Acylation
Lajoy Tucker
Teacher

Contents
What is Acylation?
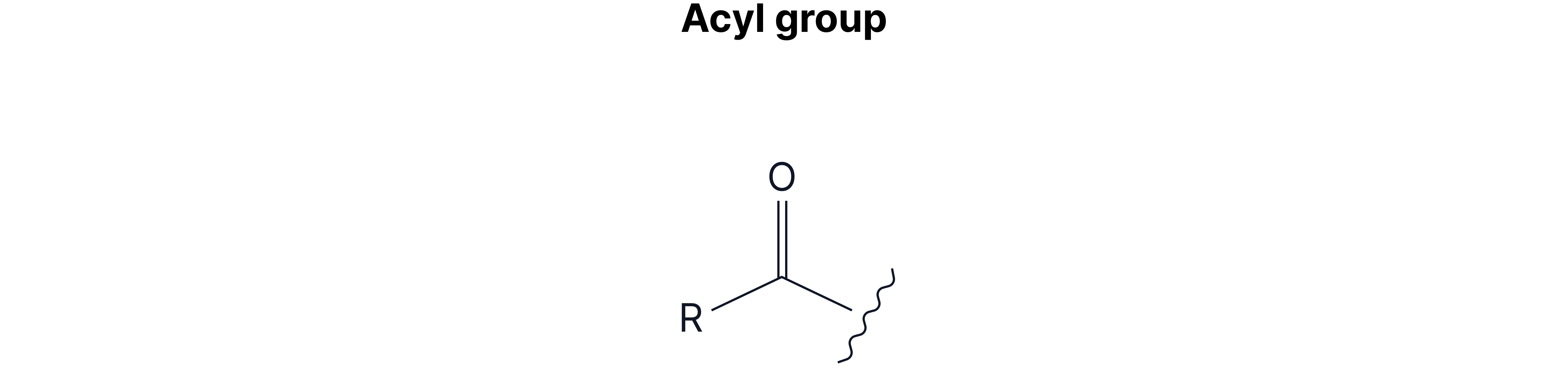
Acylation reactions introduce the acyl group using reagents including acyl chlorides and acid anhydrides.
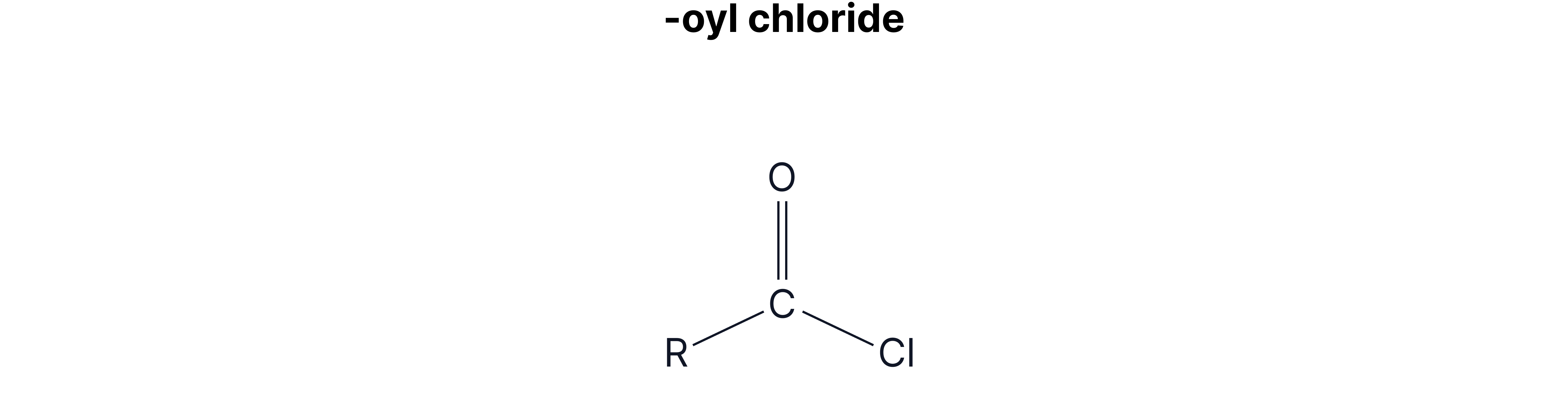
Acyl Chloride
Acyl chlorides are known to be the most reactive derivative of carboxylic acids.
