Esters
Lajoy Tucker
Teacher

Contents
What Are Esters?
Esters are organic compounds with the functional group . They are derivatives of carboxylic acids, formed via a condensation reaction with alcohols.
General formula:
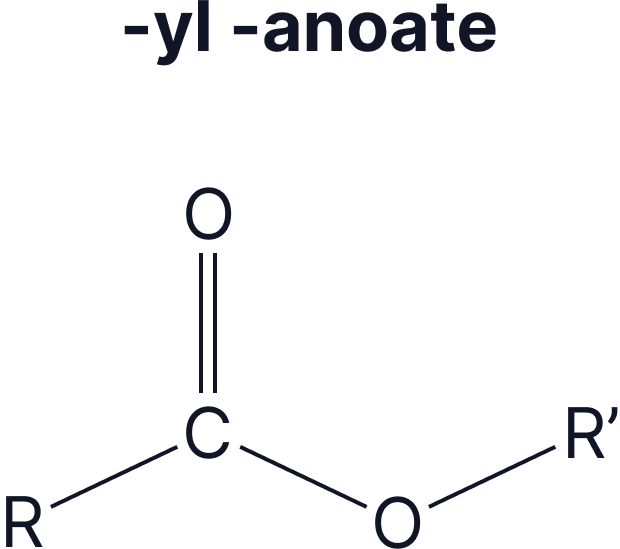
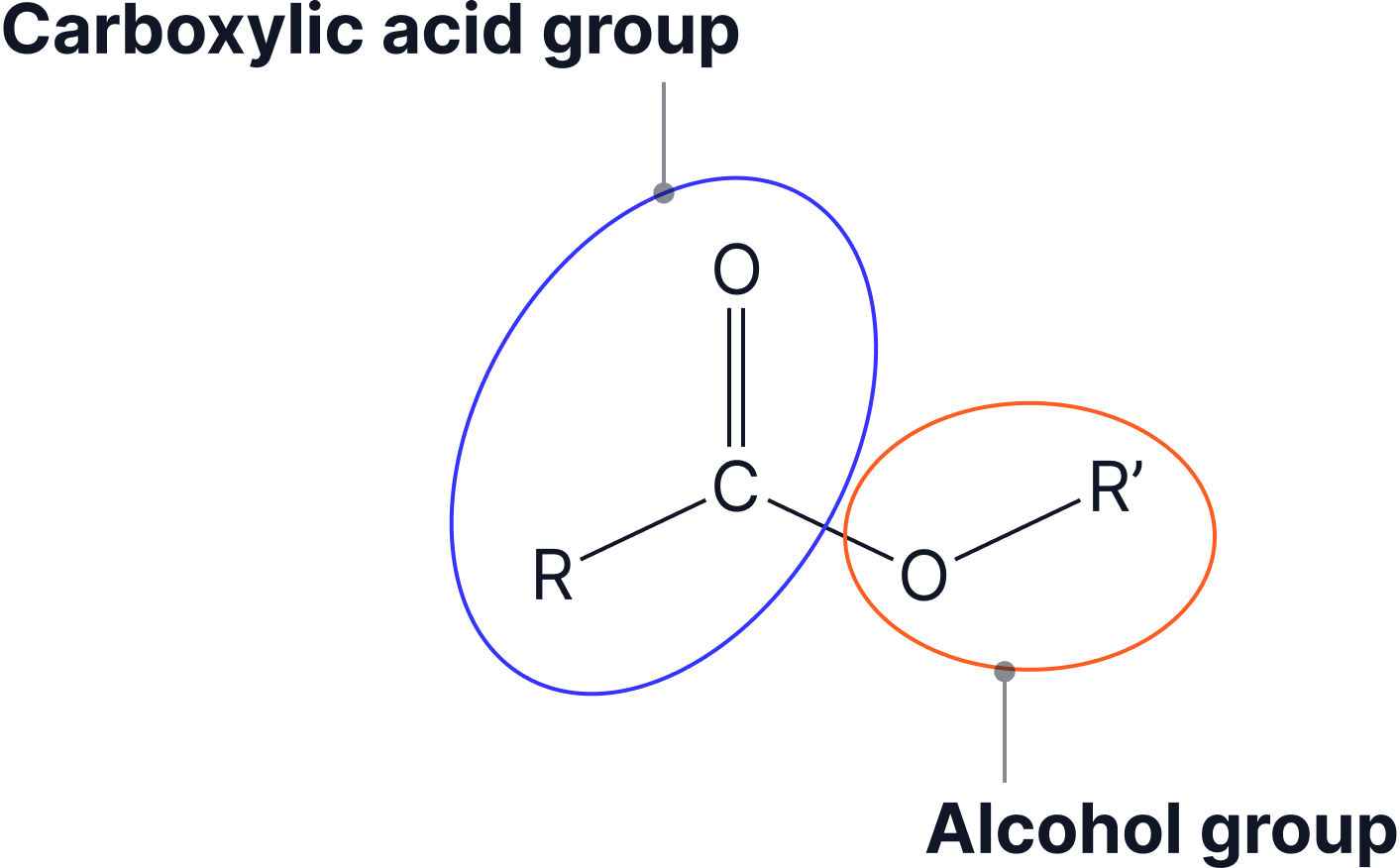
The naming of esters can be split into two: the part that comes from the alcohol and the part that comes from the carboxylic acid.
- The alcohol group gives the –yl ending
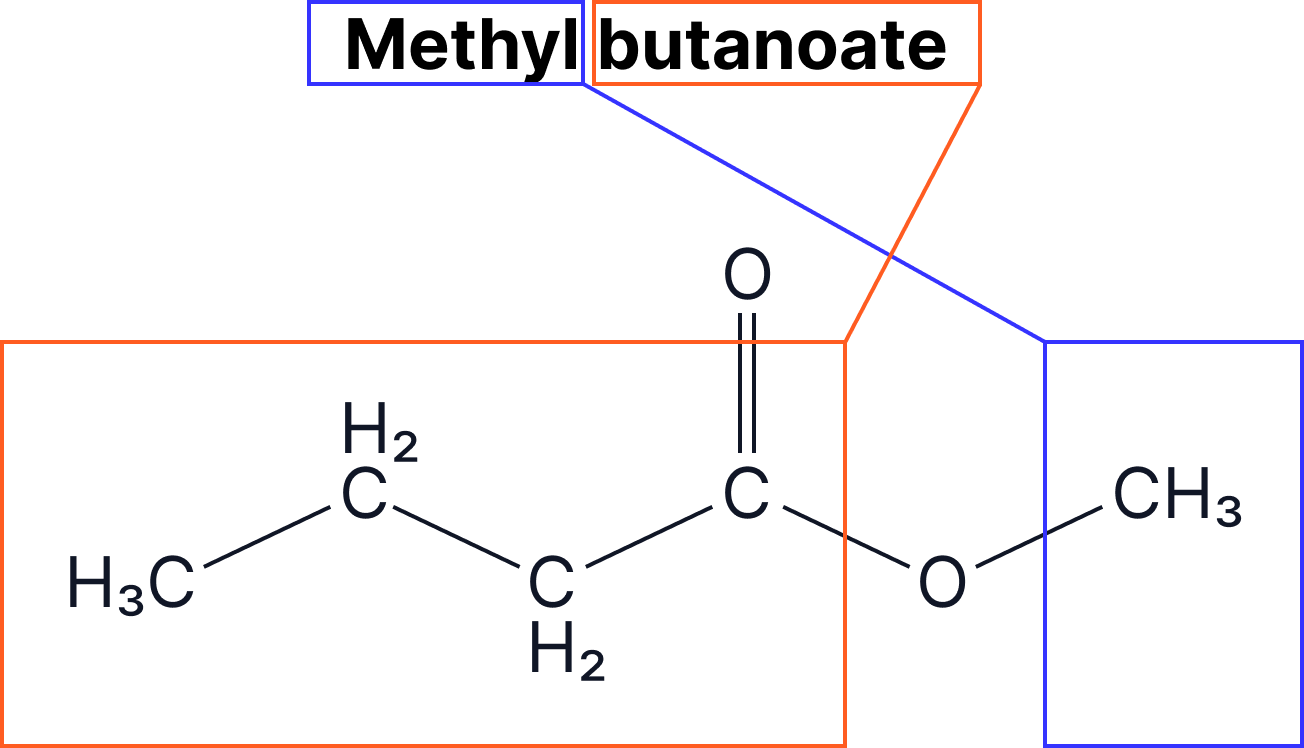
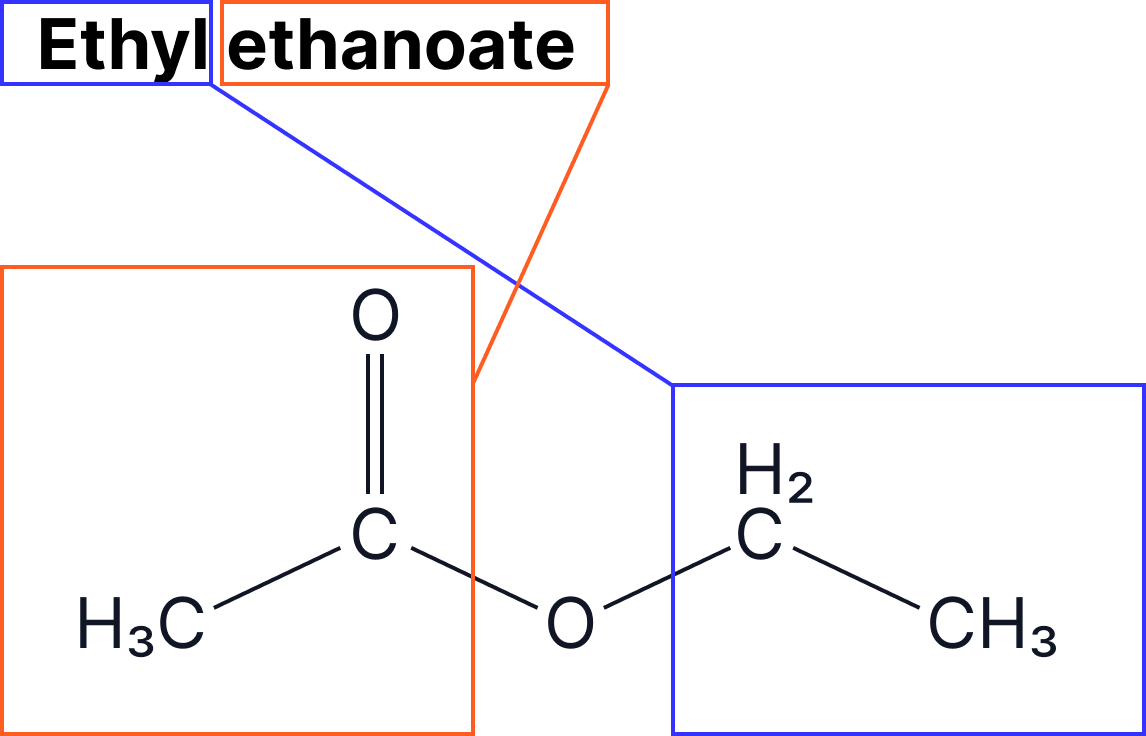
