Types of Radiation
Brook Edgar & Hannah Shuter
Teachers


Contents
Explainer Video
What is Radiation?
Most of the nuclei we encounter are stable, but some isotopes have unstable nuclei. This happens when the nucleus contains too much energy or the wrong balance of protons and neutrons.
To become stable, an unstable nucleus will release excess energy or particles in the form of radiation - this is known as radioactive decay. For GCSE Physics, you need to know the four main types of nuclear radiation: alpha, beta, gamma, and neutron radiation.
Radiation is detected using a Geiger–Müller (GM) tube, which produces a click each time it detects a particle or wave of radiation (a "count"). The number of clicks detected per second is called the count rate or activity, and it is measured in units of becquerels (Bq), where Bq = decay per second.
Radioactive decay is a random process - we can calculate the probability that a nucleus will decay or how long it will take a certain number of nuclei to decay, but we cannot predict exactly when the decay will happen and which nucleus will decay. Think of popping popcorn: we know there is a high probability that all kernels will pop in five minutes, but we cannot predict exactly which kernel pops next or exactly when the first one will pop -> we can just make a really good guess.
Worked Example:
A Geiger-Muller tube measures counts of radiation in minute from a sample of Uranium. What is the activity of this sample?
Answer:
Activity is the number of decay per second, so first we need to convert minute into seconds and then calculate the number of decays/counts each second.
Worked Example:
A sample of americium has an activity of . If a Geiger-Muller tube was left next to the sample for minutes, what would the total amount of radiation received be?
Answer:
The activity tells us the counts of radiation every second -> there are 45 counts every second. So to find the total amount of counts/radiation in four minutes, we need to multiply the activity by the total number of seconds:
First, we need to convert minutes into seconds:
Alpha, Beta, Gamma and Neutron Radiation
Alpha
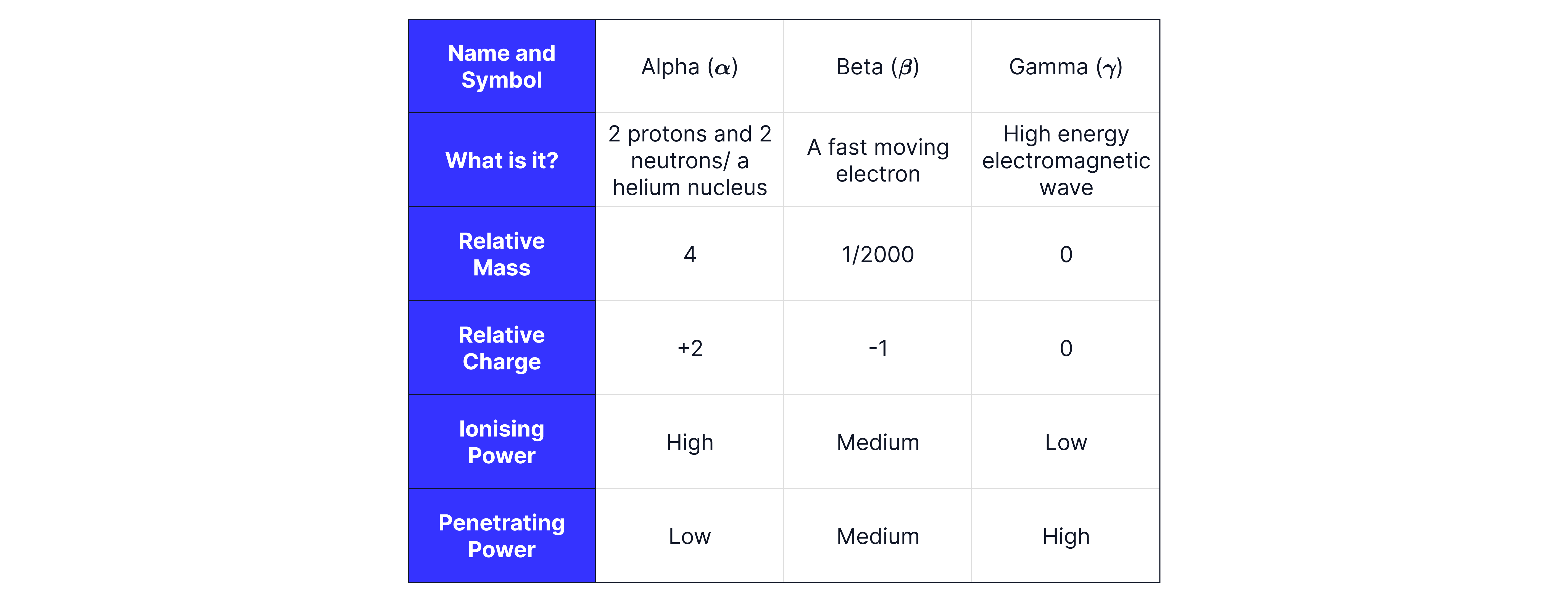
Alpha radiation is emitted from the nucleus if there are too many protons and neutrons. It consists of two protons and two neutrons, which is why it is described as a helium nucleus (not a helium atom, because it has no electrons). Because alpha particles contain two protons, it has an overall charge of , and a mass of (remember each proton has a positive charge and protons and neutrons have a mass of one). Alpha radiation is given the Greek symbol for alpha, .
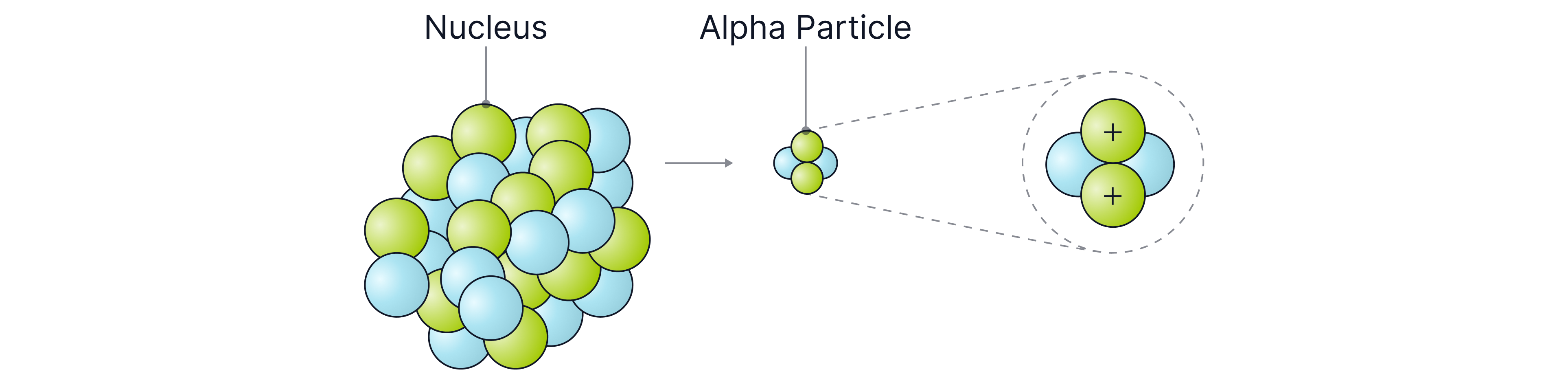
Alpha radiation interacts strongly with nearby atoms, removing electrons from them and causing them to become ions. This process is known as ionisation, and alpha particles are the most ionising type of nuclear radiation because they are the largest and most highly charged.
However, because they ionise so easily, alpha particles lose their energy quickly and cannot travel far. They can be stopped by a sheet of paper and typically have a range of around in air. This means alpha radiation is the least penetrating type of radiation.
Beta
Beta radiation consists of a fast-moving electron emitted from the nucleus. This occurs when a nucleus has too many neutrons. In this process, a neutron changes into a proton and an electron; the proton remains in the nucleus, while the electron is emitted at high speeds as a beta particle. Because it is an electron, a beta particle has a charge of and a negligible mass (approximately ). The symbol for beta radiation is .
Below, we can see that as the Cs nucleus decays into the Ba nucleus by emitting beta radiation, the atomic number (the bottom number) increases by one. This is because it gained a proton as a neutron inside the Cs nucleus changed into a proton and emitted an electron in the process.
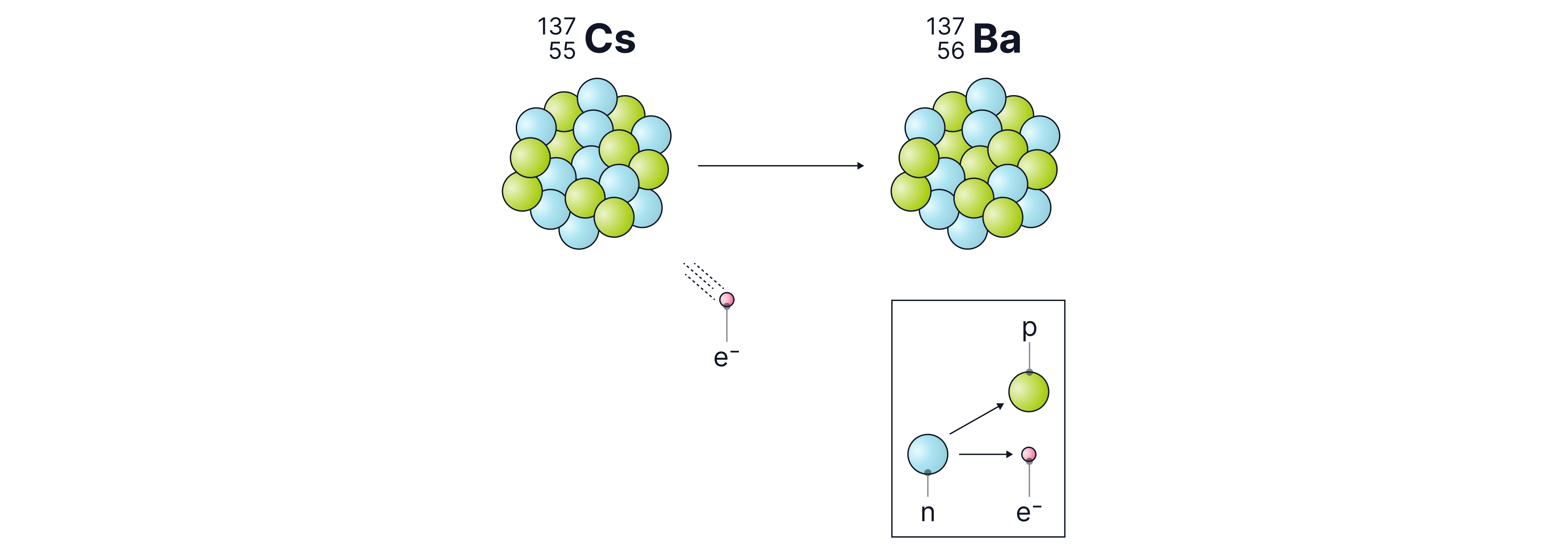
A common misconception is that this electron comes from the electron shells outside the nucleus, but this is incorrect. Beta particles are produced inside the nucleus during radioactive decay.
Beta radiation interacts less strongly with atoms than alpha particles because it has a smaller mass and a smaller charge. As a result, beta radiation is less ionising than alpha, but more ionising than gamma.
Beta particles can travel up to around in air and are stopped by a few millimetres of aluminium. This means beta radiation is more penetrating than alpha radiation, but less penetrating than gamma.
Gamma
Gamma radiation is a high-energy electromagnetic wave released from the nucleus of an unstable atom. Because it is a wave rather than a particle, gamma radiation has no mass and no charge. The symbol for gamma radiation is .
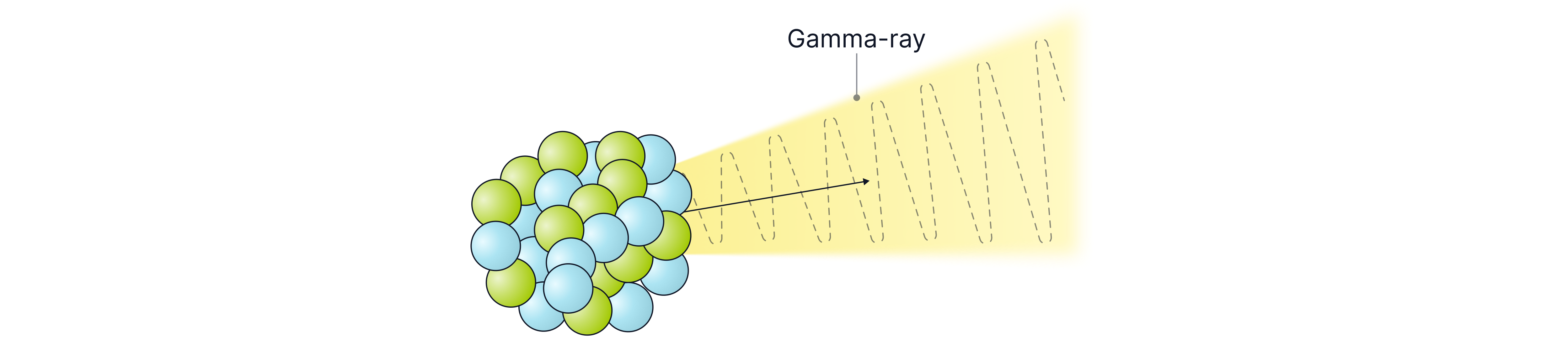
Gamma rays are the least ionising type of nuclear radiation, meaning that they can travel the furthest and thus are the most penetrating. Gamma radiation can travel unlimited distances in air (range = ) and requires thick lead or concrete to significantly reduce or absorb it.
Neutron
Neutron radiation is made up of neutrons released from the nucleus of an atom. Since a neutron has a mass of and no electrical charge, neutron radiation also has a mass of and a charge of . At GCSE, you do not need to know the penetration or ionising power of neutron radiation. The symbol for neutron radiation is simply .
Worked Example:
Complete the table for the three main types of radiation, alpha, beta and gamma.
Answer:
Worked Example:
A student is trying to determine the type of radiation emitted by a radioactive source.
They place different materials between the source and a detector and observe the following:
A sheet of paper does not reduce the count rate.
A thin sheet of aluminium causes the count rate to decrease significantly.
Adding a sheet of lead does not reduce the count rate any further compared with aluminium.
Using this information, identify the type of radiation being emitted by the source. Explain your reasoning.
Answer:
The radiation emitted by the source is beta radiation.
The count rate does not decrease when paper is placed in front of the source - so it cannot be alpha radiation, because alpha is stopped by paper.
The count rate does decrease significantly when aluminium is placed in the way, which shows the radiation is being partially blocked. This matches beta radiation, which is stopped by a few millimetres of aluminium.
Adding lead does not reduce the count rate any more than aluminium. This shows it cannot be gamma radiation, because gamma would be reduced further by thick lead.
Practice Questions
A technician uses different types of radiation to test how well materials block nuclear emissions.
State what happens to the range in air of alpha, beta and gamma radiation as you go from alpha beta gamma
Explain why beta radiation can pass through paper but is stopped by a few millimetres of aluminium.
-> Check out Hannah's video explanation for more help.
Answer:
The range increases. Alpha has the shortest range, gamma the longest.
Beta particles are smaller, faster and less ionising than alpha particles, so they penetrate further. However, they still interact with atoms enough (ionisation) to be absorbed by a thin metal sheet such as aluminium.
A student investigates how far alpha, beta and gamma radiation travel through air before they can no longer be detected by a GM tube.
State the typical range in air for alpha radiation.
State the typical range in air for beta radiation.
Explain why gamma radiation has a much longer range in air than alpha and beta radiation.
-> Check out Hannah's video explanation for more help.
Answer:
.
Around .
Gamma is an electromagnetic wave with no mass or charge, it is the least ionising, so it interacts very weakly with air particles/not at all. This means it can travel many metres through air before ionising an atom and losing some energy.