This is due to two electronegative atoms being attached to the carbonyl C, so it is more 𝛿+, and more attractive to a nucleophile.
Acylation Explained
Reactions of Acyl Chloride (condition: room temperature)
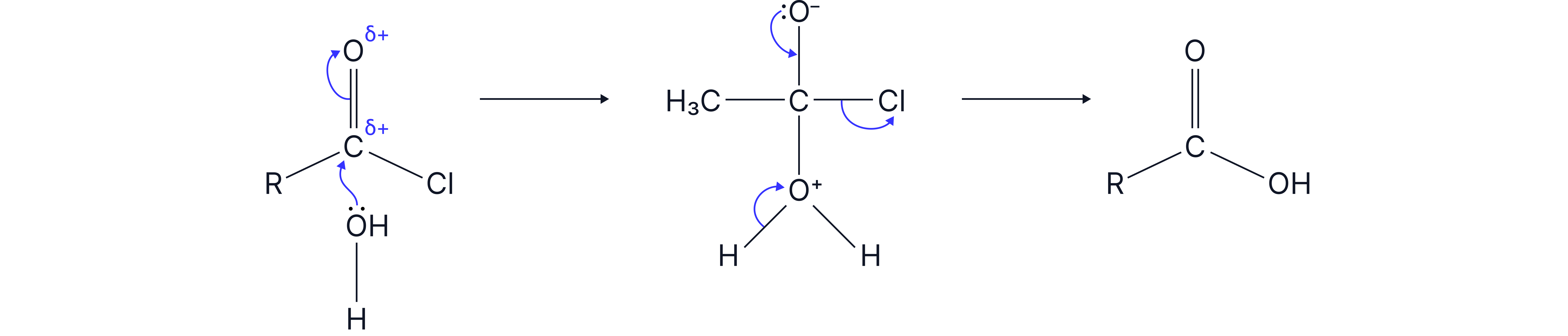
1. With water
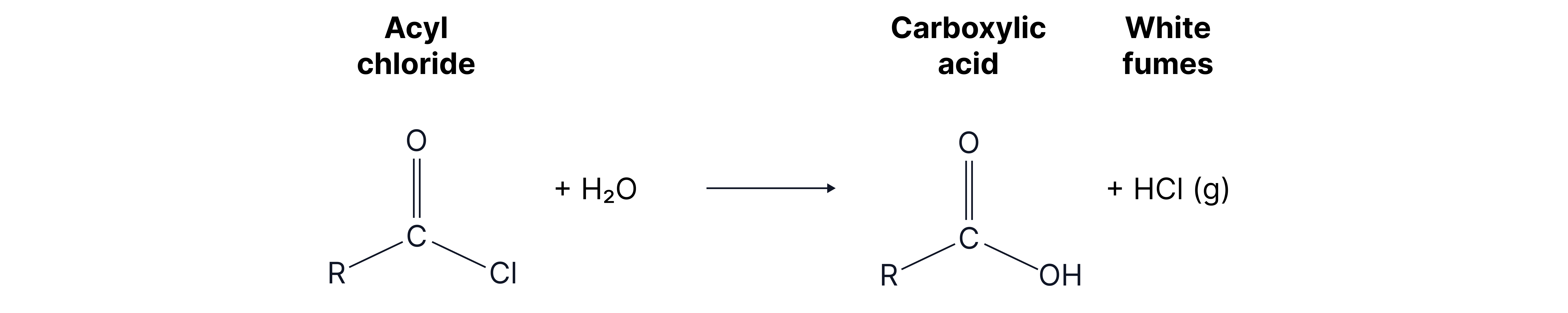
Mechanism: nucleophilic addition elimination
2. With alcohol