- The carboxylic acid gives the –anoate ending
Uses Of Esters:
-
Natural & artificial flavouring
-
Perfume
-
Solvents
-
Plasticiser
Esters Explainer Video
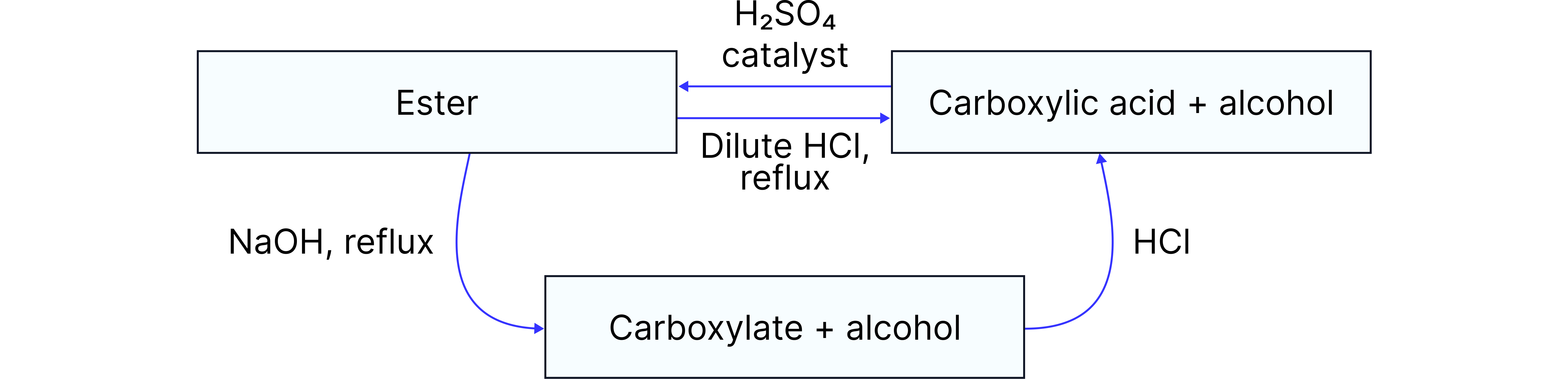
Esterification and Hydrolysis of Esters
Considering the nomenclature, one way of forming esters is the reversible reaction of carboxylic acid with an alcohol, using a strong acid catalyst and heating under reflux.
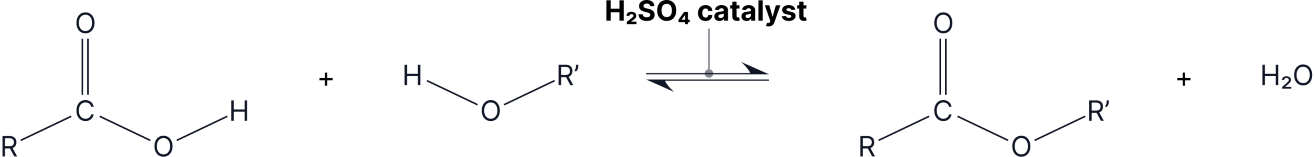
The reverse of this reaction (ester → carboxylic acid/carboxylate ion + alcohol) can happen by two different types of hydrolysis:
