Carboxylic Acids
Lajoy Tucker
Teacher

Contents
Carboxylic Acids
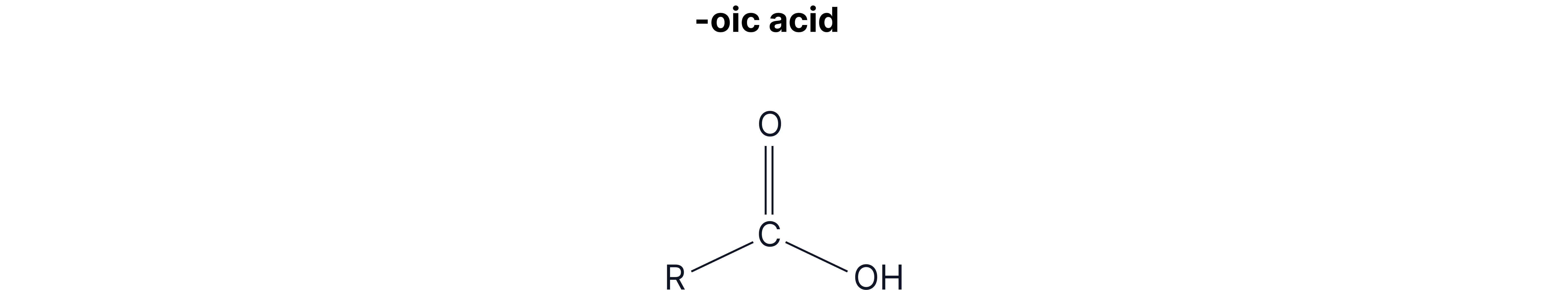
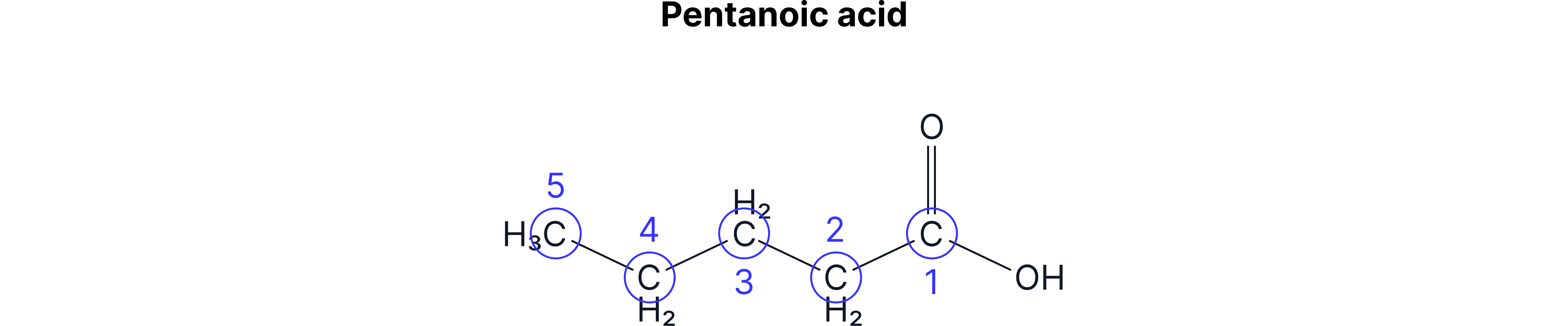
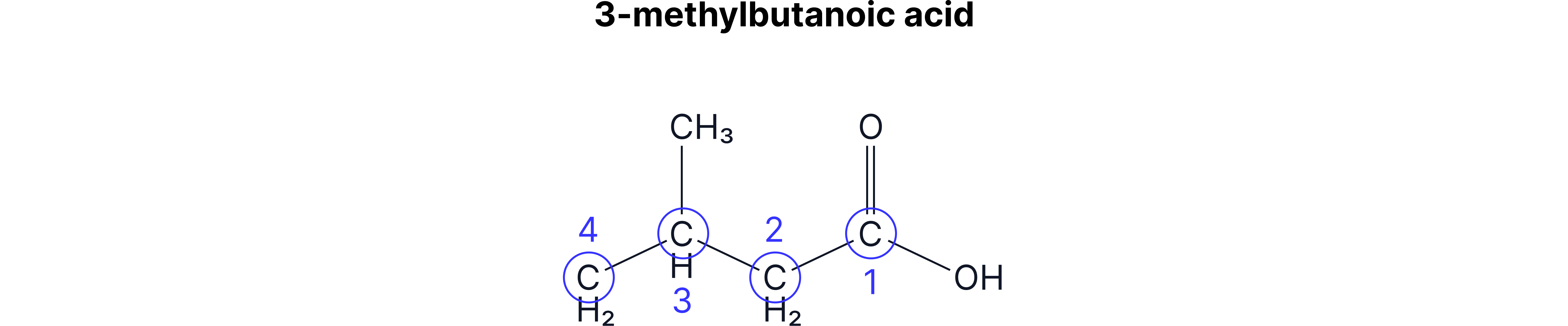
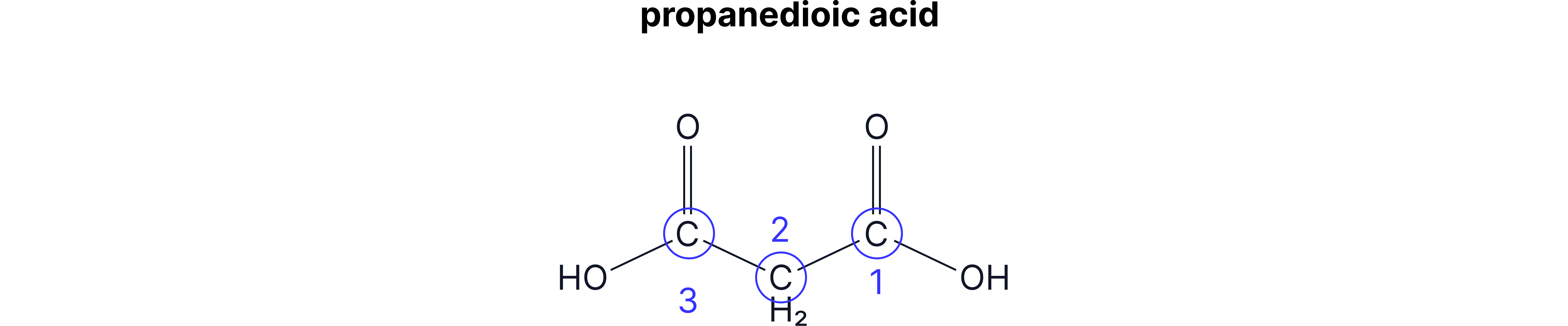
When naming carboxylic acids, you name the rest of the chain as usual (number 1 starts from the carboxylic acid C) and add –oic acid to the end.
The –COOH group will always be at the end of the chain, so you don’t need to worry about numbering the carboxylic acid group.
Carboxylic acids are weak acids which means that there is slight dissociation in water, and it is in equilibrium with its salt.
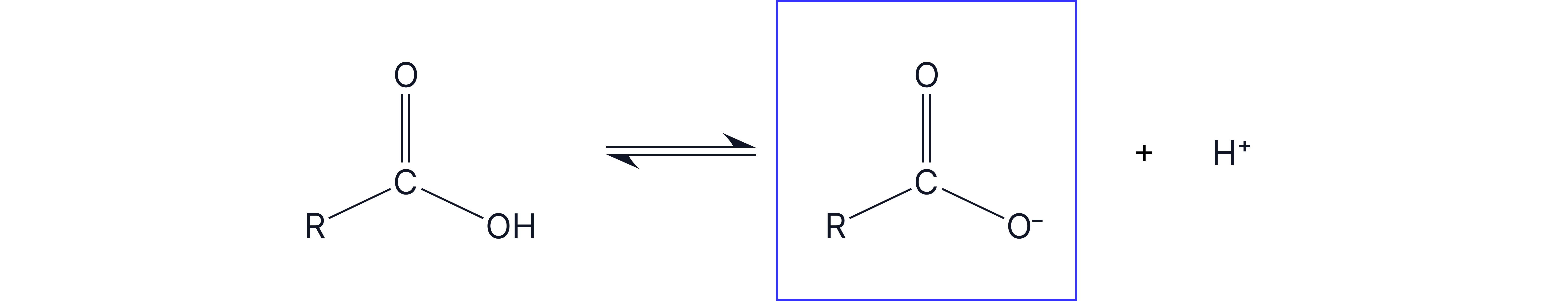
The driving force of this dissociation is the delocalisation of the carboxylate ion. In reality, the negative charge on the oxygen is spread out and distributed over the O-C-O bond, creating a cloud of π electrons.
This is a stabilisation effect.
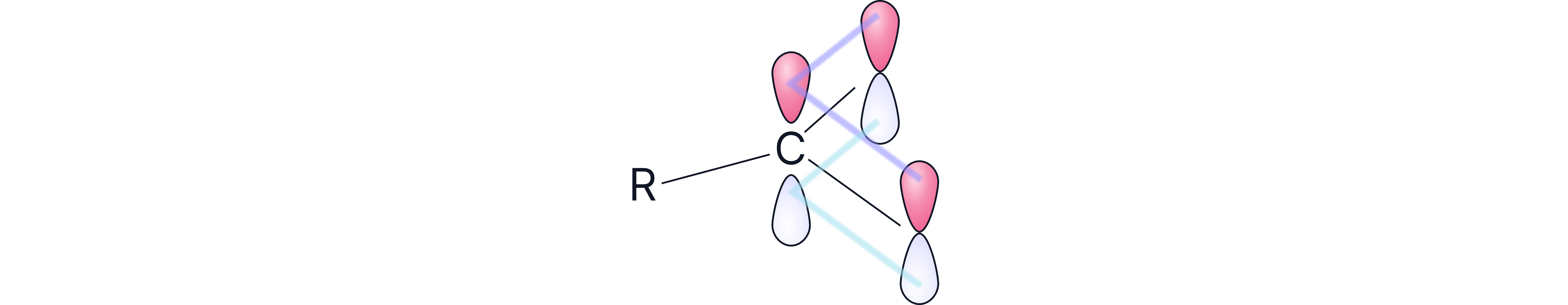
Carboxylic Acids Explained
There are two factors that affect the stability of the carboxylate ion:
If the R group is an alkyl group, then it is an electron donating group. They have a POSITIVE INDUCTIVE EFFECT. This is because alkyl groups have an electron donating nature, so they push electrons towards the cloud of π electrons on the COO-. Since there will be more electron density on COO- it is more likely to react and therefore less stable.
Note that the longer the alkyl chain, the greater electron donating power.
If the R group contains anything electron withdrawing (such as electronegative atoms F and Cl) then there is a negative inductive effect. These atoms cause withdrawal of electron density from the COO- and this results in less electron density concentrated on the COO-.
As a result, the carboxylate ion becomes more stable.
Don't forget, the more electron density on the COO-, the weaker the acid. The less electron density on the COO-, the stronger the acid.
Why is butanoic acid less acidic than ethanoic acid?
The carbon chain in butanoic acid is longer than the carbon chain on ethanoic acid.
So, the electron donating effect of the alkyl groups will be greater in butanoic acid which means there is greater electron density on the COO- than in ethanoic acid.
The greater electron density means it is less stable and more likely to attack H+ and form the carboxylic acid – so weaker acid
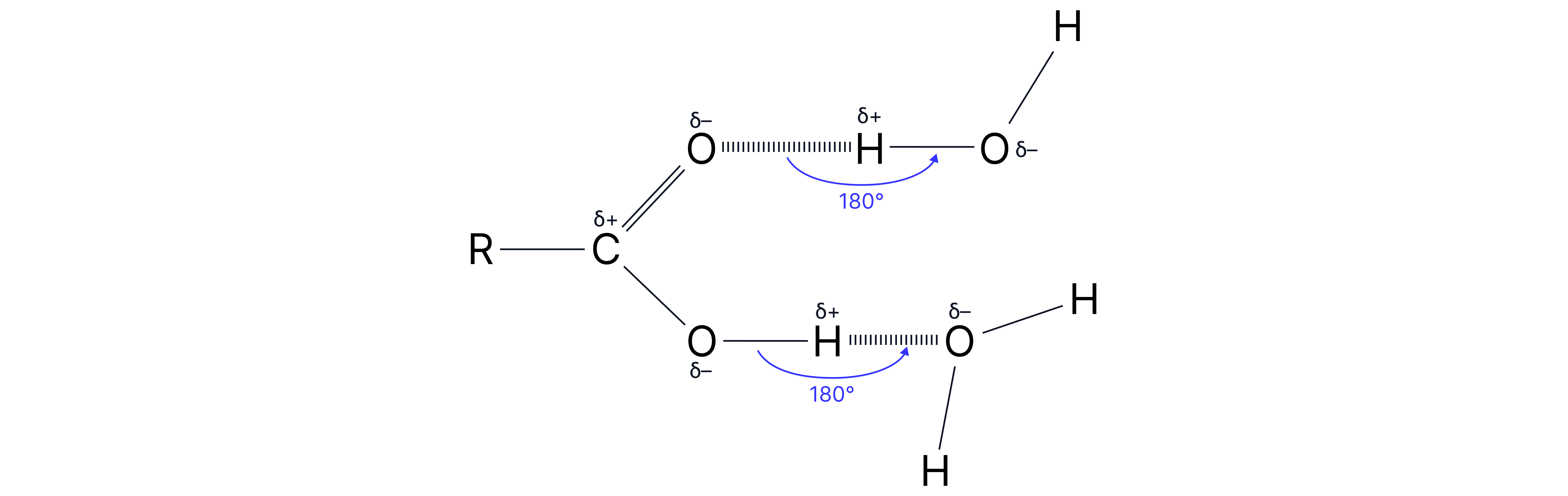
Carboxylic acids with carbon chains up to 4 carbons are soluble in water.
This is because hydrogen bonds can form between the carboxylic acid and water.
Reactions of Carboxylic Acids
1.
2.
3.
This is used as a functional group test for carboxylic acids, where the CO₂ causes effervescence.
4. Oxidation
Methanoic acid is the only carboxylic acid that can be oxidised since there is a C-H bond available for oxidation.

Practice Question
Question:
Write the balanced equation for the reaction of ethanoic acid . How is used as a functional group test for –COOH?
Answer:

The causes effervescence which is used to show the formation of a carboxylic acid.