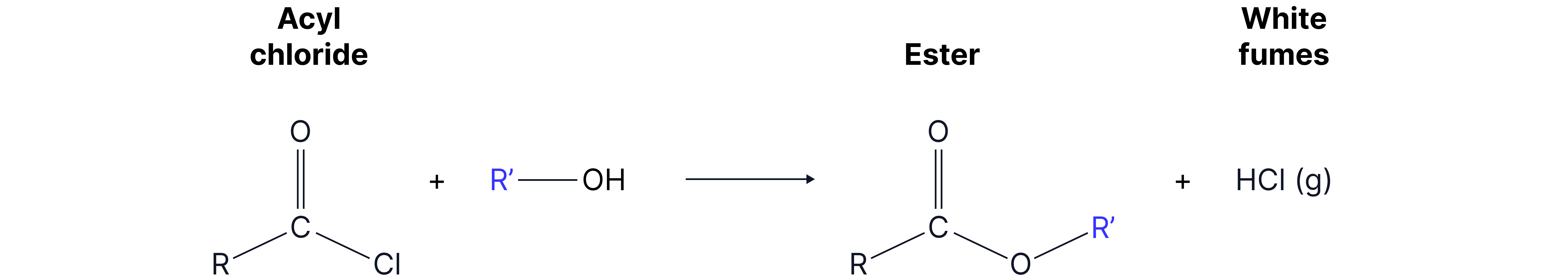
Mechanism: nucleophilic addition elimination
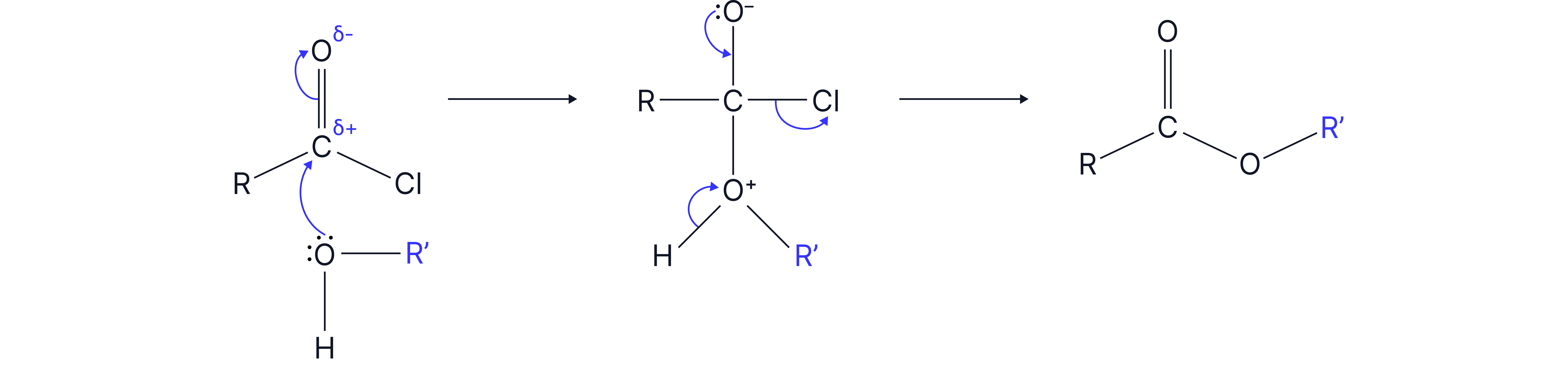
When you compare this to the synthesis of esters using –COOH, you find that using an acyl chloride is more efficient: it is an irreversible reaction, compared to a reversible reaction with –COOH. Also, since acyl chlorides are more reactive, there is no need for a catalyst.