1. Acid hydrolysis

Reversible reaction
Reagent: dilute HCl
Condition: heat under reflux
Example:
Propyl butanoate + water → butanoic acid + propanol
2. Base Hydrolysis
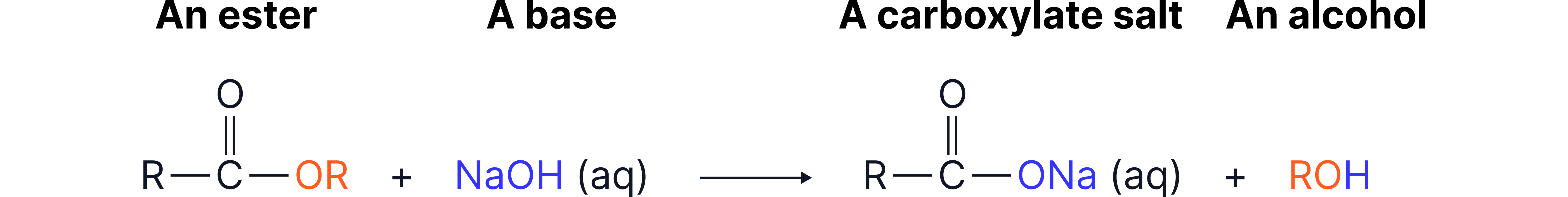
Irreversible reaction
Reagent: NaOH
Condition: heat under reflux
Addition of HCl to the carboxylate will form the carboxylic acid
Example:
Propyl butanoate + sodium hydroxide → sodium butanoate + propanol
Fats and Oils
Under alkaline conditions, animal fats and vegetable oils can undergo hydrolysis
When these fats and oils are heated with NaOH or KOH, they form soap
