Radiation Intro
Brook Edgar & Hannah Shuter
Teachers


Contents
Explainer Video
Atomic Structure
Imagine taking a sheet of paper and tearing it in half, then tearing one of those halves again, and again, and again. If you could continue this process indefinitely, eventually you’d reach a point where the piece could no longer be divided. That smallest piece is what we call an atom.
Atoms are the fundamental building blocks of everything around us, from the cells in our bodies to the stars scattered across the universe. An average atom is incredibly small (around across). There are roughly 100,000 atoms across the width of a human hair! Each atom consists of three particles: protons, neutrons, and electrons. These are known as subatomic particles.
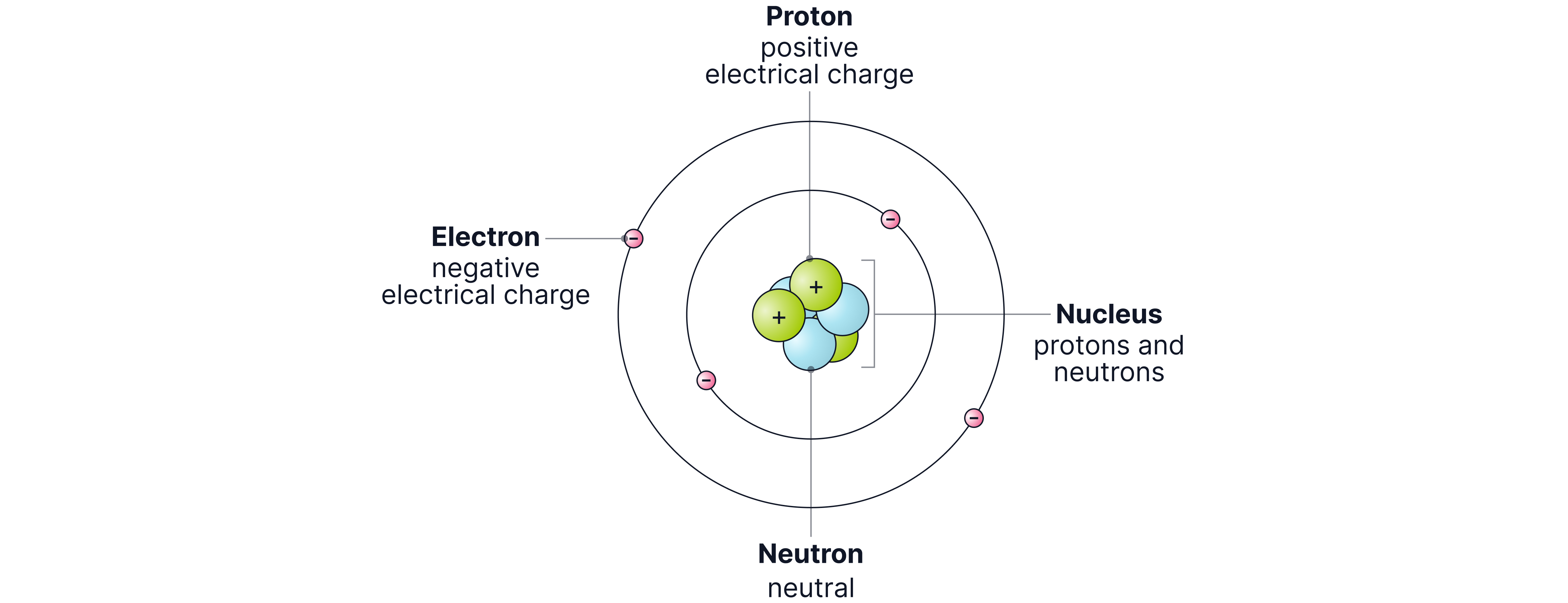
Protons
Protons are located at the centre of the atom, in a dense region called the nucleus.
Mass: (relative atomic mass)*
Charge: (protons are positive)
Neutrons
Neutrons are also located in the nucleus, packed closely alongside the protons.
Mass: (same as the proton)
Charge: (neutrons are neutral)
Electrons
Electrons move around the nucleus; they orbit the nucleus in specific shells or energy levels. From chemistry, we know that there is a maximum of two electrons in the first shell and eight in the next, and so on...
Mass: (much smaller than protons and neutrons), as the mass is so small we can ignore it, it is neglibible, so we can say the mass is zero.
Charge:
*A Mass of One What?
The actual mass of a proton is , which is extremely inconvenient to work with. Instead, we use relative atomic mass. We assigned the proton with a mass of . As the actual mass of a neutron is the same as a proton it also has a mass of . The electron has a mass roughly two thousand times less than a proton, so small that we can say it is zero.
Electrons in Shells
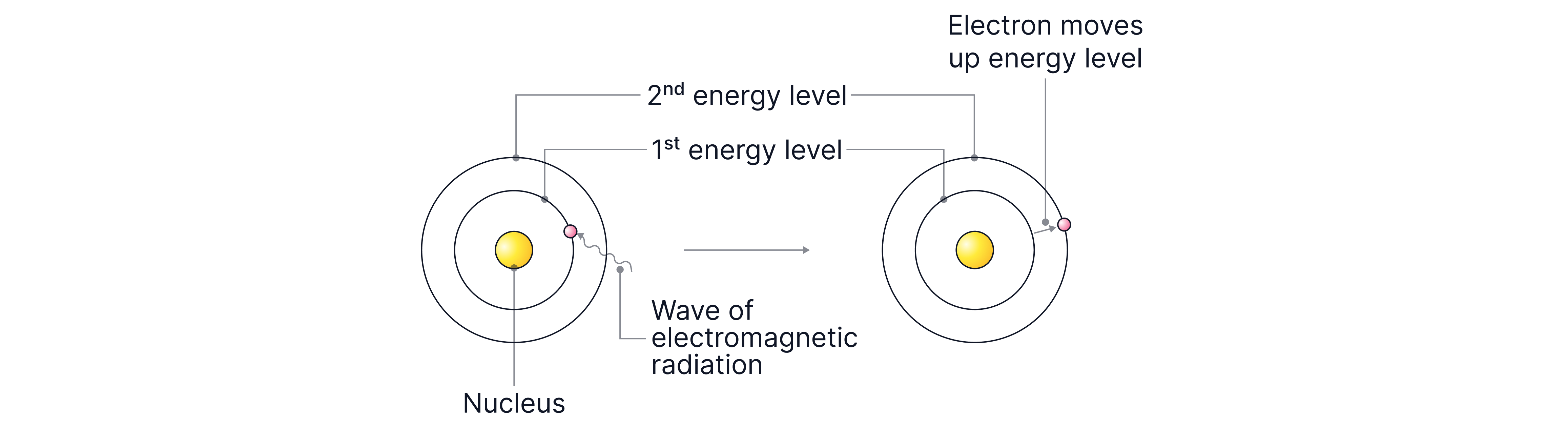
Electrons occupy shells around the nucleus, each at a specific energy, so the shells are also known as energy levels.
Electrons in shells closest to the nucleus have lower energy.
Electrons in shells further away have higher energy.
Electrons can gain energy by absorbing electromagnetic radiation (a wave of energy), causing them to jump to a higher energy level.
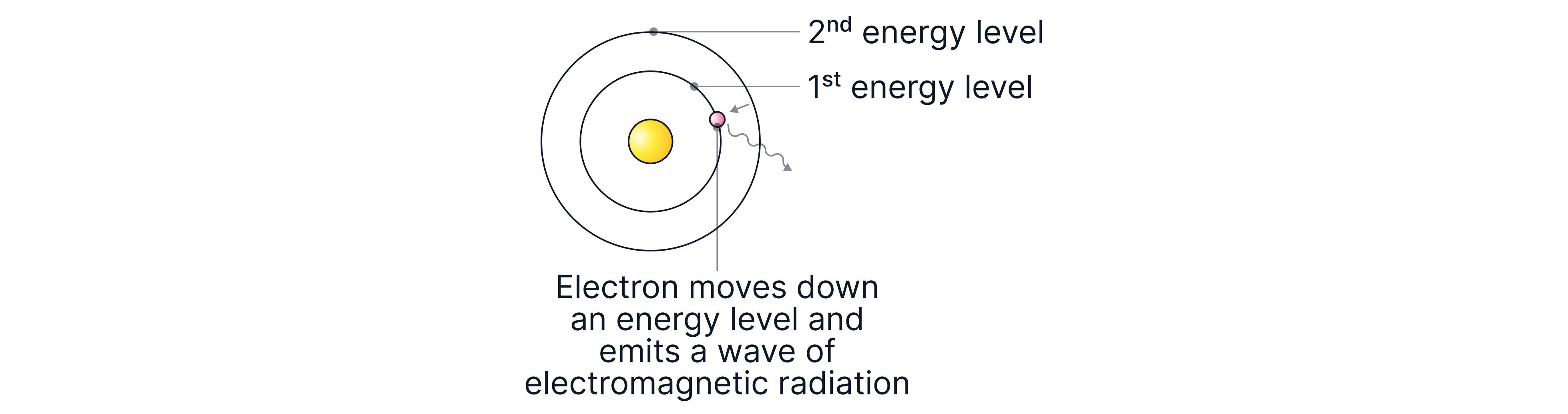
They can lose energy by emitting electromagnetic radiation, causing them to fall back to a lower energy level.
Worked Example:
Describe the position, mass and charge of each of the subatomic particles in the nucleus.
Answer:
The proton is in the nucleus. It has a mass of and a charge of .
The neutron is also in the nucleus. It has a mass of and a charge of .
The electron orbits outside the nucleus. It has a mass of or zero and a charge of .
Teacher Tip: On an exam, you will receive a mark for either of the masses you use for an electron, so I would recommend using zero, as it is an easier number to remember.
Worked Example:
Most of the mass of the atom is in the nucleus.
Explain why, in terms of the particles found in the atom.
Answer:
The protons and neutrons are found in the nucleus. Protons and neutrons each have a relative mass of , so they are massive compared to electrons. Electrons have a very small mass (about of a proton), so they contribute very little to the total mass. Therefore, almost all of the mass of the atom is concentrated in the nucleus.
Mass Number, Atomic Number and Isotopes
We can work out how many protons, neutrons, and electrons are in a neutral atom by looking at it on the periodic table.
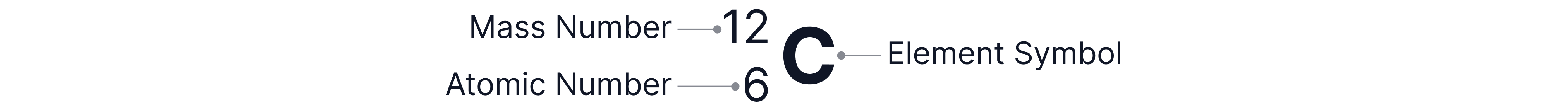
This example shows how a carbon atom appears on the periodic table.
Mass Number
The mass number is the top number. It tells us the total number of protons and neutrons in the nucleus (as both subatomic particles have mass).
For example, carbon has a mass number of , meaning it has protons and neutrons in total.
Atomic Number
The atomic number is the bottom number. It tells us the number of protons in the nucleus.
Carbon has an atomic number of , so it has protons
-> To calculate the number of neutrons:

For example, with carbon:
In a neutral atom, the number of electrons equals the number of protons (so that the charges balance). Therefore, carbon also has negative electrons to balance the positive protons.
Isotopes
An isotope is an atom of the same element that has the same number of protons, but a different number of neutrons. Here is an example using carbon, an isotope of carbon, written as .
Carbon still has an atomic number of , so it would still have protons and neutrons. But the mass number is , so to calculate the number of neutrons:
Carbon has neutrons.

Worked Example:
Complete the table below:
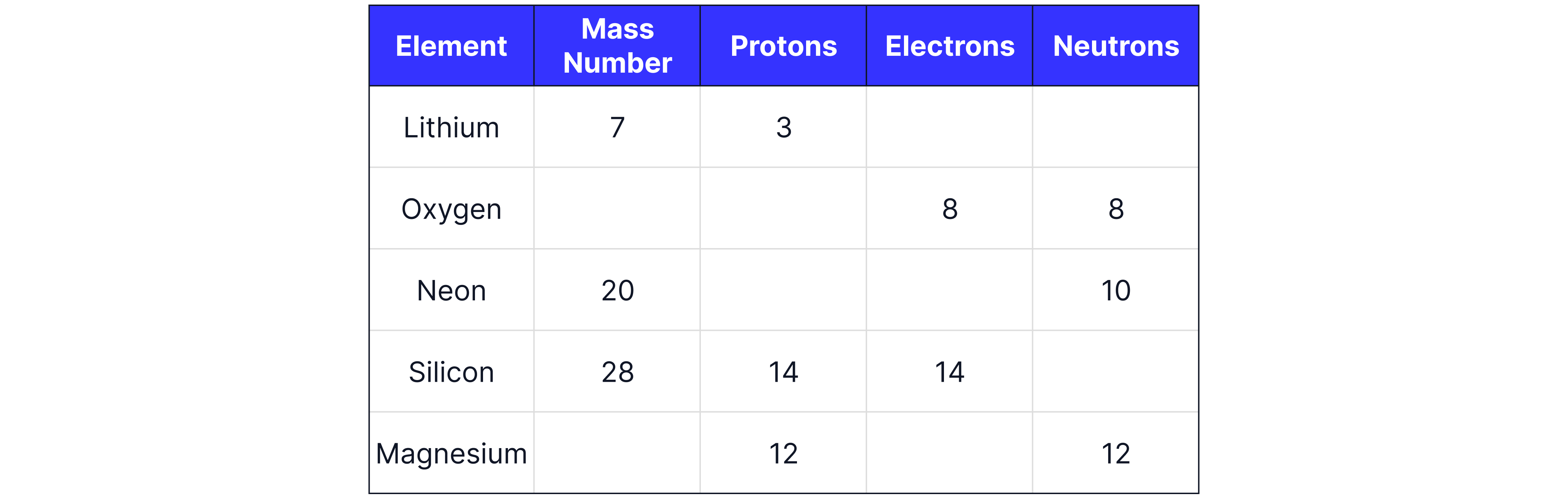
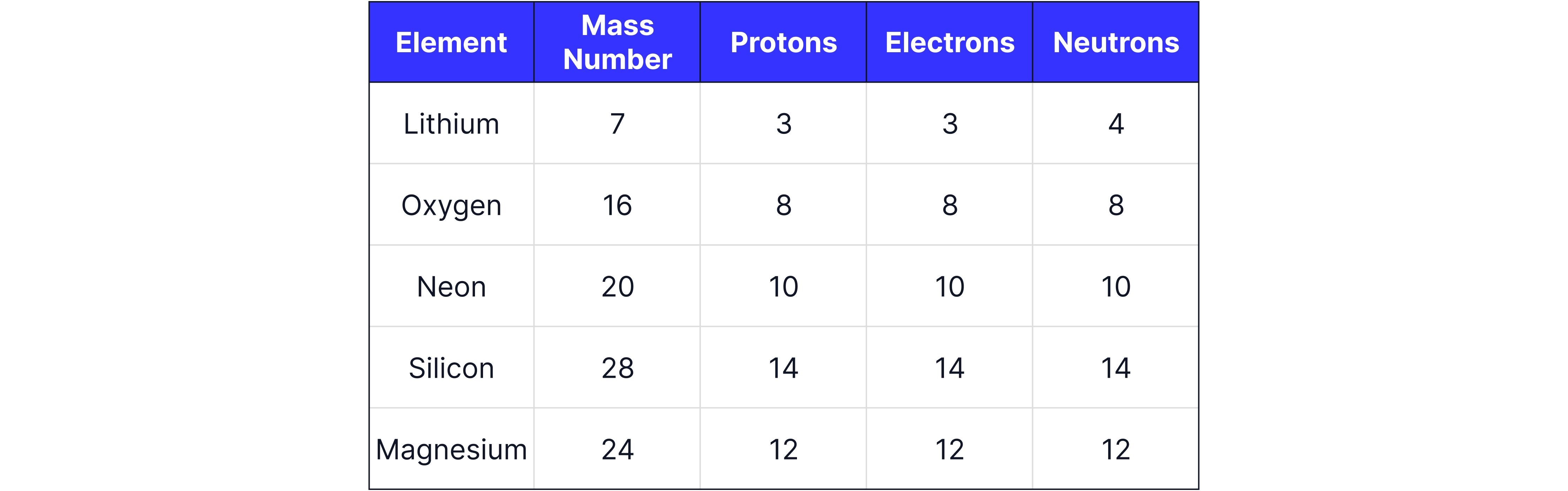
Answer:
Remember the number of protons is always the same as the number of electrons in a neutral atom, and that the mass number is the number of protons and neutrons added together.
Worked Example:
Pair isotopes of the same element from these six nuclei:
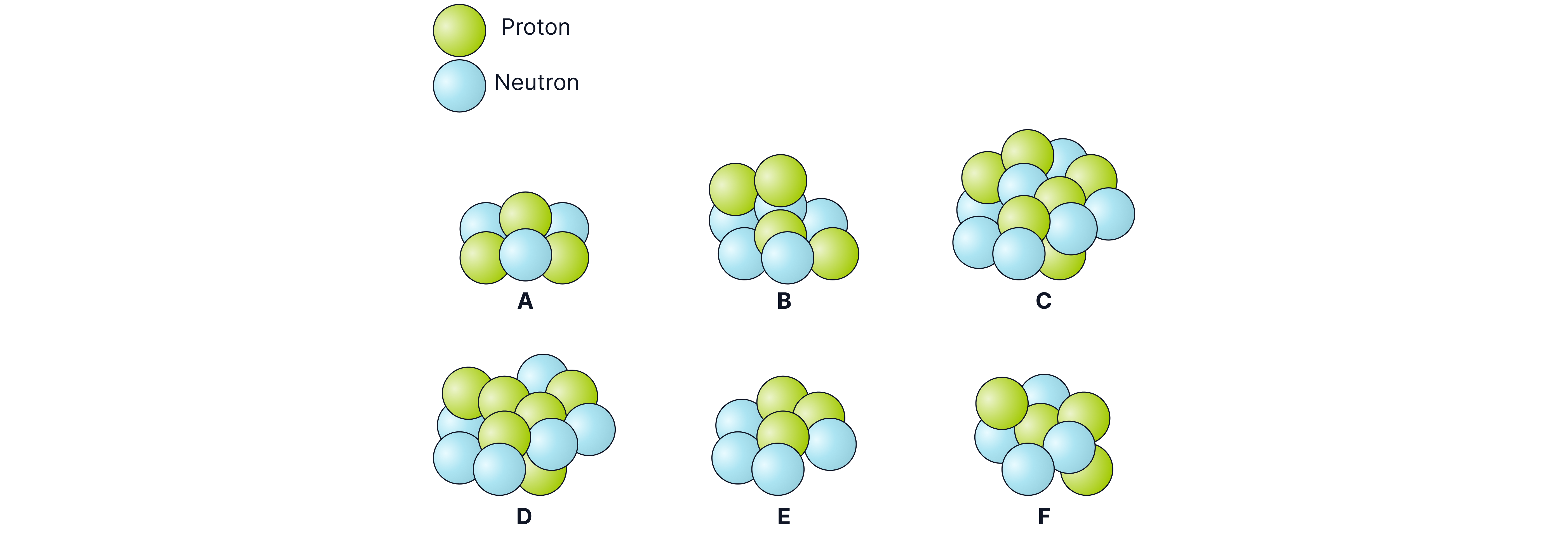
Answer:
Isotopes have the same number of protons, but different numbers of neutrons. The nuclei that have the same number of protons and are therefore isotopes of each other are:
A and E -> both have three protons
B and F -> both have four protons
C and D -> both have six protons
Practice Questions
The diagram shows a simplified model of an atom.
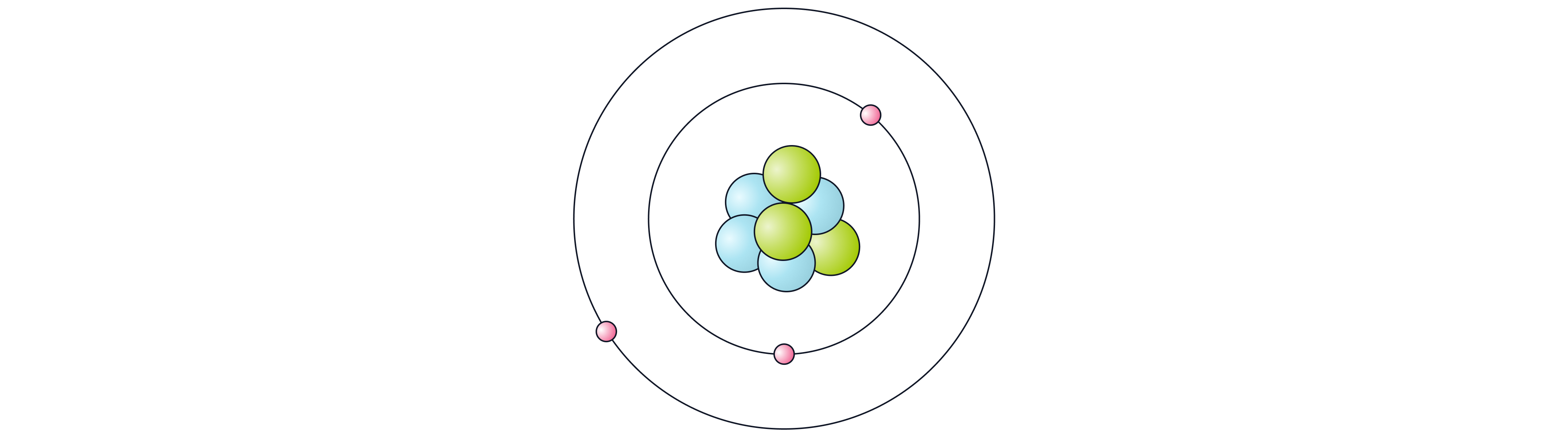
State what the nucleus is made of.
Explain why the atom has no overall charge.
An electron moves to a higher energy level. Explain what must happen for this to occur.
-> Check out Hannah's video explanation for more help.
Answers:
Protons and neutrons
Number of negative electrons equals number of positive protons, so charges cancel
The electron must absorb electromagnetic radiation/energy
The element X has the nuclear symbol .
State the number of protons in this atom.
State the number of neutrons in this atom.
Two isotopes of element X both have 15 protons. Explain what is different about the isotopes.
-> Check out Hannah's video explanation for more help.
Answers:
They have different numbers of neutrons.