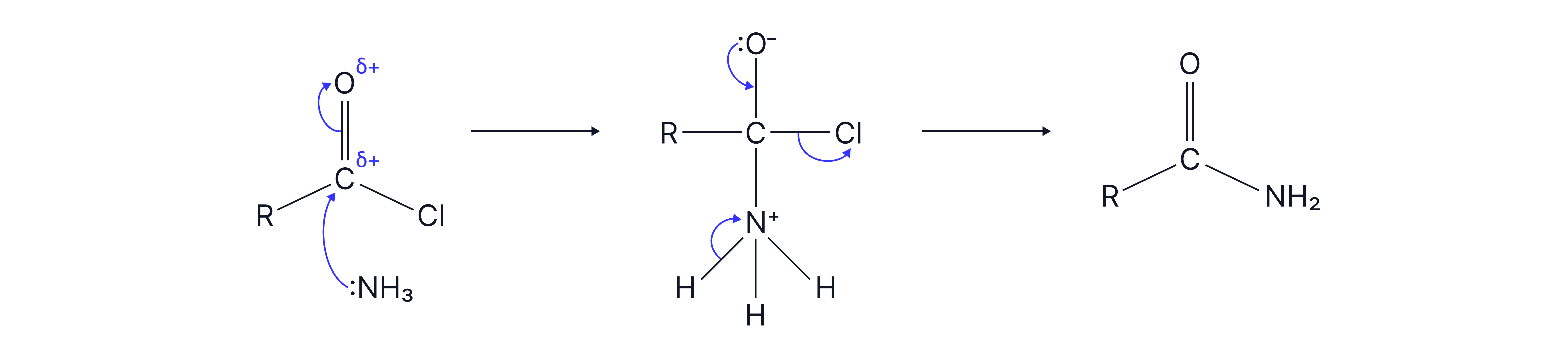
3. With ammonia
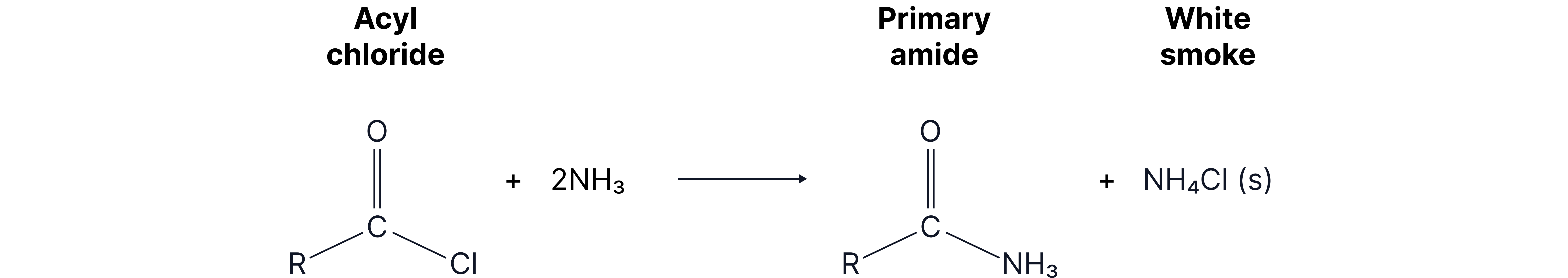
Mechanism: nucleophilic addition elimination
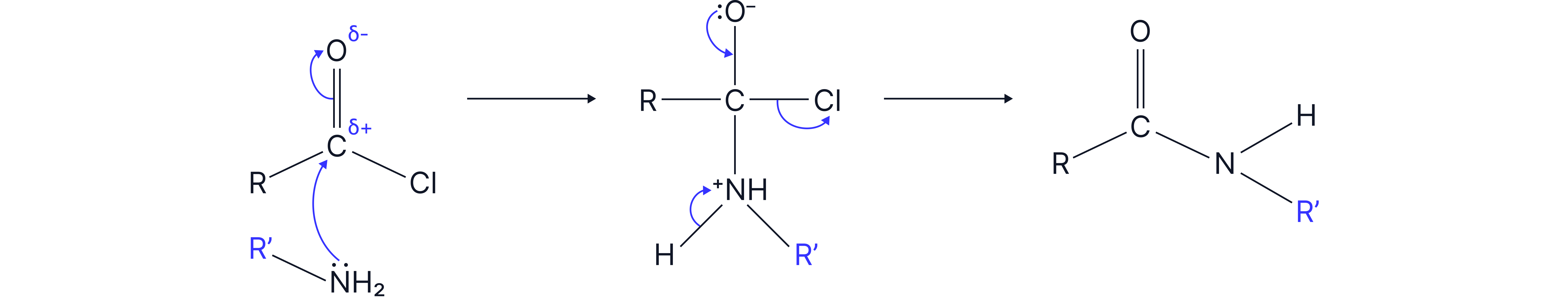
4. With primary amines
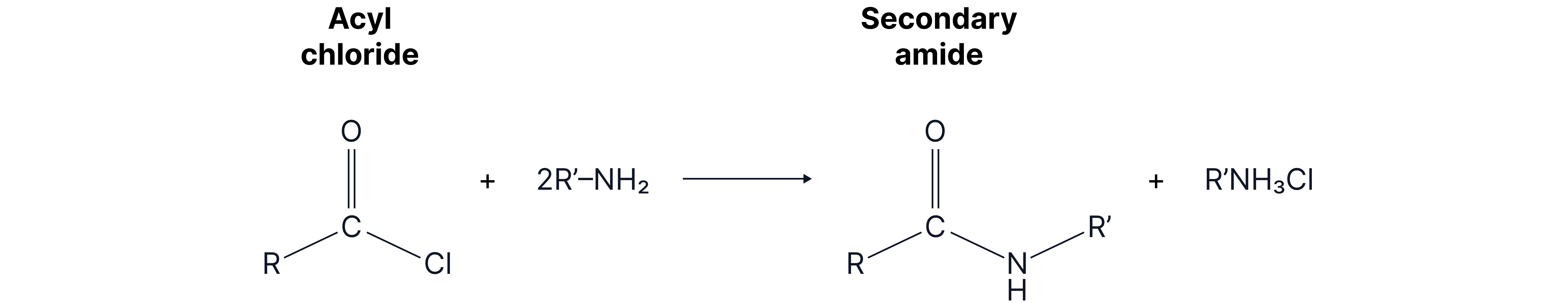
Mechanism: nucleophilic addition elimination
