Contamination and Irradiation
Brook Edgar & Hannah Shuter
Teachers


Explainer Video
Ionisation and Irradiation
When we say radiation is ionising, we mean it has enough energy to knock electrons off atoms, turning them into charged particles called ions.
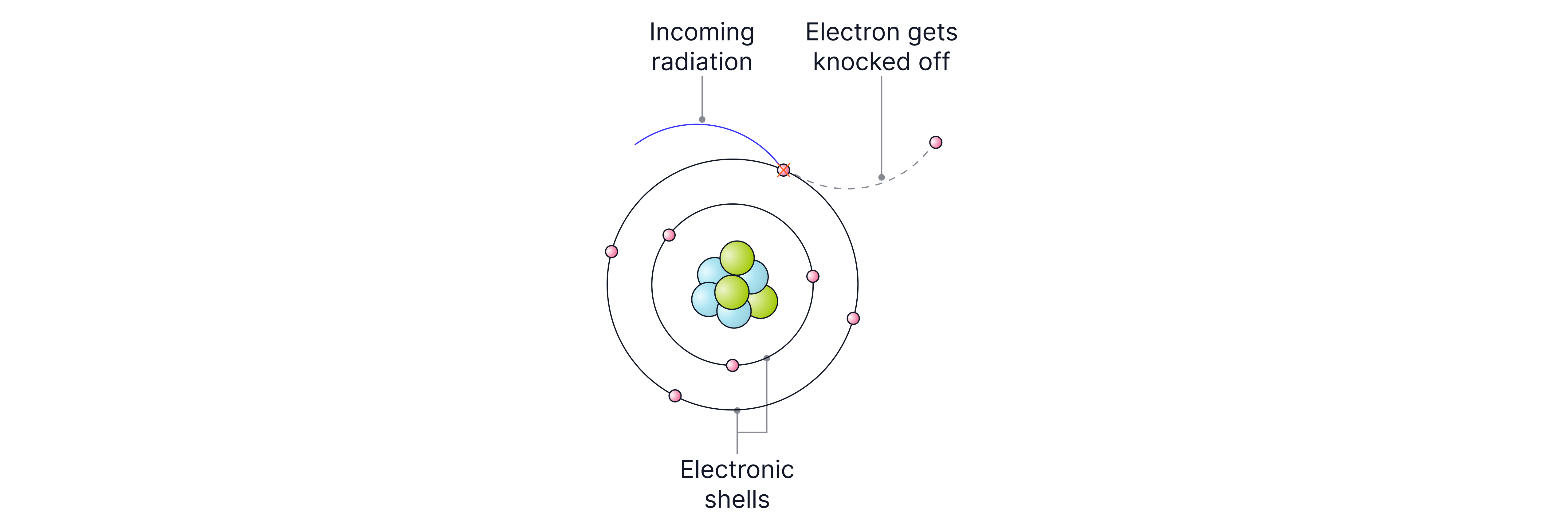
Most of the time, this isn’t a big problem, but if ionising radiation reaches our cells, it can affect important molecules like DNA (inside the nucleus of a cell). If DNA is ionised, its structure can change, which may lead to mutations and sometimes to illnesses such as cancer.
If the radiation is very strong, it can damage cells so badly that they die. This is why ionising radiation can be dangerous, but only in very large doses.
When an object is exposed to radiation, we say it is irradiated. However, irradiating an object will not make it radioactive; it will just ionise its atoms. It changes the charge of the atom, as electrons are lost, but it does not change the nucleus of the atom, so no, you won't glow green if you are exposed to gamma radiation!
Irradiation can be very useful. For example, we use high-energy radiation to sterilise medical equipment and food. The radiation kills any bacteria on the surface of medical equipment, making the items safe to use, and kills any bacteria inside the food without having to open it, making the food safe to eat.
People working with radioactive materials must carefully monitor how much radiation they are exposed to, because too much can be harmful. Exposure can be tracked using dosimeter badges (aka film badges), which record the amount of radiation a person receives.
To reduce exposure, we use safety measures such as:
handling radioactive sources with tongs (to increase the distance from our bodies)
standing behind a lead-lined screen (blocks most radiation)
storing sources in lead-lined containers (blocks most radiation)
operating sources from a different room
These precautions help keep radiation levels as low as possible.
Worked Example:
A factory uses gamma radiation to sterilise packets of herbs before they are sold. A student says, “I don’t want to eat irradiated food because it will be radioactive.”
Explain why the student is incorrect.
Answer:
Irradiating something does not make it radioactive. The food is only being exposed to gamma rays. Once the radiation source is removed, the food does not have any radiation inside it, and it does not emit any radiation of its own. The nucleus of the atoms in the food has not been changed.
Therefore, the irradiated food is not radioactive, and it is safe to eat after sterilisation.
Worked Example:
A technician is working with a gamma radiation source in a laboratory. Describe two safety precautions the technician should take and explain why they help reduce exposure.
Answer:
The technician should stand behind a lead-lined screen, as lead will absorb gamma radiation (and alpha and beta radiation if it were present).
The technician should also wear a dosimeter/film badge to measure radiation exposure over time, ensuring their dose stays within safe limits.
Teacher Tip: For this question, the answer to use tongs to increase the distance between the gamma source and the technician would not be accepted, as gamma radiation has an unlimited range in air.
Contamination
When radioactive atoms get onto or into an object, this is called contamination. The danger from contamination comes from the fact that the radioactive atoms stay with the object and continue to decay, releasing radiation.
The level of hazard depends on the type of radiation emitted and on where it is emitted.
Alpha contamination outside the body is usually a small risk, because alpha particles cannot penetrate the skin (they cannot even get through paper). But gamma contamination outside the body is more dangerous as it is highly penetrating.
Alpha contamination inside the body is extremely dangerous. Alpha particles are highly ionising, so if alpha-emitting atoms are swallowed or inhaled, they can cause severe damage to cells and organs. Gamma radiation inside the body is less harmful as it is the least ionising.
Contamination is generally more hazardous than simple irradiation because a contaminated object is radioactive itself, and the radioactive atoms can be difficult to remove.
To prevent contamination, workers use protective measures such as:
wearing gloves
wearing lab coats
wearing breathing apparatus if the radioactive source is airborne
handling sources with tongs to avoid direct contact
The main aim is to stop the radioactive material from entering or settling on the body in the first place.
The definitions of contamination and irradiation are similar, but the methods of preventing them are quite different. They are summarised in the table below:
Feature | Irradiation | Contamination |
Definition | Being exposed to radiation from an external source. | Having radioactive atoms inside or on an object. |
Does the object become radioactive? | No. Irradiation does not make the object radioactive. | Yes. The object becomes radioactive because the radioactive atoms are now part of it. |
Source of danger | Radiation from outside the body. | Radioactive atoms inside or on the body, continuously decaying. |
Duration of hazard | Hazard ends when the radiation source is removed. | Hazard continues until the contamination is removed or the material decays. |
Risk level | Usually lower, unless exposed to high doses. | Often higher, especially if radioactive material gets inside the body. |
Effect of alpha radiation | Low risk externally (cannot penetrate skin). | Very high risk internally (highly ionising). |
How to reduce risk | Increase distance, use shielding (lead), minimise exposure time. | Prevent radioactive material from making contact: use gloves, tongs, protective clothing, masks. |
Typical examples | Medical X-rays, sterilisation of equipment. | Swallowing/inhaling radioactive dust, radioactive spills. |
Worked Example:
A worker accidentally inhales a small amount of an alpha-emitting radioactive substance.
Explain why this is much more dangerous than being irradiated by an external alpha source.
Answer:
Alpha radiation cannot travel far; it has a range of around five centimetres in air and cannot penetrate the skin, so an external alpha source poses little danger.
However, once alpha-emitting atoms are inside the body, the atoms will continue to decay inside it. Alpha particles are highly ionising, so they can cause severe damage to nearby cells and DNA. This can lead to mutations, organ damage, or cancer.
Worked Example:
A scientist is working with a powdered alpha-emitting radioactive material in a laboratory. Explain two safety precautions the scientist should take to prevent contamination, and why each precaution is important.
Answer:
The scientist should wear protective clothing such as gloves and a lab coat to stop radioactive particles from getting onto the skin.
The scientist should also use a mask or breathing apparatus. Since the radioactive material is a powder, small particles could be inhaled. Wearing a mask prevents the particles from entering the lungs.
Practice Questions
A worker handles a radioactive source in a laboratory.
Explain the difference between irradiation and contamination.
State why contamination is usually more hazardous than irradiation.
Name one precaution that should be taken when using a radioactive source to irradiate objects.
-> Check out Hannah's video explanation for more help.
Answer:
Irradiation is exposure to radiation. Contamination occurs when radioactive atoms are inside or on an object, making it radioactive.
Contaminated materials continue to emit radiation, which can be harmful.
Any from: use tongs to increase distance / wear appropriate shielding / minimise exposure time / store the source in a lead container
A food-processing plant irradiates fruit to prevent it from spoiling.
Explain why irradiated fruit does not become radioactive.
A worker accidentally gets radioactive dust on their hands. Identify whether this is contamination or irradiation and explain your answer.
-> Check out Hannah's video explanation for more help.
Answer:
The fruit is only exposed to radiation; no radioactive atoms are transferred to it, the nucleus of the atoms in the fruit is not changed.
This is contamination because radioactive material has been transferred onto the worker’s skin.