Discovery of the Nucleus
Brook Edgar & Hannah Shuter
Teachers


Explainer Video
Development of the Model of the Atom
Our modern understanding of the atom did not appear all at once - it developed gradually as scientists carried out new experiments and gathered better evidence.
Early Ideas
The ancient Greek philosopher Democritus proposed that all matter was composed of tiny, indivisible (cannot be broken down) particles called atoms. At this point, atoms were imagined simply as solid spheres.
This idea remained mostly unchanged for centuries.
Thomson and the Plum Pudding Model
In , J.J. Thomson discovered the electron, proving that atoms can be broken down. His discovery led to the plum pudding model, which described the atom as:
A solid sphere with its mass and positive charge evenly distributed (spread throughout)
Negative electrons scattered within it, like raisins or, in his case, plums in a pudding
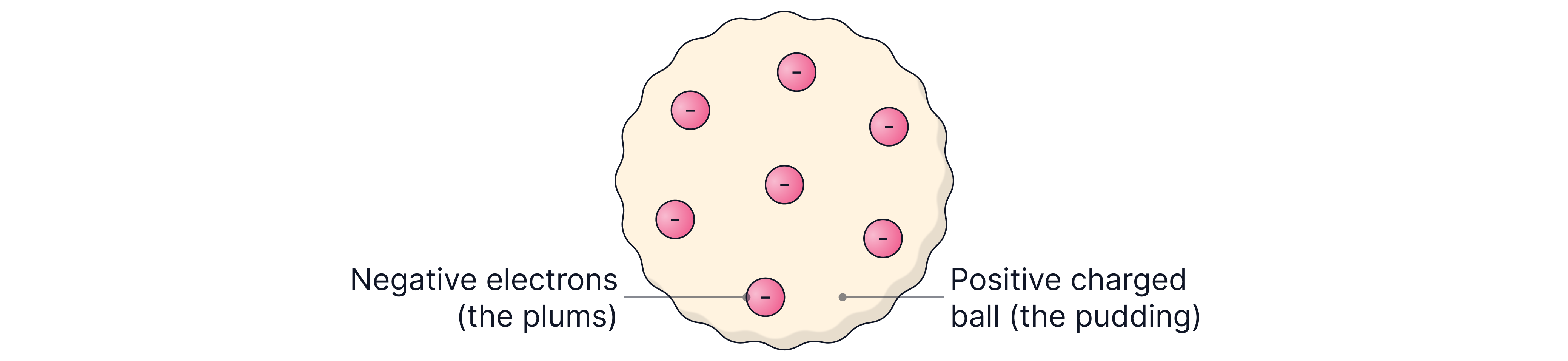
This model explained why atoms have no overall charge, as the sphere of positive charge equals the charge of the electrons.
Rutherford's Alpha Scattering Experiment
Rutherford and his team fired alpha particles (positively charged helium nuclei) at a very thin sheet of gold foil to test the plum pudding model.
Thomson actually predicted that the positive sphere was more like a dense liquid than a pure solid, so they thought that if the plum pudding model were correct, as the positive charge is spread out through the atom, the alpha particles would pass through, experiencing very small forces of repulsion, causing slight deflections to their paths of around . What actually happened was that:
A small number of alpha particles were deflected at large angles , with some even coming backwards. As these alpha particles experienced strong repulsive forces, this suggested that there is a small, dense positive charge at the centre of the atom, as only a few were deflected at these large angles. This was named the nucleus.
Most alpha particles passed straight through, suggesting most of the atom was empty space.
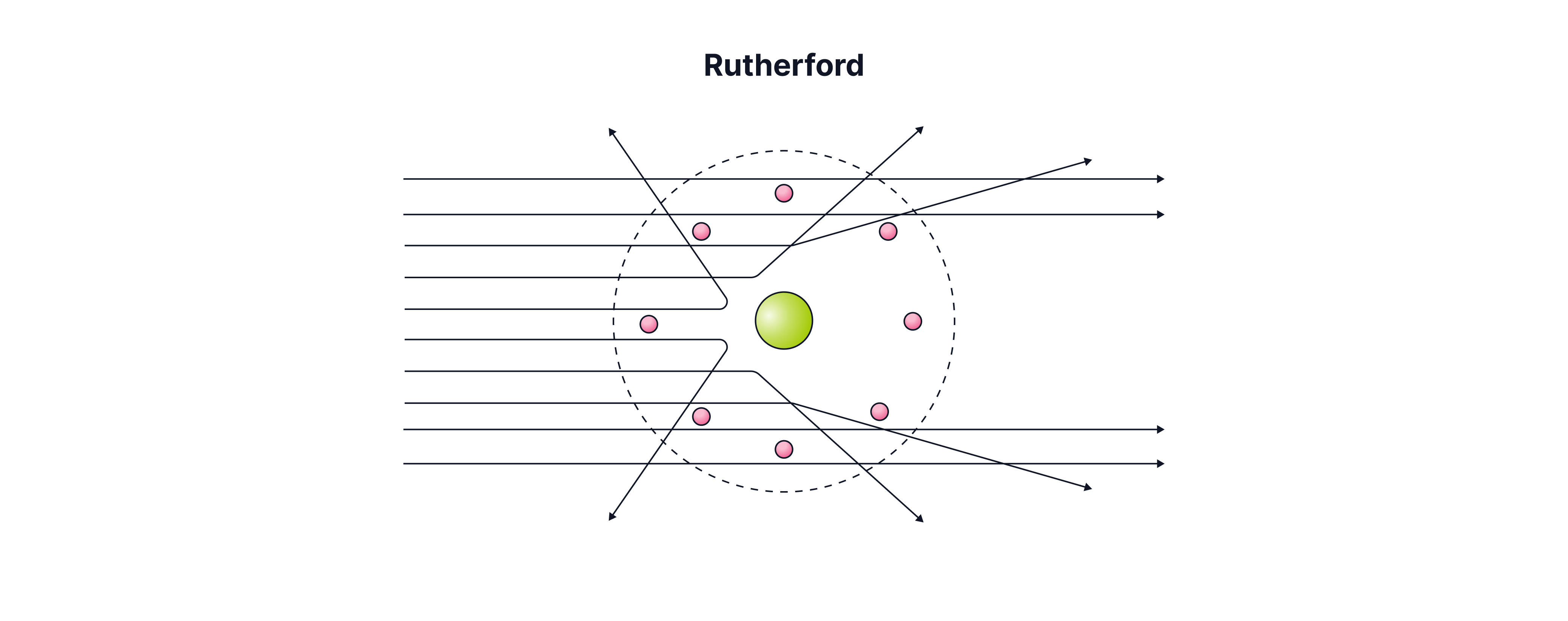
This became the nuclear model of the atom and forced scientists to abandon the plum pudding model entirely.
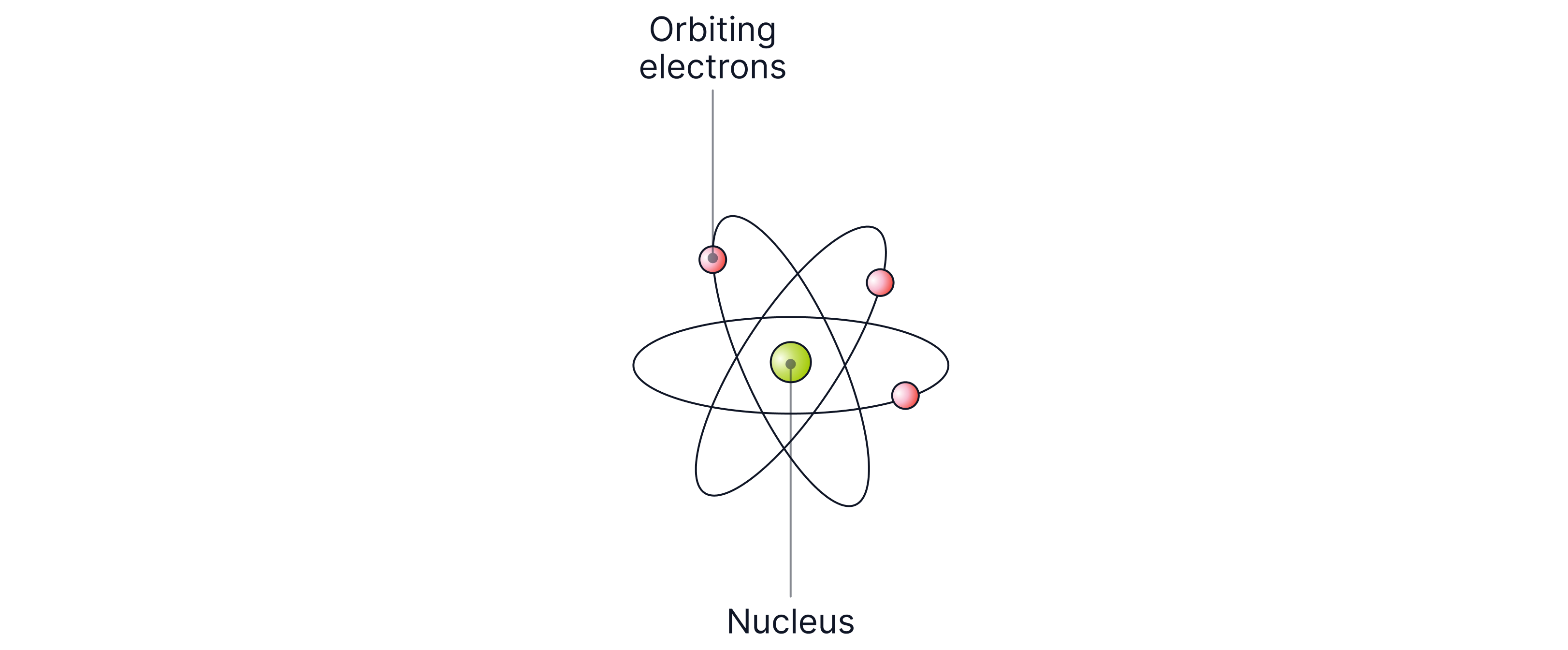
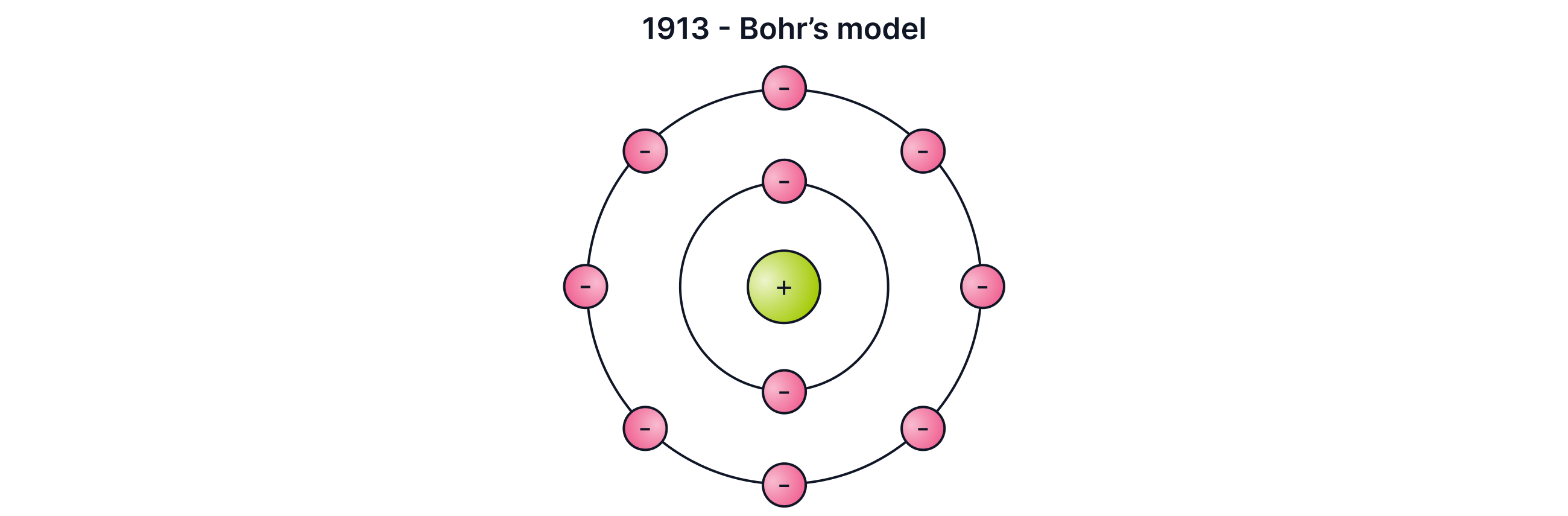
Bohr and Electron Energy Levels
In , Niels Bohr refined Rutherford’s model. He proposed that:
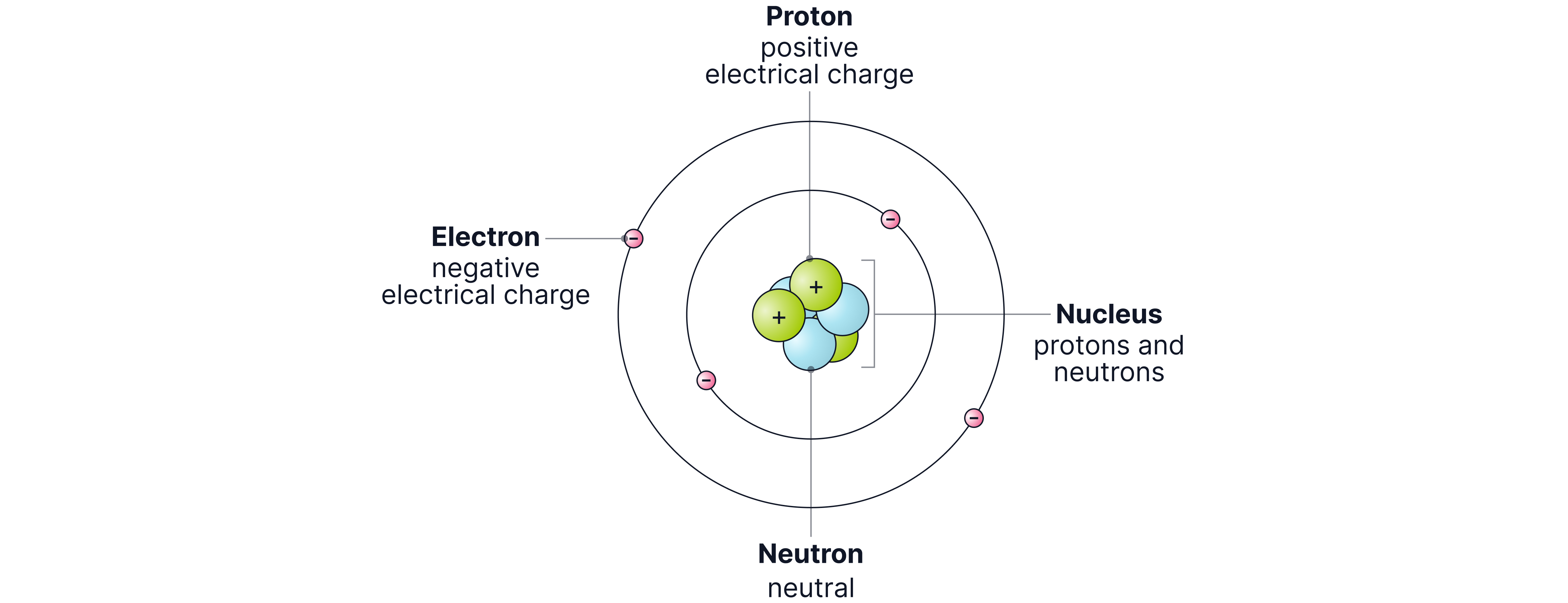
Electrons orbit the nucleus at fixed distances (energy levels or shells)
Electrons can move to higher or lower orbits by absorbing or emitting energy (electromagnetic waves)
This explained why electrons do not spiral into the nucleus.
Chadwick and the Neutron
Rutherford’s model could not explain why nuclei containing more than one proton did not break apart due to repulsion. In , James Chadwick discovered the neutron, a neutral particle inside the nucleus.
This discovery explained:
The extra mass in the nucleus
Why the nucelus didnt break up from the positive protons repelling each other (the neutrons hold them together)
Why isotopes exist (same number of protons, different number of neutrons)
With this final discovery, the modern atomic model was established.
