Changes in State
Brook Edgar & Hannah Shuter
Teachers


Contents
Explainer Video
States of Matter
There are three main states of matter: solids, liquids, and gases. All matter is made of tiny particles (atoms/molecules).
In solids, the particles are all touching in a regular arrangement and can only vibrate in place. They are held together by strong intermolecular forces (forces of attraction between the molecules/particles). Their volume is fixed.
When a solid is heated, like putting a saucepan on the stove, the particles gain kinetic energy, so their vibrations increase, but they remain in fixed positions. Because there is very little space between the particles, solids cannot be compressed.

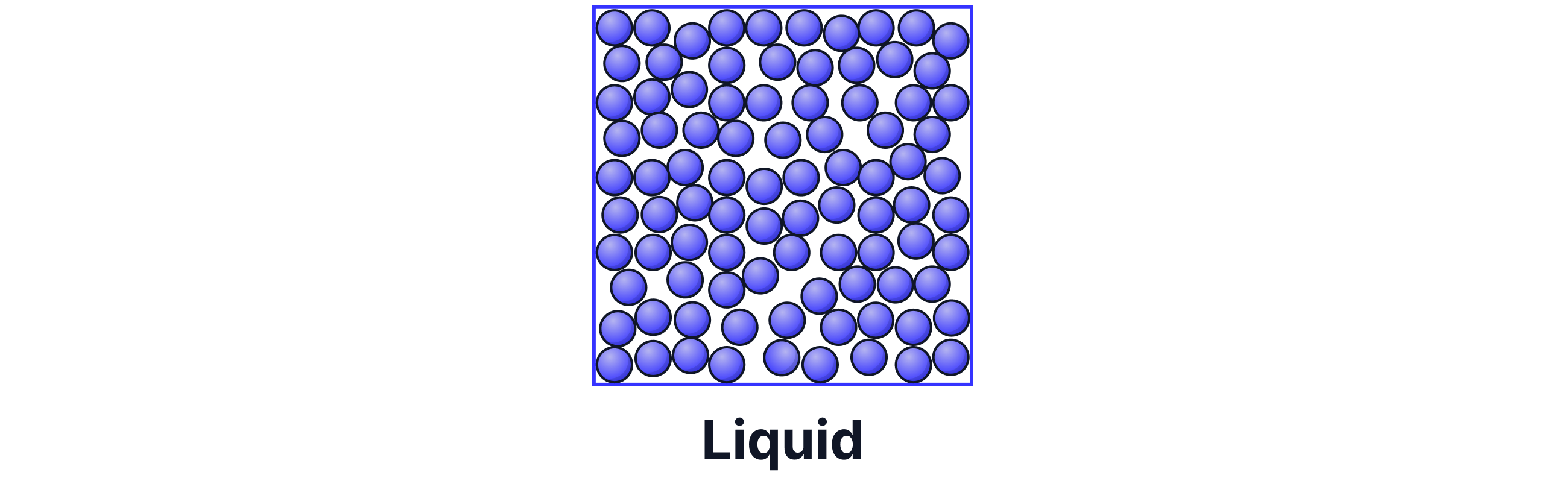
In liquids, the particles are all touching, but the intermolecular forces between them are weaker, allowing the particles to slide past one another. They move randomly. Liquids do not have a fixed shape and instead take the shape of their container.
As a liquid is heated, the particles gain kinetic energy and move faster.
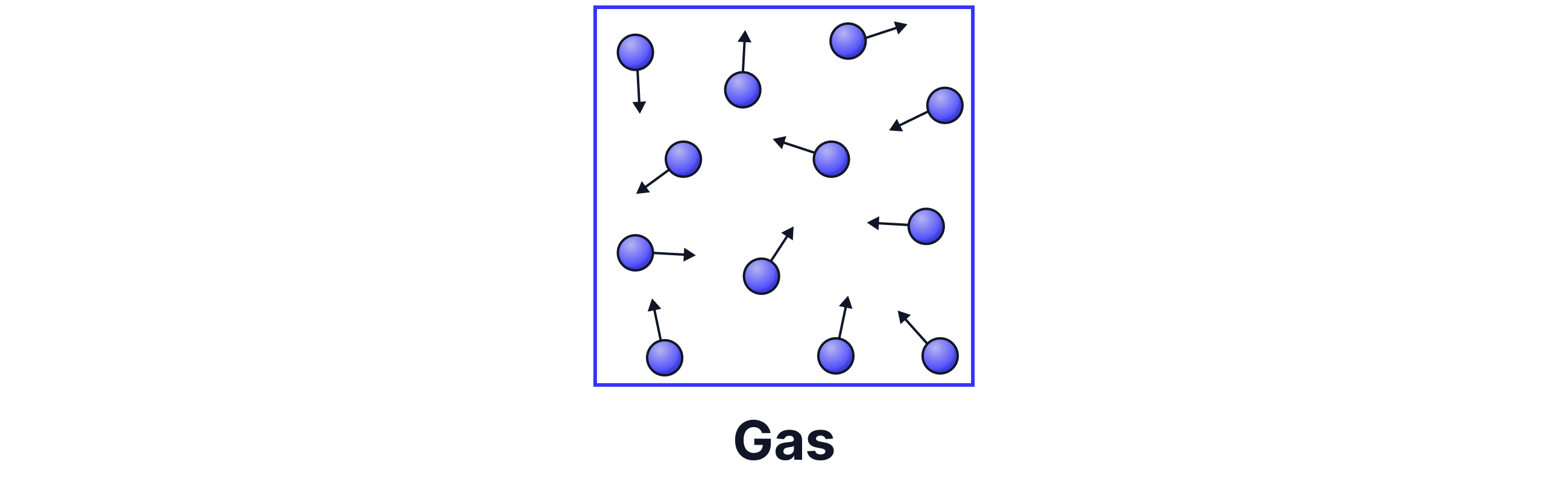
In gases, particles do not touch because they are far apart. There are no forces of attraction between the particles, so the particles move freely and randomly in all directions. They move rapidly, spreading out to fill their entire container. When a gas is heated, its particles gain even more kinetic energy and move faster still.
Worked Example:
Complete the table with the correct state of matter and the explanation:
Property | State / States | Explanation |
Keeps its shape | ||
Cannot be compressed / volume is fixed | ||
Expands to fill the container | ||
Can be compressed | ||
Flows and takes the shape of the container | ||
Highest density | ||
Lowest density |
Answer:
Property | State / States | Explanation |
Keeps its shape | Solid | Strong forces between the particles |
Cannot be compressed / volume is fixed | Solid and Liquid | Particles are all touching |
Expands to fill the container | Gas | Particles move randomly in all directions |
Can be compressed | Gas | Have space between particles |
Flows and takes the shape of the container | Liquid | Weaker forces between particles so they can flow over one another |
Highest density | Solid | Lots of particles in a small volume |
Lowest density | Gas | Fewer particles in the same volume than a solid or a liquid |
Worked Example:
A solid metal object is heated and does not melt (remains a solid). Explain what happens, if anything, to the arrangement and movement of particles in the metal.
Answer:
Particles in a solid vibrate around fixed positions. As the solid is heated up, the particles will gain kinetic energy and their vibrations will increase, but the arrangement of the particles will not change. They still have a fixed, regular arrangement.
Kinetic Theory
Kinetic Theory describes matter as being made of tiny particles that are constantly moving. Differences in the arrangement and movement of particles in a solid, a liquid, and a gas can be explained using kinetic theory.
Solids and gas particles differ as solid particles are close together, touching, vibrating about fixed positions as they are held together by strong forces of attraction, so solid particles can not be compressed. Gas particles are far apart, moving randomly as there are no forces of attraction between them, so they can be forced closer together -> they can be compressed.
Kinetic theory explains why equal masses of a solid and a gas will take up different volumes. Particles in a solid have strong forces of attraction between the particles, so they are held close together, touching, in a fixed arrangement, whereas particles in a gas spread out; they are not close together, as there are no forces of attraction between the particles.
Worked Example:
Use your knowledge of kinetic theory to explain the spacing between particles and the movement of particles in a gas.
Answer:
In a gas, the particles are far apart as there are no forces of attraction between them. A gas can therefore be compressed. The particles move randomly and spread out in all directions to fill their container.