Acid Anhydrides
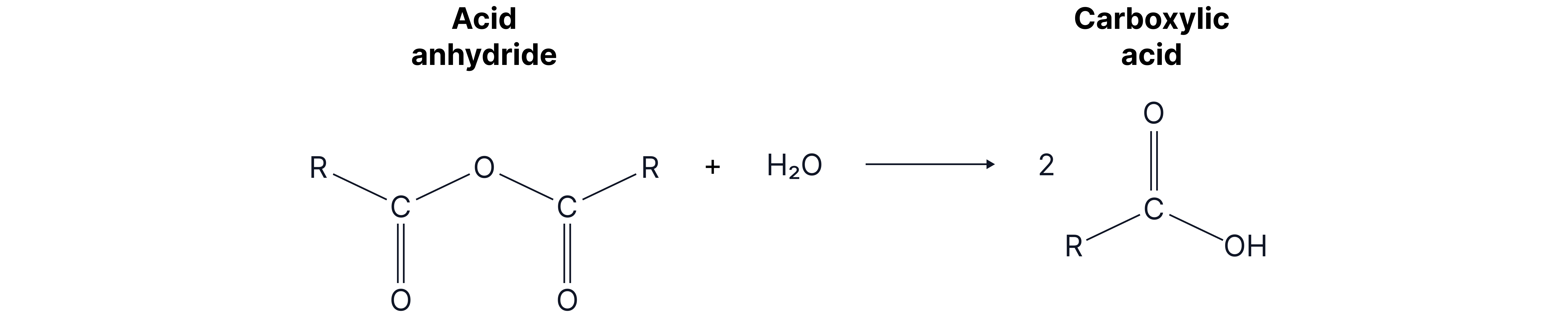
The reactivity of acid anhydrides is very similar to acyl chlorides, so the same reactions happen and functional group of products form. Acid anhydrides are often used instead of acyl chlorides as no toxic and corrosive HCl fumes are formed.