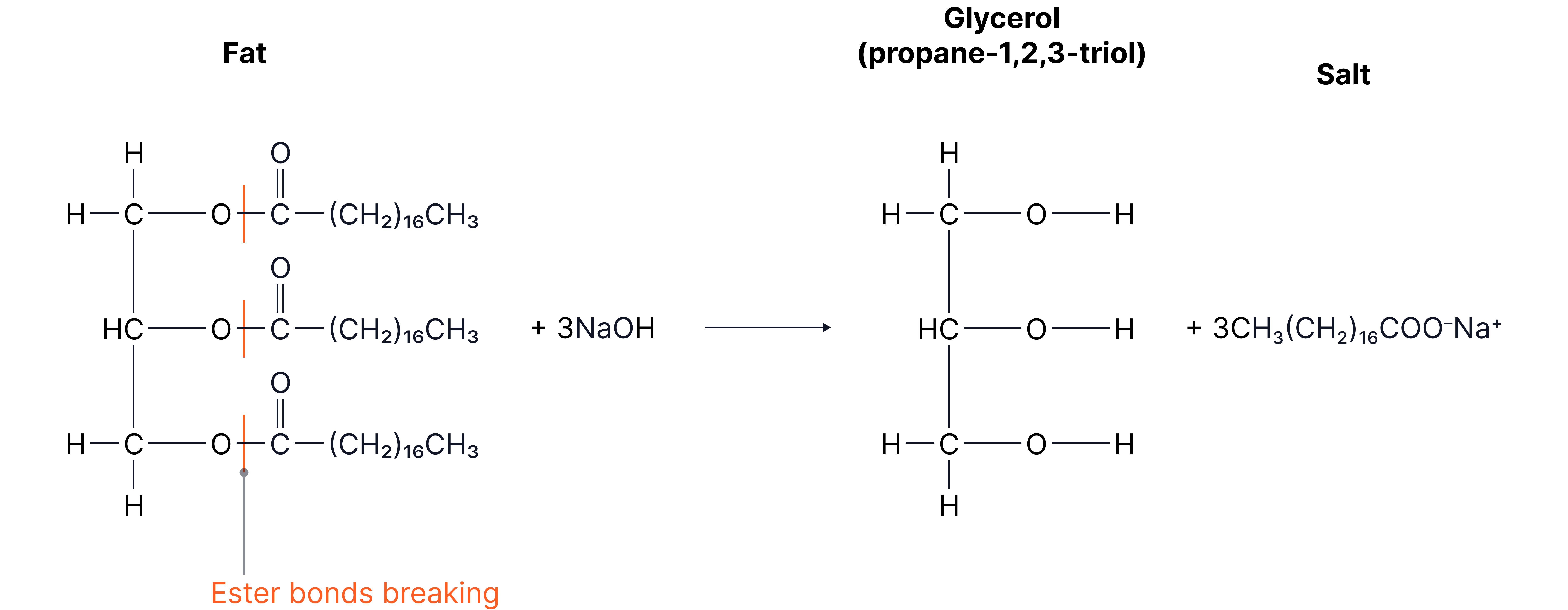
Glycerol (propane-1,2,3-triol)
Glycerol has 3 alcohol groups which means it can form hydrogen bonds quite easily
They are used in glues and in food
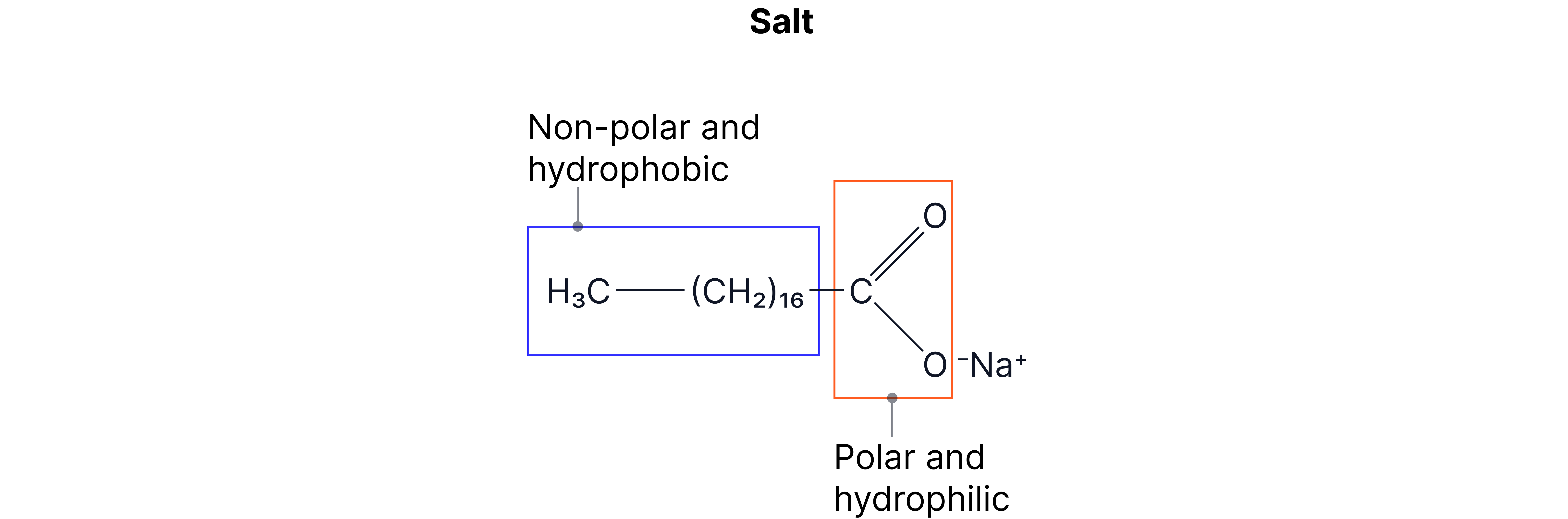
The carboxylate salts can act as soaps
Soap has a hydrophilic and hydrophobic end which makes it useful to wash grease away. The hydrophilic end mixes with water and the hydrophobic end mixes with grease. As a result, water and grease can be mixed and washed away.
Question 1
Which of the following would produce ethyl ethanoate?
A. Propanoic acid + Ethanol
B. Ethanoic acid + Propan-1-ol
C. Ethanoic acid + Propan-2-ol
D. Propanoic acid + Propan-1-ol
Answer:
B
Question 2
Ethyl ethanoate is heated under reflux with dilute hydrochloric acid.
A. Name the type of reaction that takes place
B. Write a balanced equation for this reaction
Answers:
A. Acid hydrolysis (or hydrolysis is acceptable – but specifying acid hydrolysis is better for clarity).
B.
(Ethyl ethanoate + water ⇌ ethanoic acid + ethanol)
Question 3
A student wants to prepare an ester from butanoic acid and methanol.
A. Write the structural formula of the ester formed
B. State the catalyst and conditions needed.
Answers:
a) (This is methyl butanoate)
b) Catalyst: Concentrated sulfuric acid . Conditions: Heat under reflux
Summary Table
Reaction | Reagents | Conditions | Products |
|---|---|---|---|
Esterification | Alcohol + Carboxylic Acid | Conc. H₂SO₄, reflux | Ester + Water |
Acid Hydrolysis of Ester | Ester + Water + H⁺ | Reflux with dilute acid | Carboxylic Acid + Alcohol |
Base Hydrolysis of Ester | Ester + NaOH (aq) | Reflux | Carboxylate Salt + Alcohol |