Particle Motion in Gases
Brook Edgar & Hannah Shuter
Teachers


Explainer Video
Gas Pressure
In gases, particles move rapidly and randomly in all directions. There are very weak attractive forces between them, but as they are so weak, we say these forces are negligible (so small that we can ignore them).
The temperature of a gas is directly linked to the kinetic energy of its particles. Kinetic energy is the energy due to motion, so the faster the particles move, the more kinetic energy they have. As the temperature increases, so does the speed (and energy) of the particles.
Gas pressure is caused by collisions between gas particles and the walls of their container. Each time a particle hits the wall of its container, it exerts a tiny force. With billions of collisions happening every second, this adds up to a measurable pressure, as pressure is defined as force per unit area.
For example, the air around us is made of gas particles constantly bouncing off our bodies. This produces atmospheric pressure. The gas inside a balloon constantly collides with the walls of the balloon, keeping it inflated.
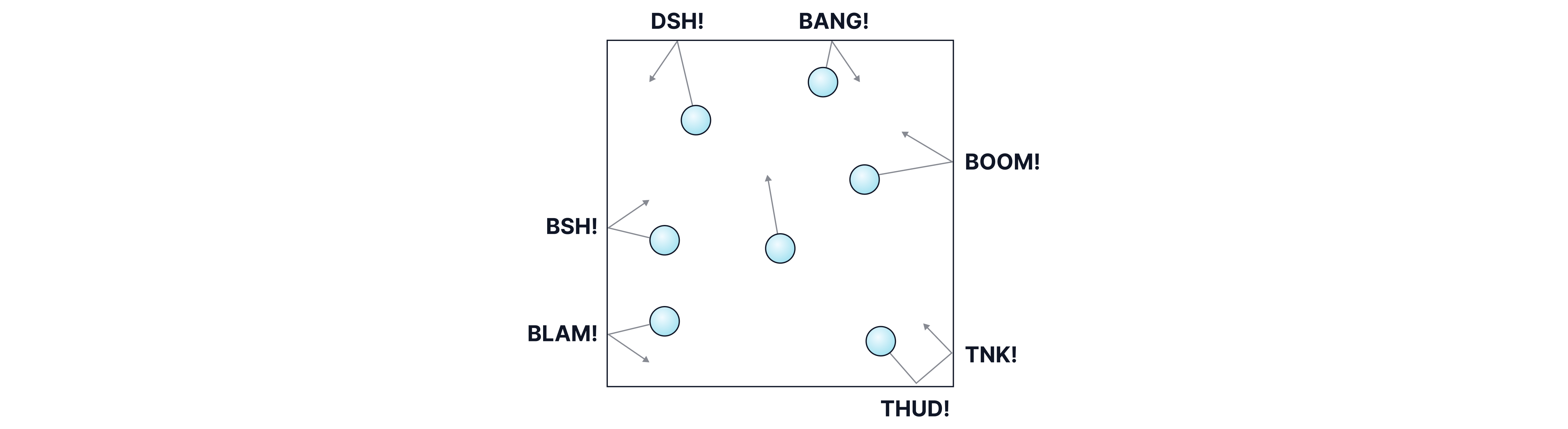
Gas pressure is affected by three main factors: temperature, particle number, and volume. For combined science, you only need to know how temperature affects pressure. In exam questions, they might talk about "sealed containers" or "constant volume", this is just so you know the only thing affecting the pressure is the temperature.
Temperature
Imagine a sealed container of gas. If we increase the temperature:
Particles gain kinetic energy and move faster.
They hit the container walls more often, so they exert more force on a given area.
This leads to higher pressure.
If we cool the gas down:
Particles move more slowly.
Collisions are less frequent and therefore less force is exerted on a given area.
The pressure decreases.
Reminder: Any answer regarding gas pressure should include a reference to the frequency of collisions between particles and the container walls.
Worked Example:
A sealed container full of air is heated. State and explain what happens to the pressure inside the container.
Answer:
As the temperature increases, the kinetic energy of the particles increases. This means they are moving faster. The particles collide more frequently and with more force with the walls of the container. This increases the air pressure.
Challenge Question:
Explain why the pressure doesn’t change if an unsealed container of air is cooled.
Answer:
Unsealed means that air can flow freely into and out of the container. As the container cools, the particles' kinetic energy decreases, causing them to slow down. The pressure inside the container momentarily drops as there are fewer collisions, but because the pressure inside is now lower than outside, more air particles move into the container, leading to more frequent collisions and a rise in pressure again. Overall, the pressure then doesn't change.
Practice Questions
A sealed container holds a fixed mass of gas at constant volume. The gas is heated from to .
State how heating the gas affects the motion of its molecules.
Explain why the pressure of the gas increases when the temperature rises.
-> Check out Hannah's video explanation for more help.
Answer:
The molecules have greater average speed.
Molecules collide more frequently with the container walls. Each collision exerts a greater force because the molecules are moving faster.
A student states: “If the temperature of a gas is reduced, the pressure must increase at constant volume.”
State whether the student’s statement is correct and explain your answer using the particle model of gases.
-> Check out Hannah's video explanation for more help.
Answer:
The student is incorrect. Lower temperature means molecules have less kinetic energy and move more slowly. They collide with the container walls less often and with less force, so pressure decreases.