Worked Example:
Match the evidence to the conclusion for Rutherford's scattering experiment:

Answer:

Worked Example:
Scientists used the results of the alpha scattering experiment to replace the plum pudding model with the nuclear model of the atom.
Explain two differences between the plum pudding model and the nuclear model.
In your answer, describe what each model suggests about the structure of the atom.
Answer:
In the plum pudding model, the atom is described as a sphere of positive charge with negative electrons embedded throughout it, like raisins in a pudding. There is no nucleus.
In the nuclear model, all of the mass and positive charge are concentrated in a small, central nucleus, with electrons orbiting this nucleus in shells. Most of the atom is empty space, unlike the solid sphere of positive “pudding” proposed by Thomson.
Practice Questions
In Rutherford’s alpha scattering experiment, a narrow beam of alpha particles was directed at a thin sheet of gold foil.
State what most of the alpha particles did when they hit the gold foil.
A very tiny fraction of the alpha particles bounced back toward the source. Explain what this observation suggested about the structure of the atom.
-> Check out Hannah's video explanation for more help.
Answer:
Most alpha particles passed straight through the foil.
It showed that there is a small, dense nucleus containing most of the atom’s mass. The nucleus must be positively charged because it repelled the positively charged alpha particles.
Before Rutherford’s experiment, the plum-pudding model described the atom as a sphere of positive charge with electrons spread throughout it.
Explain why the plum-pudding model could not explain the results of the alpha scattering experiment.
Describe two conclusions about atomic structure that were supported by Rutherford’s findings.
-> Check out Hannah's video explanation for more help.
Answer:
In the plum-pudding model, positive charge is spread evenly, so alpha particles would be expected to pass through with only very slight or no deflection. It cannot explain large deflections or particles bouncing back.
The atom is mostly empty space. The positive charge and most of the mass are concentrated in a very small central nucleus.