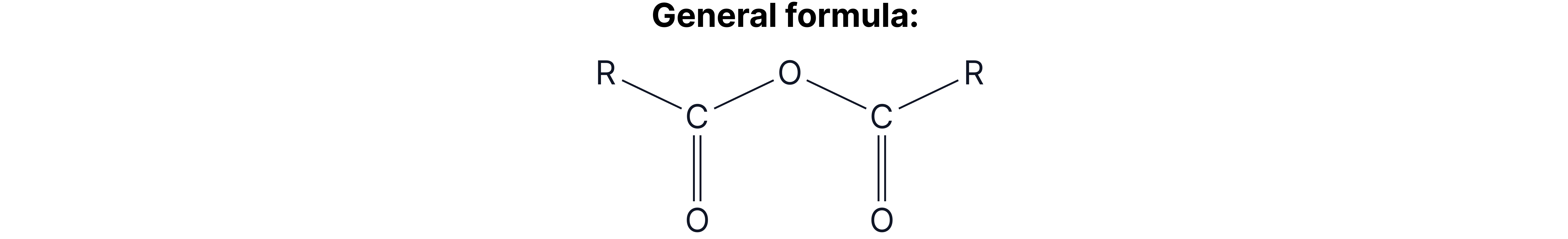
Reactions of Acid Anhydrides (condition: room temperature)
1. With water
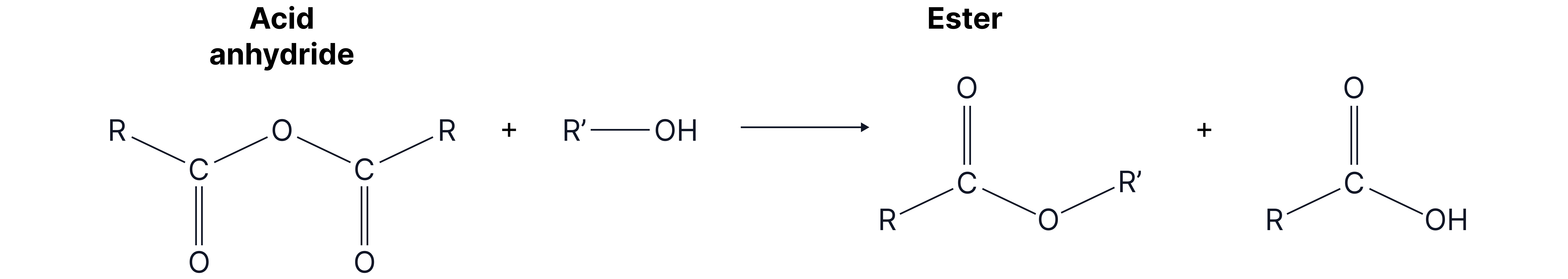
2. With alcohol
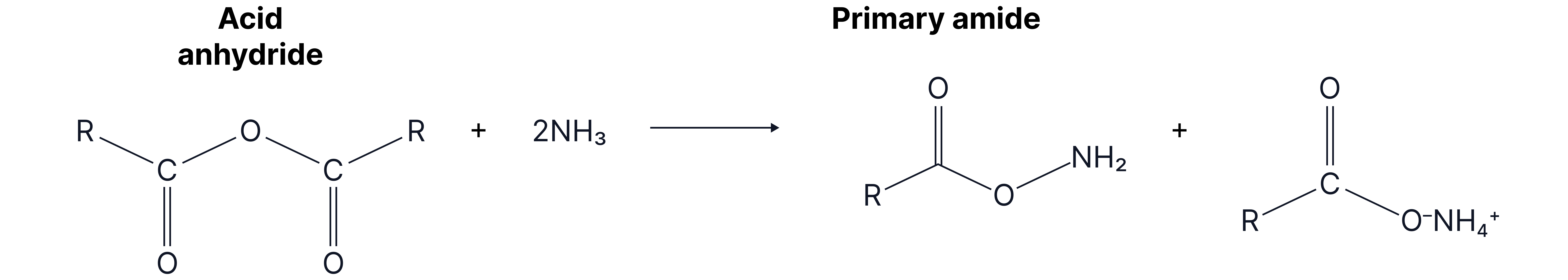
3. With ammonia
4. With primary amines
