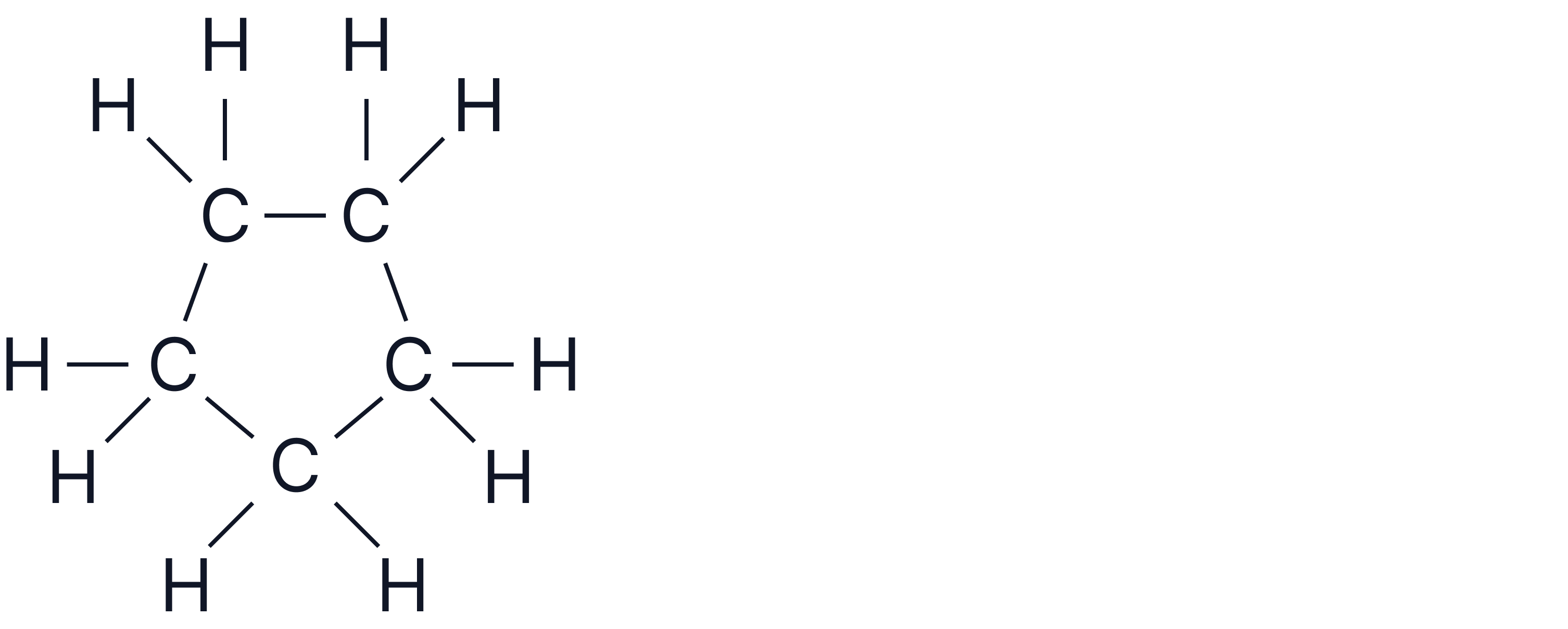¹³C NMR
Lajoy Tucker
Teacher

Contents
What is ¹³C NMR?
Carbon-13 NMR spectroscopy is a technique used to determine the structure of organic compounds by identifying the different carbon environments in a molecule.
Only carbon atoms of the isotope (about 1.1% of natural carbon) are NMR active.
Produces a spectrum where each peak represents a unique carbon environment.
Key Principles of ¹³C NMR
1. Number of Peaks
Each peak = one chemically unique carbon environment.
Symmetrical molecules may have fewer peaks than the total number of carbon atoms.
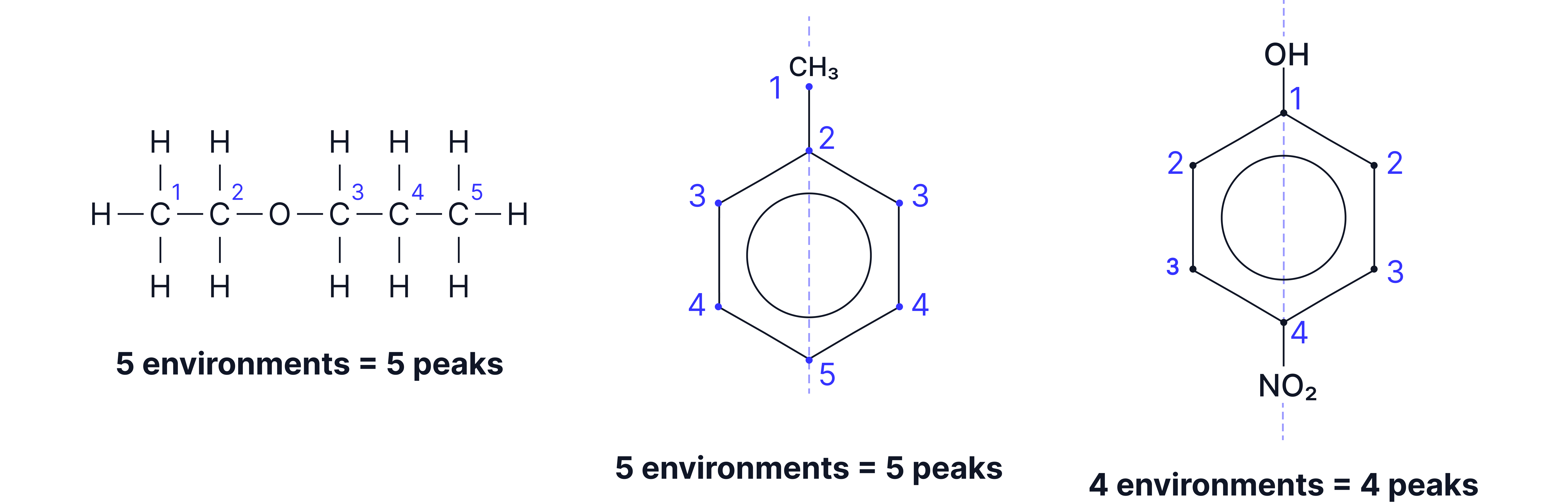
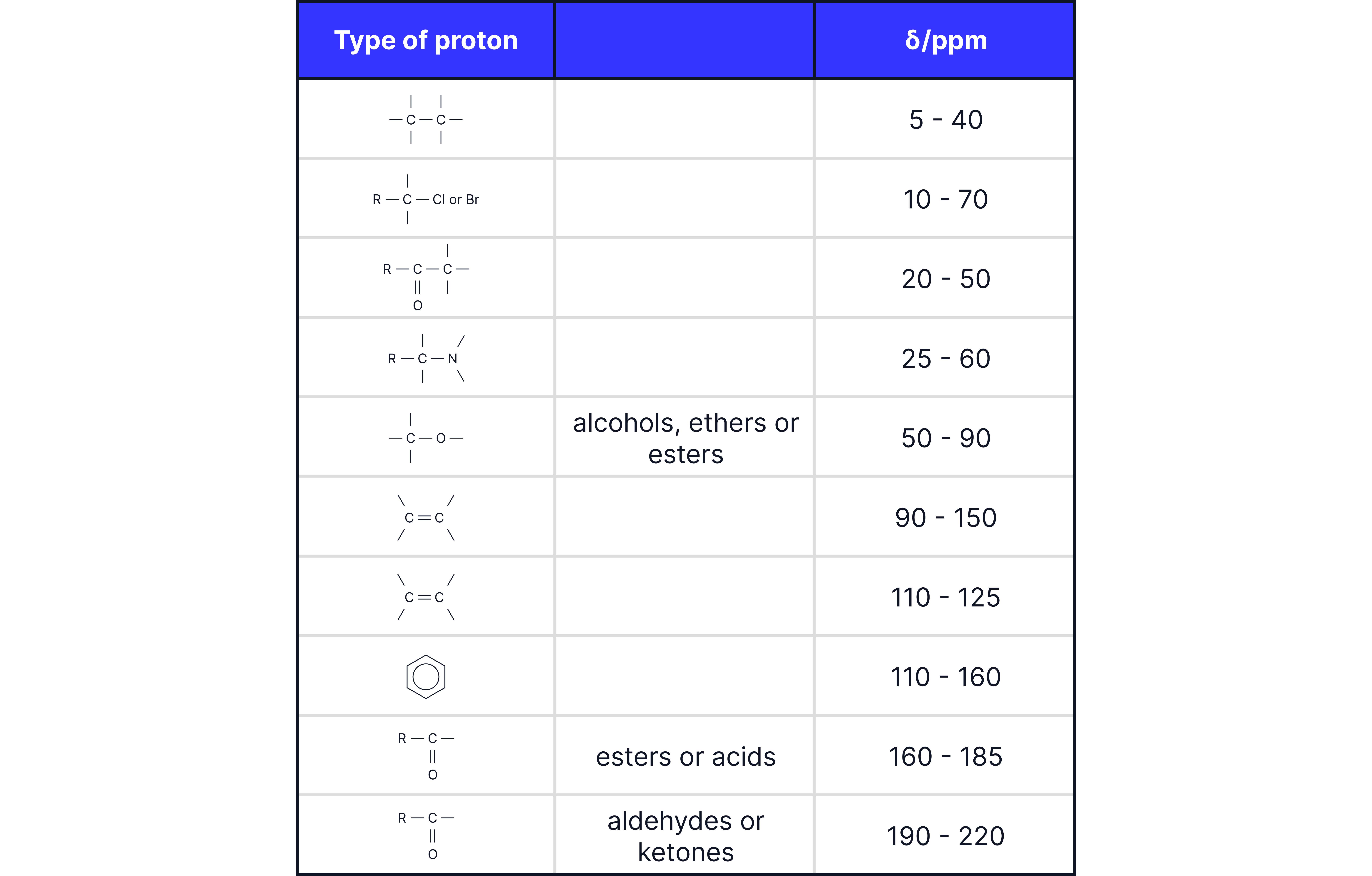
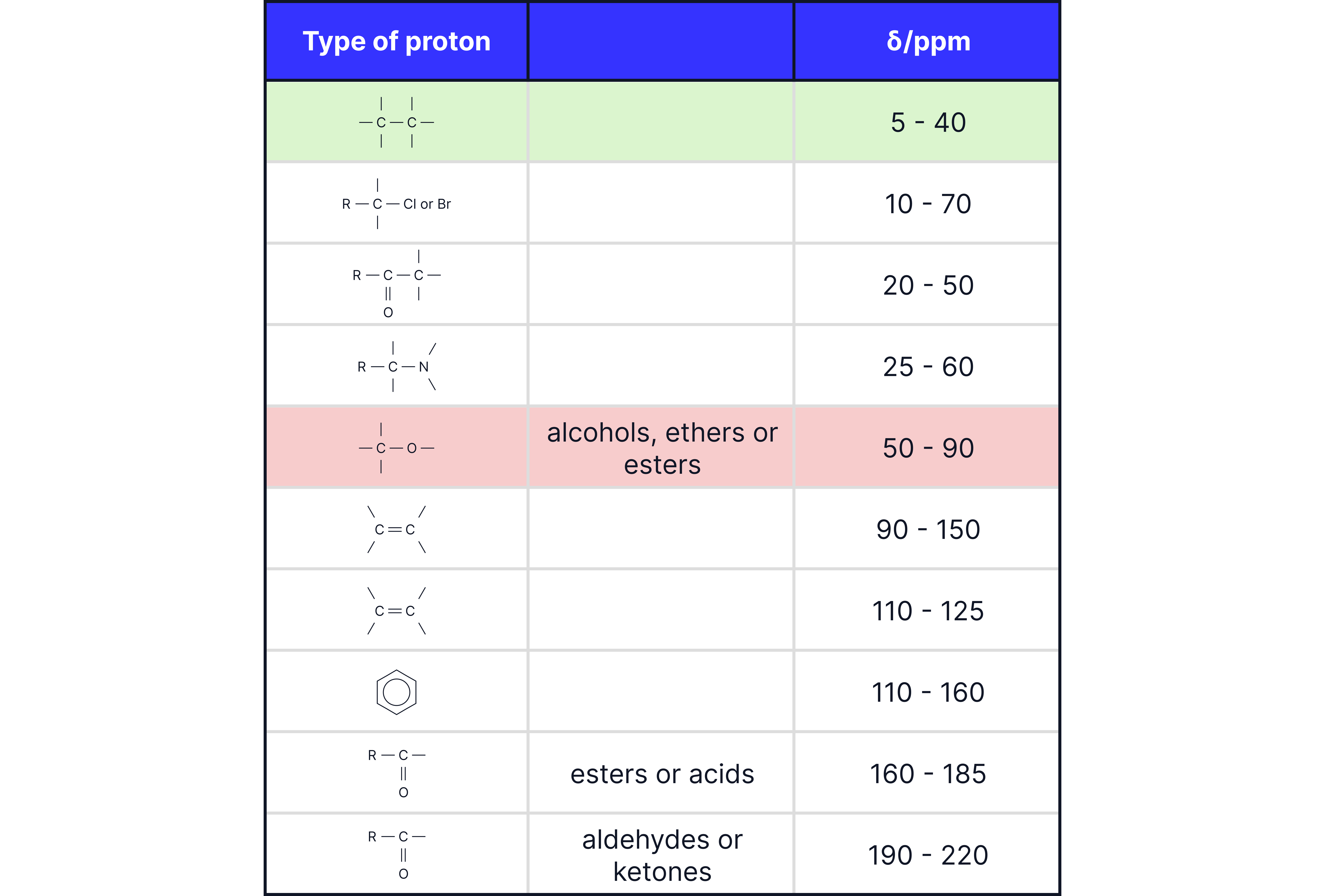
2. Chemical Shift ()
Measured in parts per million (ppm).
Indicates the type of carbon environment, influenced by:
Nearby electronegative atoms (e.g., , )
π systems (e.g., , aromatic rings, carbonyls)
TMS (tetramethylsilane) is used as the reference (0 ppm).
Interpreting a ¹³C NMR Spectrum
Count the number of peaks → tells you how many different carbon environments there are.
Use the chemical shift values to identify the type of each carbon.
Combine with other data (e.g. molecular formula, , ) to deduce the full structure.
Worked Example
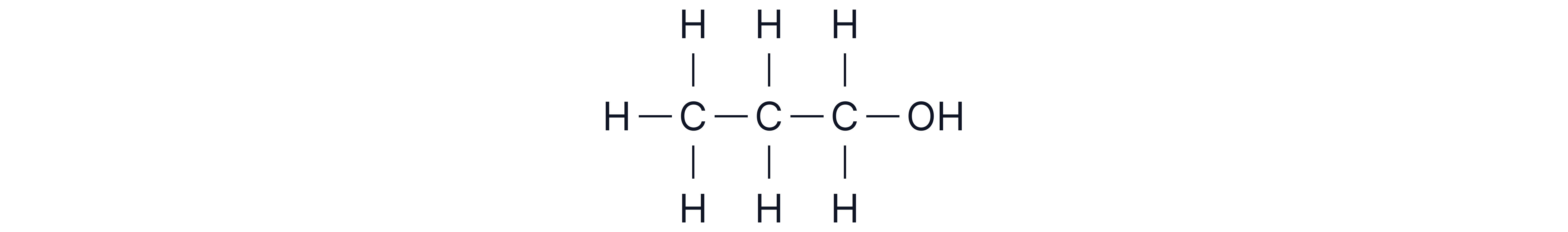
A compound has molecular formula and turns acidified potassium dichromate green. Use this information with the spectrum below to deduce its structure.
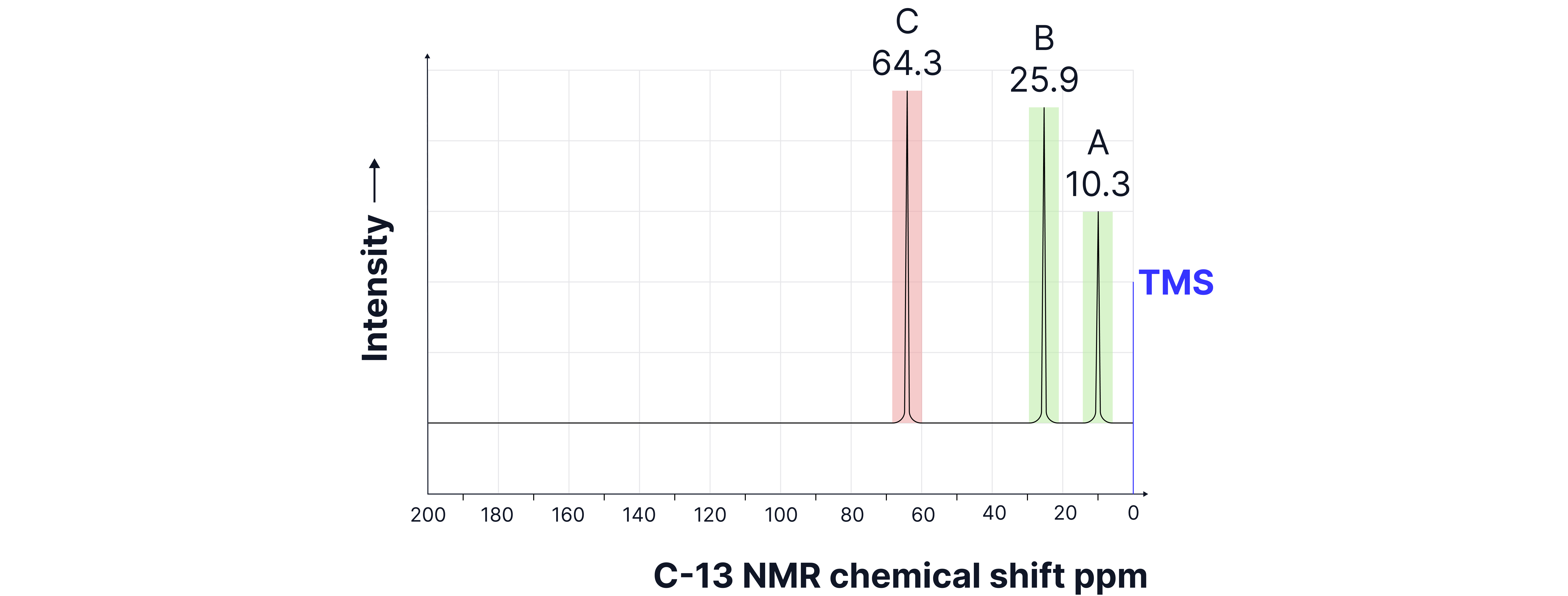
3 peaks therefore 3 unique carbon environments
Chemical shifts
Although peak c could correspond to the environment, the molecular formula given tells us there are no N atoms in this compound.
Acidified potassium dichromate turns green indicating a primary or secondary alcohol (or aldehyde but the data indicates a NOT a )
Suggested structure:
¹³C NMR Practice Questions Explainer
Practice Questions
Question 1
Compound A has 3 carbons and an absorption at in its infrared spectrum.
It has two peaks in its spectrum.
Deduce its structure.
Answer
Infrared peak indicates bond.
3 carbons but only 2 peaks indicates symmetry.

Question 2
A primary amine with 5 carbons has three peaks in its .
Deduce its structure.
Answer
Primary amine has the general structure .
5 carbons but only 3 peaks indicates symmetry.

Question 3
Compound D has the molecular formula .
and has only 1 peak in its spectrum.
Deduce its structure.
Answer
5 carbons but only 1 peak indicates symmetry.
Formula indicates alkene or cycloalkane.
Cycloalkane would give 5 identical carbon atoms.