Half-Life
Brook Edgar & Hannah Shuter
Teachers


Explainer Video
Half-Life
Definition
The half-life of a radioactive isotope is the time it takes for the number of nuclei in a sample to be reduced by half.
Half-life is defined as the time it takes for the count rate/activity/number of nuclei in a sample to fall to half of its initial value.
For example, suppose you begin with radioactive nuclei.
After one half-life, half of these nuclei will have decayed, leaving remaining.
After two half-lives, half of the remaining nuclei decay, leaving .
After three half-lives, the number falls again to .
The half-life (the time it takes for the number of nuclei to be reduced by half) is always constant. If it takes a certain amount of time for the number of nuclei to drop from to , let's say thirty seconds, it will take exactly the same amount of time for the number to drop from to , thirty seconds, and so on.
Some radioactive isotopes have half-lives of a few seconds, some have half-lives of millions of years, so make sure you read the units used in the question carefully.
Calculating Half-Life
You may be expected to find the half-life of a sample or the count rate after a certain number of half-lives from written information given to you.
Example 1: A sample of a radioactive isotope has an initial count rate of counts per minute and the count rate falls to counts per minute after hours. We can work out how many half-lives have passed by taking the initial count rate, and dividing it by until we find the final count rate:
half-life
half-lives
Therefore, to go from counts per minute to counts per minute was two half-lives.
If half-lives have passed in hours, then we can find how long one half-life is:
Example 2: A sample has an initial activity of . After 10 half-lives, what is the new activity?
We could divide by two until ten half lives have past,
half-life
half-lives
half-lives
half-lives
But you can see that this would take a long time. An alternative quick way to do it is to use the equation, , where represents the number of half-lives.
Finding Half-Life from a Graph
You may also be asked to find the half-life of an isotope from a graph like the one below. The definition of half life is the time taken for the count rate to decrease by half, so we take our initial count rate (t =) of and divide it by two - this would give us a count rate of .
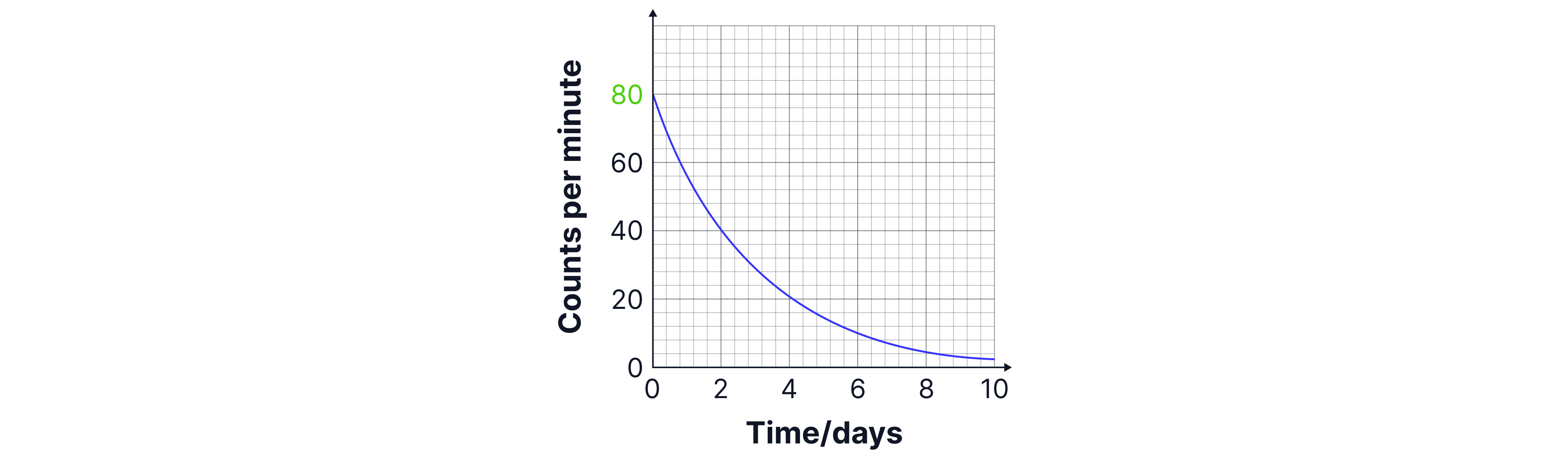
We then draw construction lines across the graph on the y-axis at to find the time. In the graph below this would give us a half-life of days.
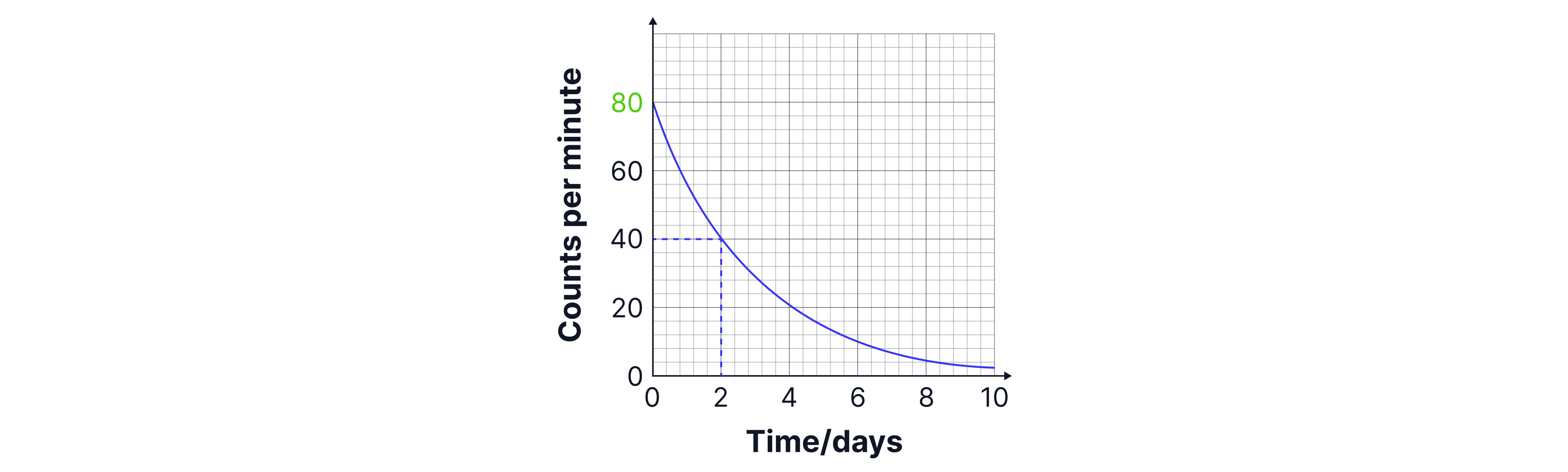
Remember: Check the axis on the graph for the correct units, and don't forget that in an exam, you will be given credit for drawing construction lines on your graph. So take the time to mark these lines onto your graph!
Worked Example:
A radioactive isotope contains undecayed nuclei. After minutes, the number of undecayed nuclei has fallen to .
How many times smaller is the number of nuclei compared to the original?
How many half-lives have passed?
Calculate the half-life of the isotope.
Answer:
We know from the previous question that half-lives have passed in minutes so the time for one half-life,
Worked Example:
Calculate the half-life of the iodine shown in the graph below:
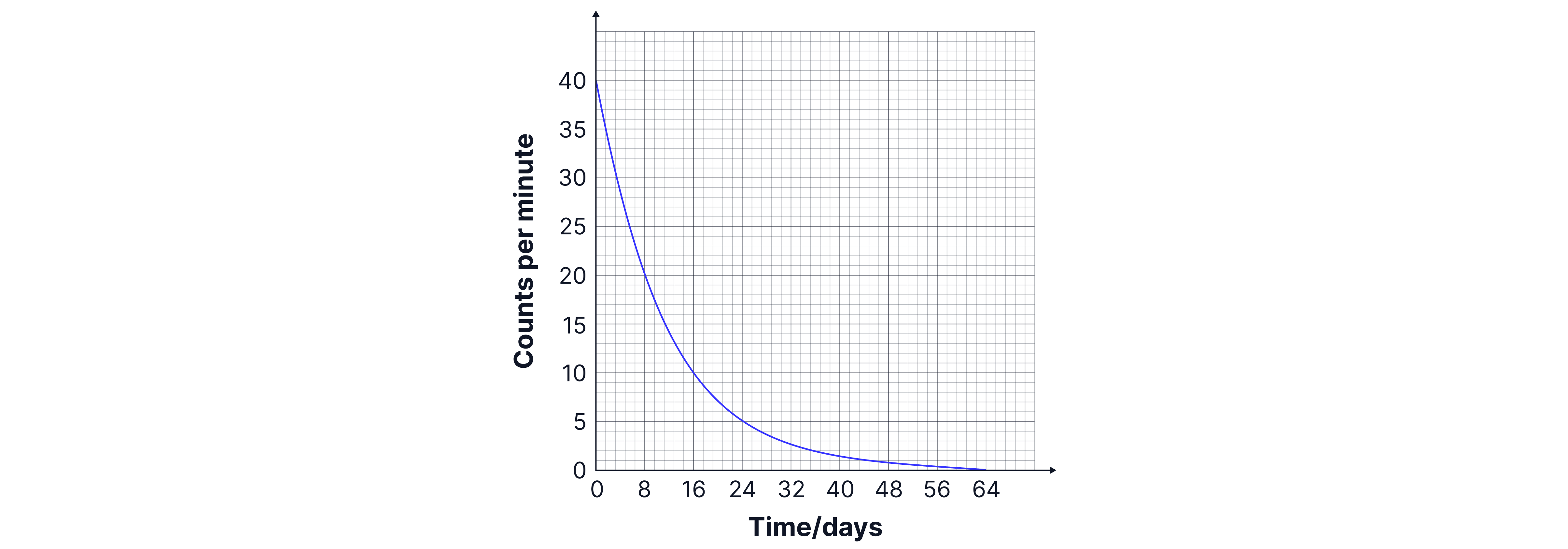
Answer:
The definition of half-life is the time taken for the number of radioactive nuclei to decrease by half. The initial mass of nuclei in this sample is , so after one half-life there will be left. We need to draw construction lines on our graph at (as below) to find that the time taken to reach this point is days, so one half-life is days.
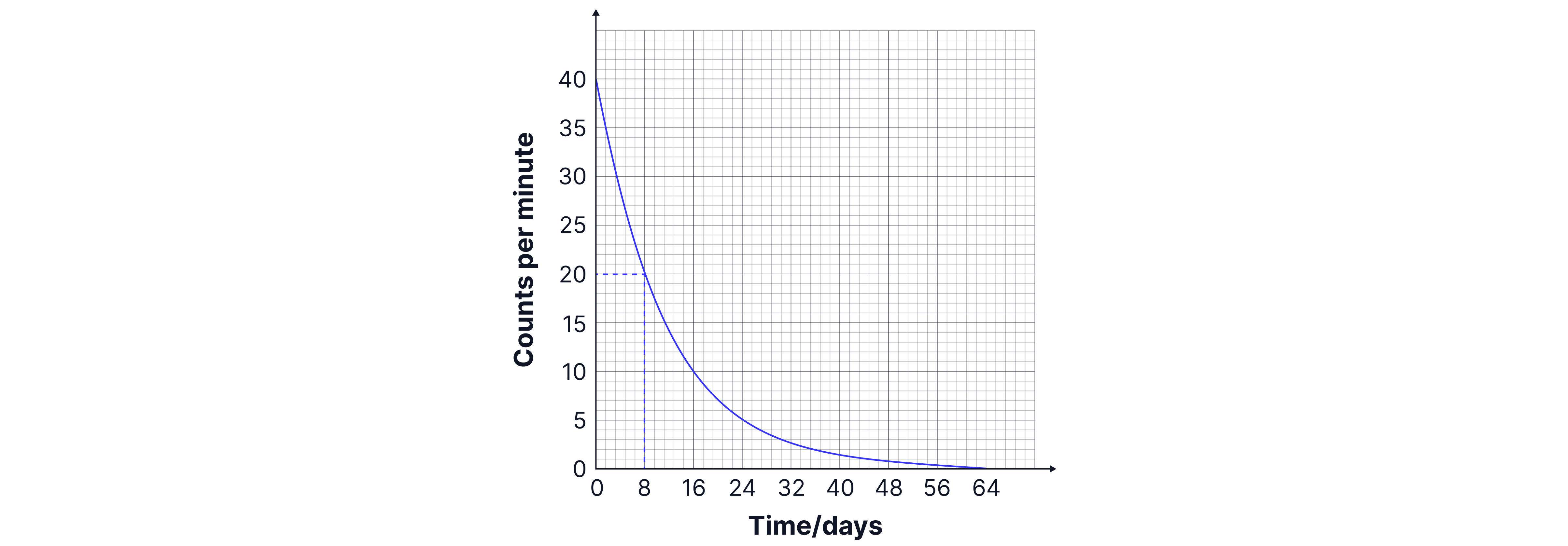
Practice Questions
A radioactive isotope has a count-rate of counts per second. After hours, the count-rate has fallen to counts per second.
State what is meant by the term half-life.
Calculate the number of half-lives that have passed in hours.
Calculate the half-life of the isotope.
-> Check out Hannah's video explanation for more help.
Answer:
The time taken for the count-rate of a radioactive isotope to fall to half its original value.
A sample of a radioactive isotope has an initial activity of .
Explain why it is not possible to predict exactly when any individual nucleus in the sample will decay.
The half-life of the isotope is minutes. Calculate the activity after minutes.
-> Check out Hannah's video explanation for more help.
Answer:
Radioactive decay is a random process.