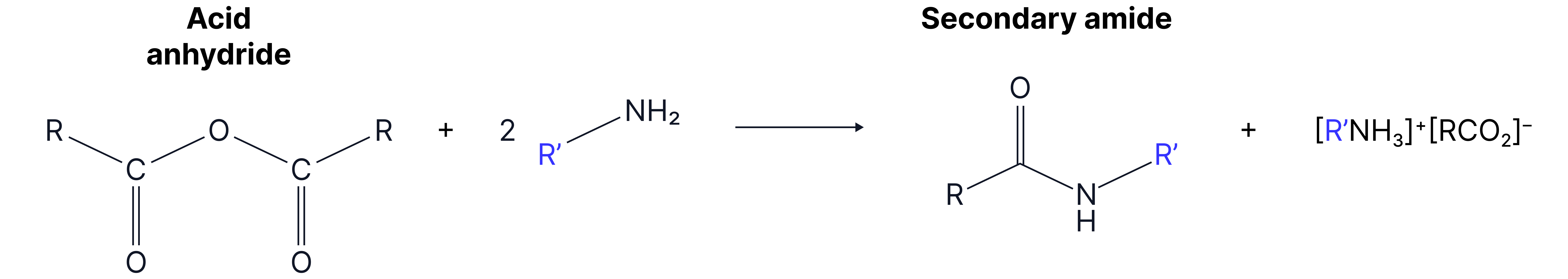
Practice Question
Question:
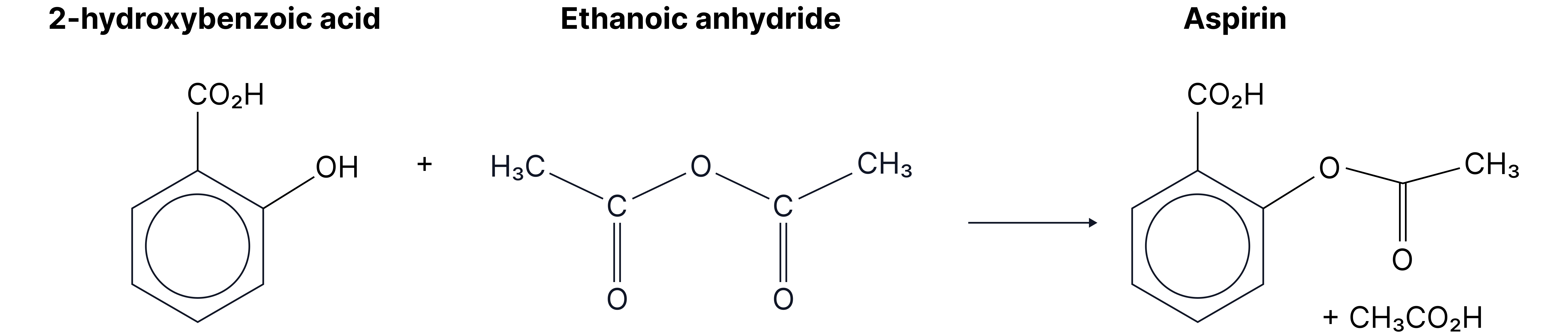
Complete the equation for the reaction between 2-hydroxybenzenecarboxylic acid and ethanoic anhydride.
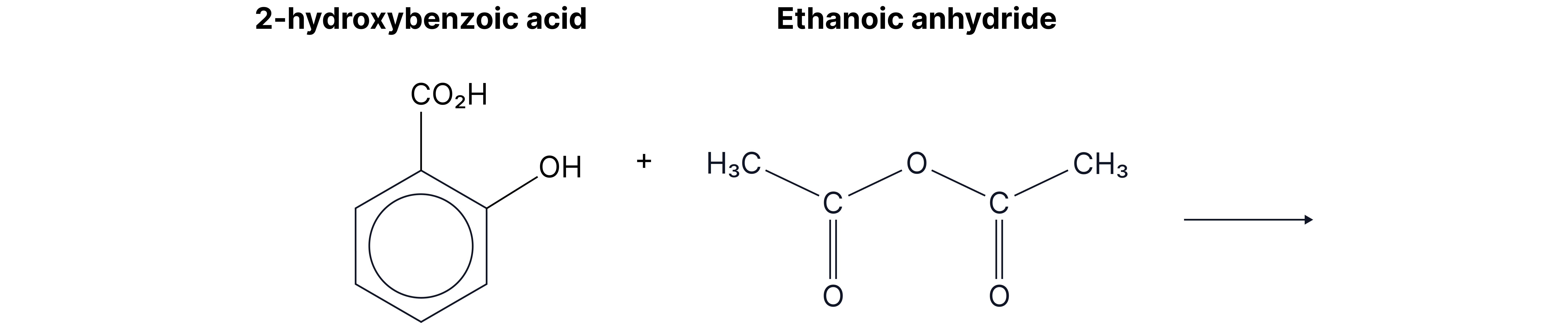
Answer: