Internal Energy
Brook Edgar & Hannah Shuter
Teachers


Explainer Video
Internal Energy
The internal energy of a substance is the total energy stored in its particles. It is made up of two parts:
Kinetic energy – from the motion of the particles
Potential energy – from the positions of the particles relative to one another
Often, an exam question is to define internal energy.
Internal energy of a substance is the sum of the kinetic and potential energies of the particles in a substance.
We can increase a substance's internal energy by heating. When a substance is heated, energy is transferred to the particles. Either when heating:
The temperature of the substance increases as the particles move faster -> the particles increase their kinetic energy
Or the substance changes state when heating -> the particles increase their potential energy.
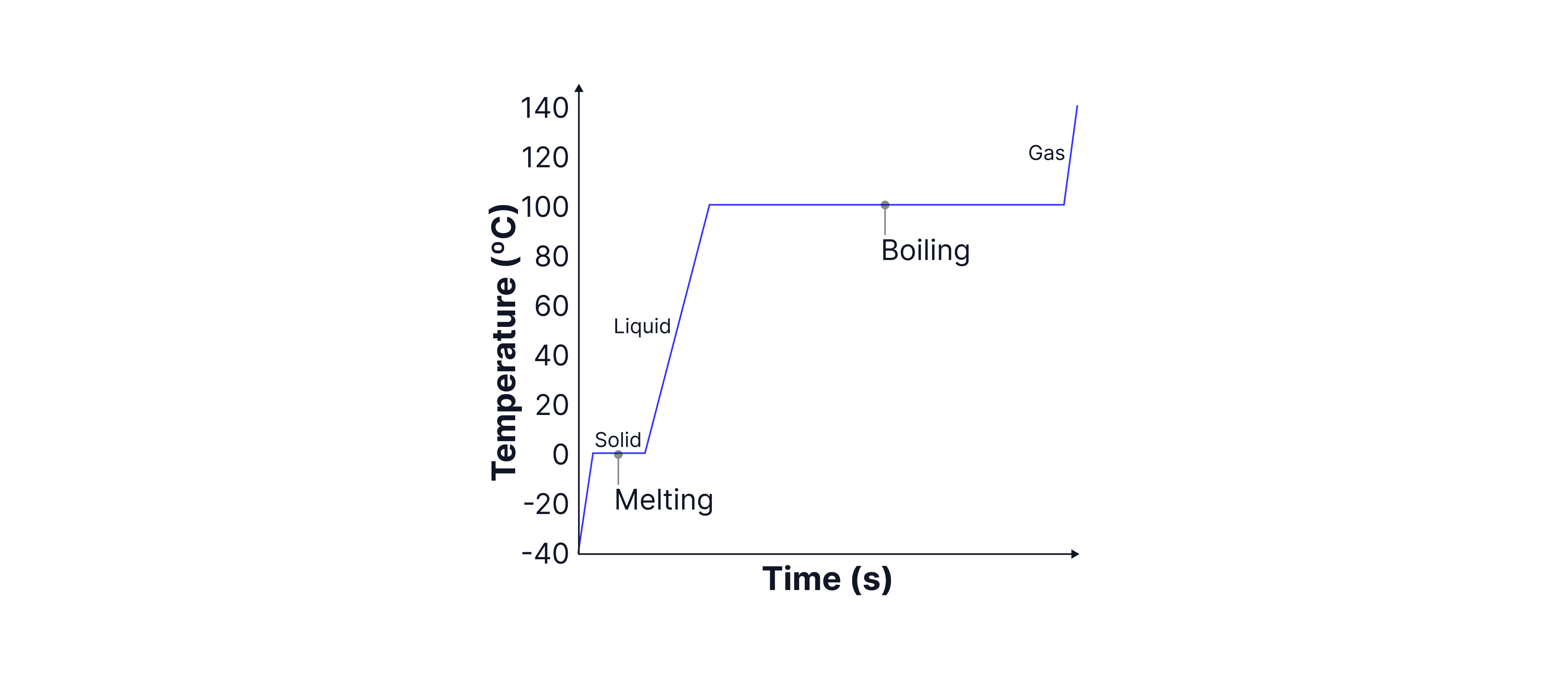
The graph below shows water being heated from a solid to a gas. As the temperature increases, the kinetic energy increases. As a change in state occurs (melting or boiling), shown by a flat line on the graph, the potential energy of the particles increases.
Kinetic Energy
Kinetic energy is the energy particles have because they are moving. Particles move in all states of matter, but they move fastest in gases, so gases have the highest kinetic energy.
The temperature of a substance depends on the average kinetic energy of its particles. As we heat a substance, thermal energy causes the particles to move faster, increasing their kinetic energy and raising the temperature.
Potential Energy
Potential energy is the energy stored due to the spacing between particles.
Particles close together (as in solids) have low potential energy.
Particles far apart (as in gases) have high potential energy.
When a substance is heated and changes state, the potential energy of its particles changes. Thermal energy from heating breaks the forces of attraction between particles, allowing them to move farther apart and increasing their potential energy. Because all the thermal energy is used to break the intermolecular forces between the particles, the temperature remains the same; the particles' kinetic energy does not change. This is why changes of state happen at fixed temperatures.
Worked Example:
How does the internal, kinetic and potential energy of particles change in these situations?
A material is heated and doesn’t change state.
A material changes from liquid to solid at its melting point.
The temperature of water is raised from 20°C to 80°C.
Water in a kettle reaches 100°C. The kettle keeps supplying thermal energy.
The kinetic energy of the particles will increase and the potential energy will stay the same, so the internal energy will increase.
The potential energy of the particles decreases and the kinetic energy stays the same, so the internal energy will decrease.
The kinetic energy of the particles is increasing and the potential energy stays the same, so the internal energy increases.
The water will change state to steam. This will increase the potential energy of the particles and the kinetic energy of the particles will remain the same, so the internal energy will increase.
Worked Example:
A sample of candle wax is heated. The temperature of the wax is recorded every seconds and the results are shown in the graph below:
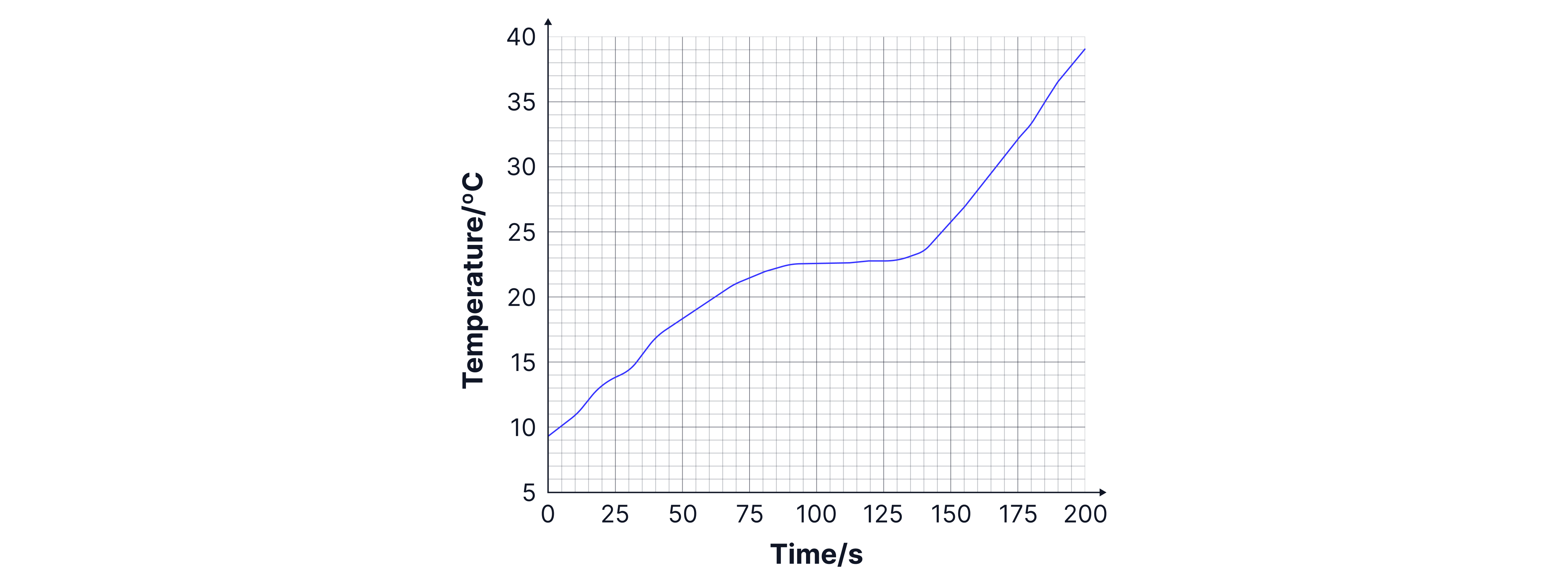
Use the graph to determine the melting point of the candle wax
Explain the shape of the graph in terms of the energy of the particles
Answer:
The candle wax will be changing state at the point on the graph when the temperature is constant. This is at .
When the temperature of the candle wax is increasing, the kinetic energy of the particles is increasing. When the temperature is constant, the candle wax is changing state so the potential energy of the particles in increasing.
Changes in State revisited
Heating a solid-> gas
As the substance is heated, thermal energy is transferred to it. First, the temperature of the solid increases as its particles' kinetic energy increases. At the melting point, the particles gain enough energy to overcome the forces of attraction (intermolecular forces) holding them together, and the solid changes state to a liquid. The particles gain potential energy. The particles change from a regular to an irregular arrangement. They change from vibrating in a fixed position to moving randomly over one another. As the liquid continues to be heated, the particles gain even more kinetic energy and move faster, causing the temperature of the liquid to rise. At the boiling point, the remaining intermolecular forces are overcome, and the liquid becomes a gas. The particles gain potential energy, change from touching to not touching, and move randomly in all directions, filling the space of their container.
Cooling a gas-> solid
As a gas is cooled, it loses thermal energy. Its particles lose kinetic energy, move more slowly, and come closer together as the temperature drops. At the condensing point, they form forces of attraction between the particles (intermolecular forces), lose potential energy and change state to a liquid. The particles change from moving randomly to moving over each other. As the liquid cools further, the particles lose kinetic energy, and their motion slows. At the freezing point, more forces of attraction form between the particles, and the liquid becomes a solid. The particles change from an irregular to a regular arrangement. The particles lose potential energy.
Remember: During a change of state, the temperature does not change. The temperature at which a solid melts to form a liquid is the same temperature at which a liquid freezes to form a solid. For example, water freezes to form ice at, and ice melts to form water at . The same is true for condensation and boiling.
Practice Questions
A student heats a sample of a solid from to .
Describe what is meant by the internal energy of a substance.
Explain why the internal energy of the substance increases when it is heated.
State the effect that heating has on the temperature of the solid in this situation.
-> Check out Hannah's video explanation for more help.
Answer:
Internal energy is the sum of the total kinetic and potential energy of the particles in a substance.
Heating increases the kinetic energy of the particles in the substance as the temperature increases, making them move faster.
The temperature increases as shown in the question.
Triple Only:
A student rapidly compresses a fixed mass of gas using a piston. No heat is transferred to the surroundings during compression.
Explain why the internal energy of the gas increases during compression.
State and explain what happens to the temperature of the gas during rapid compression.
-> Check out Hannah's video explanation for more help.
Answer:
Work is done on the gas by the piston. This transfers energy to the particles, increasing their kinetic energy which is part of their internal energy
The temperature increases. This is because the increased kinetic energy of particles raises their average speed.