Changes of State
The state of matter can be changed by heating or cooling. The diagram below gives the names of the changes of state:
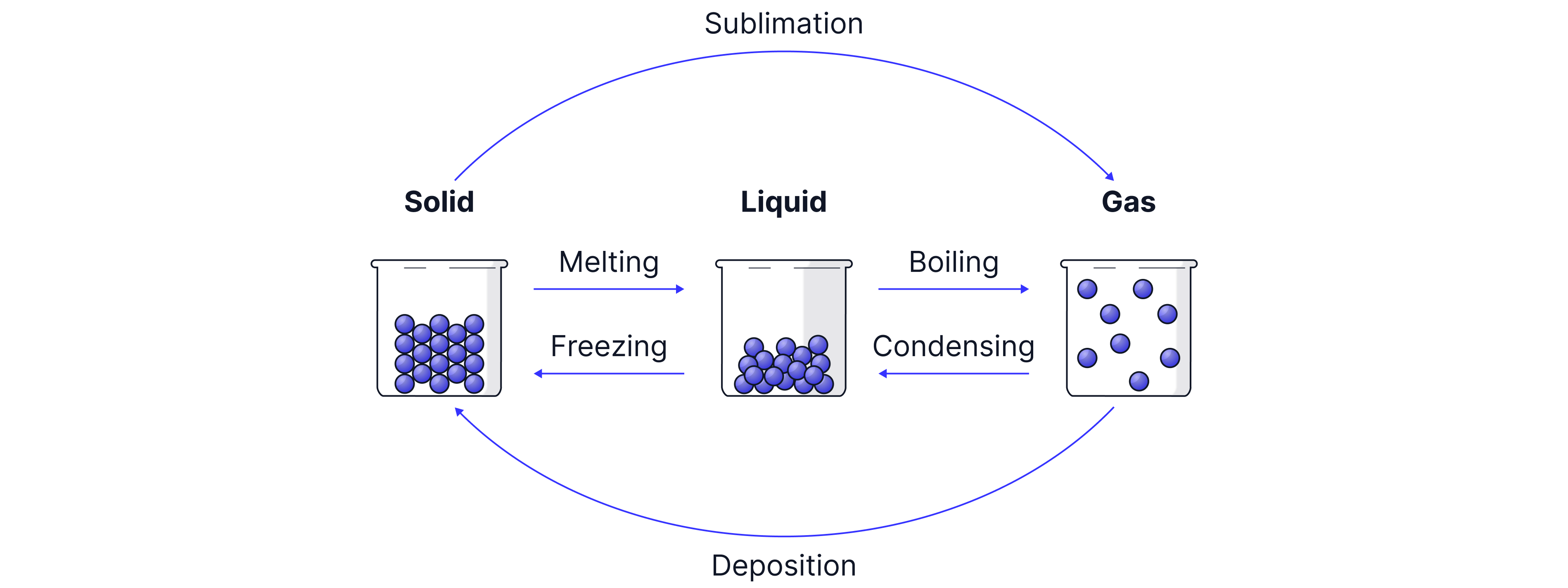
The same temperature that a solid changes state to a liquid -> the melting point, is the same temperature that a liquid changes state to a solid -> the freezing point. For water this is .
The temperature at which condensation occurs is the same as the temperature at which boiling occurs. For water this is .
Physical and Chemical Changes
Changes of state are considered physical changes because they do not produce new substances and can be reversed. In contrast, chemical changes involve chemical reactions that create new substances, and these changes are typically not reversible.
Conservation of Mass
Mass is conserved in all processes, including changes of state. A common misconception is that mass is lost when a liquid boils because, if the experiment is carried out on a balance, the measured mass appears to decrease. This happens because the gas particles escape into the air, so they are no longer pushing down on the balance. If the gas were captured and condensed back into a liquid, you would find that the total mass is exactly the same as it was before boiling.
Remember: There is a difference between evaporating and boiling.
-> Evaporation happens at the surface of a liquid and can occur at any temperature. For example, the liquid particles on the surface of your hot tea gain enough energy to become gas particles, which happens below .
-> Boiling happens throughout the liquid, not just at the surface, and occurs at the boiling point. The entire liquid gains enough kinetic energy to change state into a gas.
Worked Example:
Mercury has a melting point of and a boiling point of , Chlorine has a melting point of and a boiling point of . What state are these two materials in at:
Answer:
Mercury would be above the melting point but below the boiling point, so it will be a liquid. Chlorine would be above its boiling point so it would be a gas.
Mercury would be below its melting point, it has not melted yet, so is a solid. Chlorine would be between its melting point and boiling point, so it would be a liquid.
Mercury would be between its melting point and boiling point, so it would be a liquid. Chlorine would be above its boiling point, so it has boiled to become a gas.
Practice Questions
The melting point of iron is . Describe the arrangement and movement of iron particles above this temperature.
-> Check out Hannah's video explanation for more help.
Answer:
Above , iron is in the liquid state. The particles (atoms) are no longer in a fixed, regular pattern like in a solid. Instead, they are randomly arranged and not held in fixed positions. The particles have more energy and can move freely past each other. They are constantly moving in all directions, though they are still close together.
A student heats a beaker of liquid until it fully evaporates. The mass of the liquid before heating is . After evaporation, the student places a cold surface above the beaker and the vapour condenses back into liquid.
Explain why the total mass of the substance remains the same during evaporation and condensation.
Explain why evaporation and condensation are examples of physical changes rather than chemical changes.
-> Check out Hannah's video explanation for more help.
Answer:
Mass is conserved because the particles themselves are not created or destroyed. The substance changes state but the number of particles stays the same in each state.
No new substance is formed during the process. The original properties of the substance can be recovered when the state change is reversed.