Addition Polymers
Lajoy Tucker
Teacher

Contents
Introduction and Definition
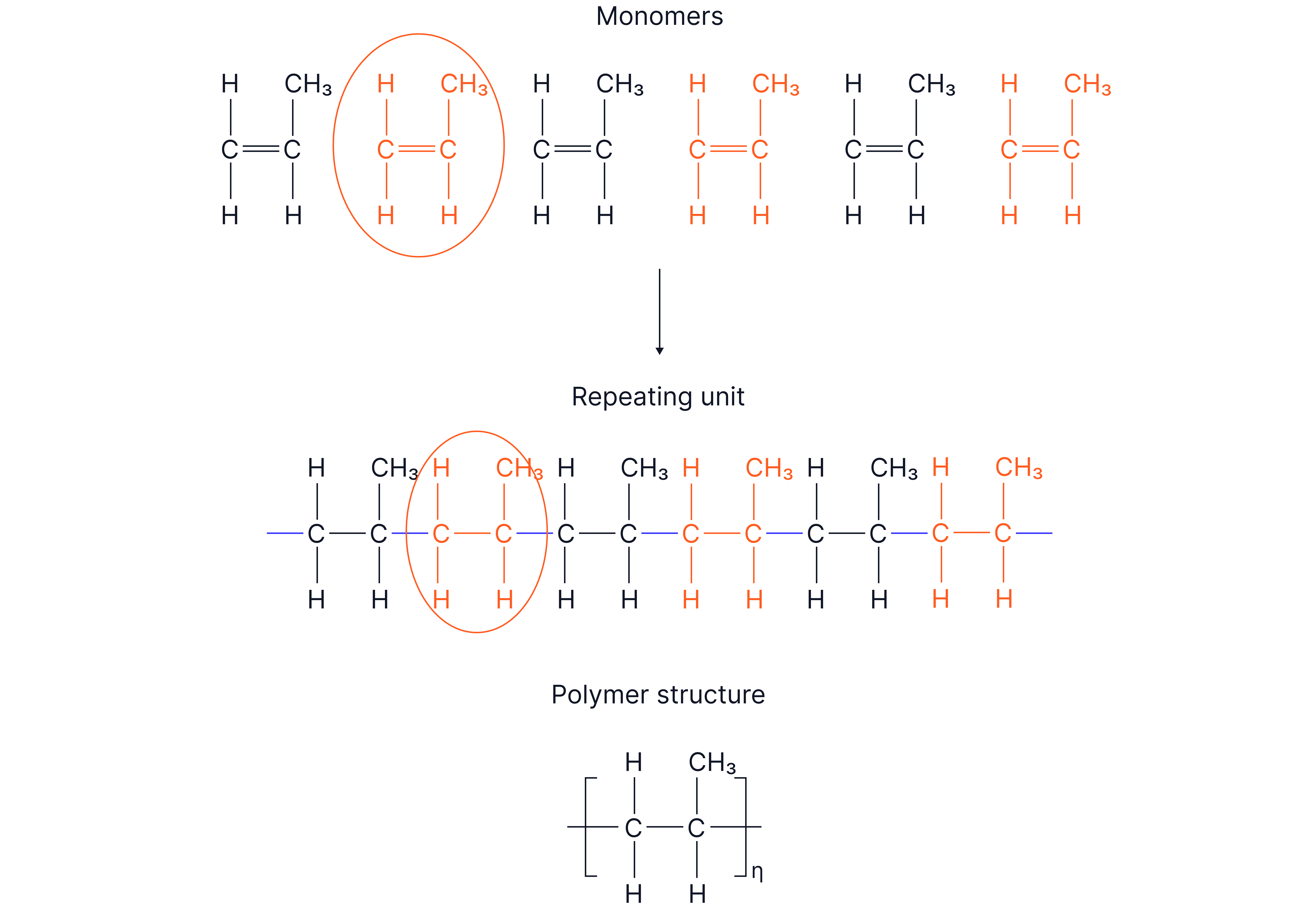
Addition polymerisation – many alkene molecules join to make one long polymer chain.
The C=C opens. New C–C single bonds form between units.
Key Terms
Monomer – the small repeating molecule (usually an alkene).
Polymer – large molecule made from many monomers.
Repeating unit – the smallest section of the polymer chain that repeats.
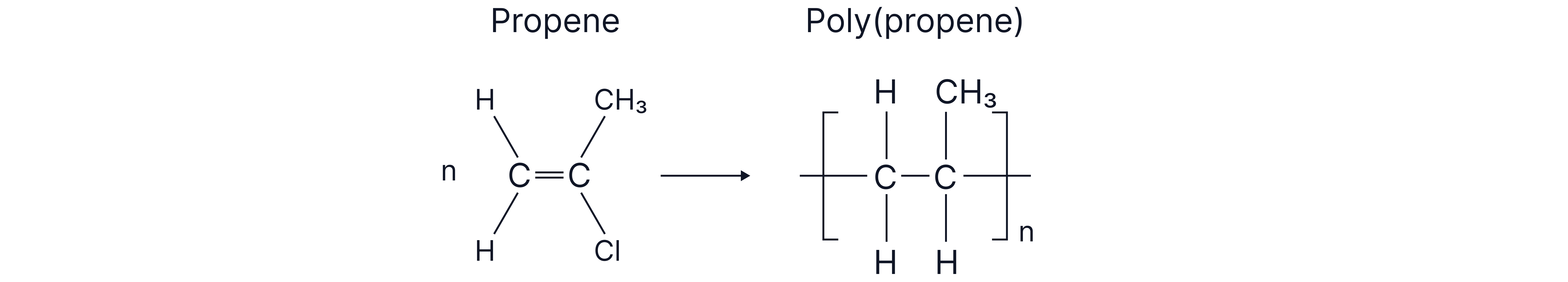
How to draw the polymer (from a monomer)
1. Draw the monomer clearly around its C=C.
2. Open the double bond to two single bonds that continue out of the unit.
3. Put square brackets around the unit and add a small n outside.
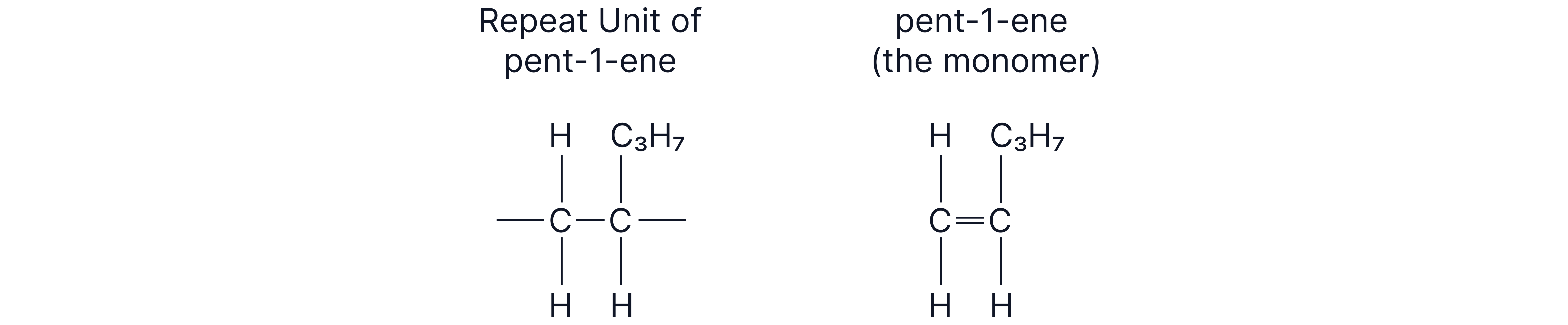
How to find the monomer (from a polymer)
1. Take one repeating unit from the chain.
2. Close the opened bonds to form a double bond.
3. Name the alkene monomer.
Exam Tips
For a repeating unit, draw no “n” on the unit itself.
Show the continuation bonds at each end of the brackets.
Name as poly(monomer name) – e.g. poly(propene).
Forgetting the continuation bonds at each bracket end.
Writing n next to the monomer when the question only asks for the repeating unit.
Misnaming: write poly(propene), not “polypropylene” unless the spec allows it.
Reactivity of Addition Polymers
Addition polymers are unreactive due to:
Strong C–C and C–H bonds
No reactive functional groups
They are saturated, chemically stable, and resist biological degradation.
Intermolecular Forces in Polymers
Intermolecular forces determine physical properties like melting point, flexibility, and hardness.
Examples:
Poly(ethene): flexible, low melting point due to weak van der Waals forces.
Poly(chloroethene) (PVC): stronger dipole–dipole interactions due to polar C–Cl bonds. Waterproof.
Use of PVC and Modification with Plasticisers
PVC (poly(chloroethene)) is naturally rigid due to strong intermolecular forces between chains.
Addition of plasticisers:
Weakens intermolecular forces
Increases flexibility
Allows PVC to be used in applications like cable insulation, flooring, and clothing.
Practice Question
Example 1
Draw the repeating unit of the polymer formed from propene.
Answer:
-[–CH₂–CH(CH₃)–]-ₙ
(Ensure square brackets and extension bonds are drawn.)
Example 2
Name the monomer used to form poly(phenylethene).
Answer:
Phenylethene
Example 3
The structure of a polymer is shown. Deduce the structure of the monomer.
-[–CH₂–CHBr–]-ₙ
Answer:
CH₂=CHBr (bromoethene)