remember to read the volume from the bottom of the curve (the miniscus).
Using your values of the mass and volume of a ring-pull and the table below, determine which metal the ring-pulls are made from.
Metal | Density |
Stainless steel | 7.9 |
Iron | 7.2 |
Aluminium | 2.7 |
Tin | 7.3 |
Answer:
First, you need to find the change in mass between the empty beaker and the beaker filled with ring pulls. This will give us the mass of ring pulls.
Finding the mass of a single ring pull:
First we need to find the difference between the measuring cylinders before and after the ring pulls were added:
Finding the volume of a single ring pull:
So the material is aluminium.
Challenge Question:
A student wants to determine the volume of a coin.
Volume = surface area thickness
They need to know the coin's diameter to calculate its surface area using the equation . The diameter of the coin is . The thickness of one coin is too small to measure with a ruler.
Suggest how the student could find the thickness of a single coin.
Explain how the student will use the measurements to determine the volume.
Answer:
The student could stack coins together, measure the thickness of this stack using a micrometer, then divide the thickness by to find the thickness of one coin.
As volume = surface area thickness, they need to first divide the diameter of the coin by two to get its radius, and then use the equation to find the surface area of one coin face. As a coin has two faces, they need to multiply this answer by two and then by the thickness of the coin to get its volume.
Practice Questions
A student investigates the density of a metal block. The block has a mass of . Its dimensions are: length = , width = , height = .

Calculate the volume of the block in .
Calculate the density of the block in .
-> Check out Hannah's video explanation for more help.
Answer:
A student wants to calculate the density of a small irregular-shaped stone. Describe the measurements the student should take.
-> Check out Hannah's video explanation for more help.
Answer:
Measure the mass using a balance
Place some water in a measuring cylinder and record the initial volume. Carefully lower the stone into the water.
Record the new volume of water.
(displacement method).
Once the volume is known, density can be calculated using the formula: