Pressure in Gases
(Triple Only)
Brook Edgar & Hannah Shuter
Teachers


Explainer Video
Pressure and Volume
Gas pressure is caused by particles colliding with surfaces. Each time a gas particle hits the wall of a container, it exerts a force at right angles to the surface of the wall/container. The more frequent these collisions, the greater the force exerted on a given area, the greater the pressure.
If we place particles in a sealed box and decrease the volume, the particles are squeezed closer together. This increases the frequency of the collisions as they travel less distance before hitting the wall of the container, thus increasing the pressure. Think of trying to squash a balloon: as you try to reduce its volume, the pressure becomes so great that the balloon may pop!
As volume decreases, the pressure increases. We can see this on the graph below.
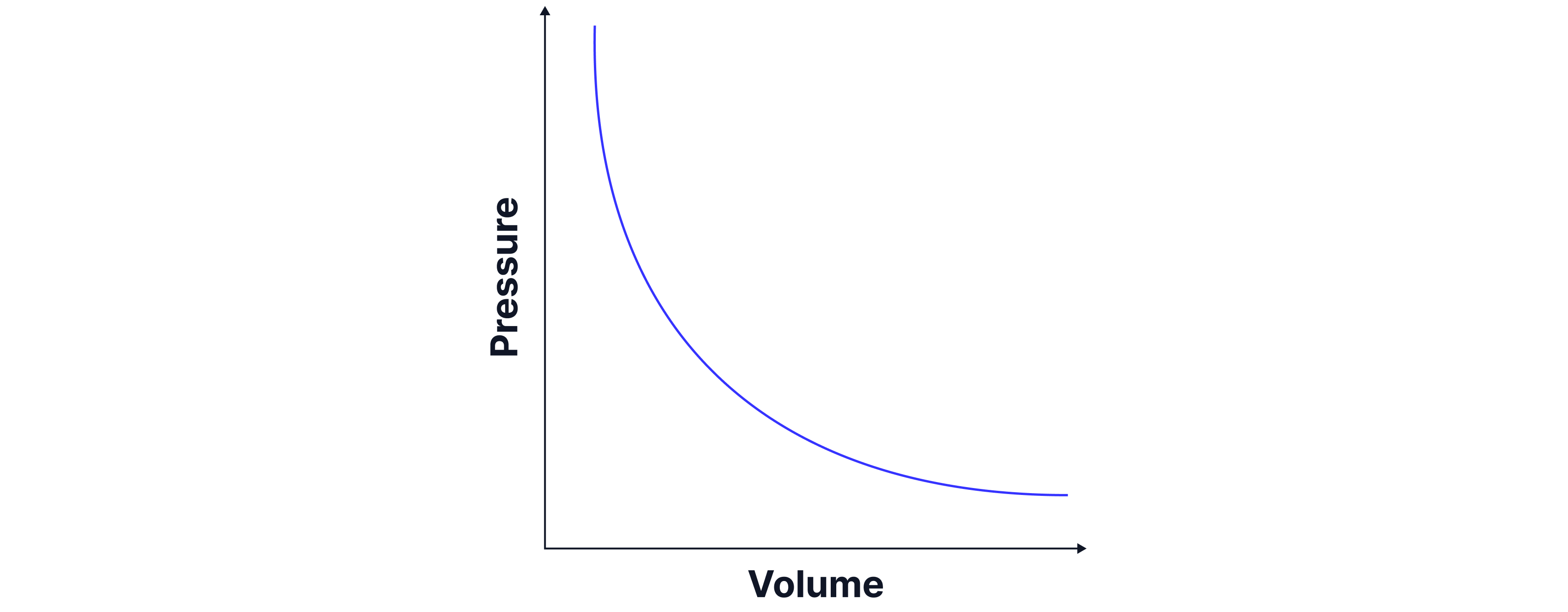
The graph above is plotted from the table of data below, where we can see that if the temperature and number of particles stay the same, that pressure volume always equals the same value -> a constant.
Formula:
Pressure / | Volume / | Pressure x volume / |
100 | 36 | 3600 |
120 | 30 | 3600 |
150 | 24 | 3600 |
180 | 20 | 3600 |
Because pressure times volume is constant, if the volume decreases by half, the pressure must double. Conversely, if the pressure halves, the volume must double. This is called an inversely proportional relationship.
Example: I can use this equation to work out the new volume of a sample of gas at , when it started at and a volume of .
We first need to calculate the constant:
The new pressure multiplied by the new volume will also equal , the constant, so we can work out the new volume:
Remember: We don't need to convert our units when using this equation - if the volume given/used is in , we will get our volume back in . You just need to be consistent across the equation.
Worked Example:
The pressure of a sample of gas in a sealed container is changed with the temperature remaining the same. Which statement is correct?
A. If pressure is doubled, the volume also doubles.
B. If pressure is halved, the volume is doubled.
C. If pressure is halved, the volume is also halved.
D. If pressure is doubled, the volume reduces to a quarter.
Answer:
Pressure and volume and inversely proportional, so if one factor increases, the other decreases at the same rate. This means our answer is B - as the volume doubles, the pressure halves.
Worked Example:
In an experiment of gas was at an initial pressure of .
Calculate the new volume if the pressure is dropped to at a constant temperature.
Answer:
First - we need to get our pressures into the same unit - either both into Pa or both into kPa.
Then we need to find the constant before the pressure is changed:
Now we can use this constant to find the new volume:
As our initial volume was in , our final volume will also be in .
Worked Example:
A sealed balloon is compressed. Explain, in terms of particles, what happens to the pressure inside the balloon.
Answer:
As the balloon is compressed, the volume decreases. This means the particles are forced closer together. There is less distance in between container walls, so the particles collide more frequently. This increases the pressure.
Work Done on a Gas
When we compress a gas, we force its particles into a smaller volume. This process requires us to do work on the gas -> to transfer energy to the gas particles.
As a result, the particles gain kinetic energy, meaning they move faster. As temperature correlates to the average kinetic energy of the gas particles, the temperature of the gas increases.
Think about using a bicycle pump. As you pump, you push more air into the tyre, increasing the pressure because there are more frequent collisions between the tyre walls and air particles. As work is done on the gas as you push it into the tyre, the kinetic energy of the particles increases. The air particles have a greater mean (average) kinetic energy -> the temperature of the gas increases. This is why the pump and the valve often feel warm after inflating a tyre.
Worked Example:
Describe how inflating a balloon can increase the temperature of the gas inside the balloon.
Answer:
To inflate a balloon, you have to push more gas particles in to increase the pressure. When you push the gas particles, you will do work on them, so energy will be transferred to the particles. This increases the kinetic energy of the gas particles, which increases the temperature of the gas inside the balloon.
Worked Example:
A carbon dioxide gas cylinder contains of gas at a pressure of .
Calculate the volume of the gas at atmospheric pressure, .
Suggest why the pressure of the gas must be increased slowly.
Answer:
The pressure must be increased slowly so that energy is transferred to the gas slowly. This means that the temperature of the gas will not increase very much.
Practice Questions
A sealed syringe contains a fixed mass of gas at a constant temperature. The volume of the gas is and the pressure is .
State the relationship between pressure and volume for a fixed mass of gas at constant temperature.
Calculate the new pressure if the volume is reduced to .
Explain why decreasing the volume increases the pressure, in terms of particle collisions.
-> Check out Hannah's video explanation for more help.
Answer:
As volume increases, pressure decreases. They are inversely proportional.
Particles collide with the walls more frequently, increasing the pressure
A student compresses a fixed mass of gas inside a pump. Initially, the gas has a pressure of and volume of . The temperature remains constant. Calculate the new volume of the gas if the pressure increases to .
-> Check out Hannah's video explanation for more help.
Answer: