Isomerism in Alkenes
Lajoy Tucker
Teacher

Contents
Introductions and Definitions
Structural isomers are molecules with the same molecular formula but different structural formula.
Stereoisomers are molecules with the same structural formula but different arrangements of atoms in space.
Structural Isomerism in Alkenes
Relevant forms:
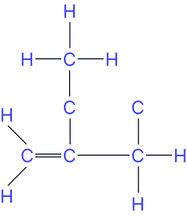
Chain isomers (different carbon chain branching)
e.g. but-1-ene and 2-methylpropene
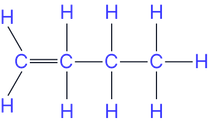
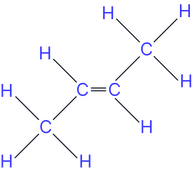
Position isomers (position of the double bond)
e.g. but-1-ene and but-2-ene

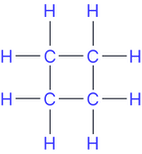
Functional group isomers (alkenes and their cycloalkane isomer)
e.g.but-1-ene and cyclobutane

E/Z Isomerism
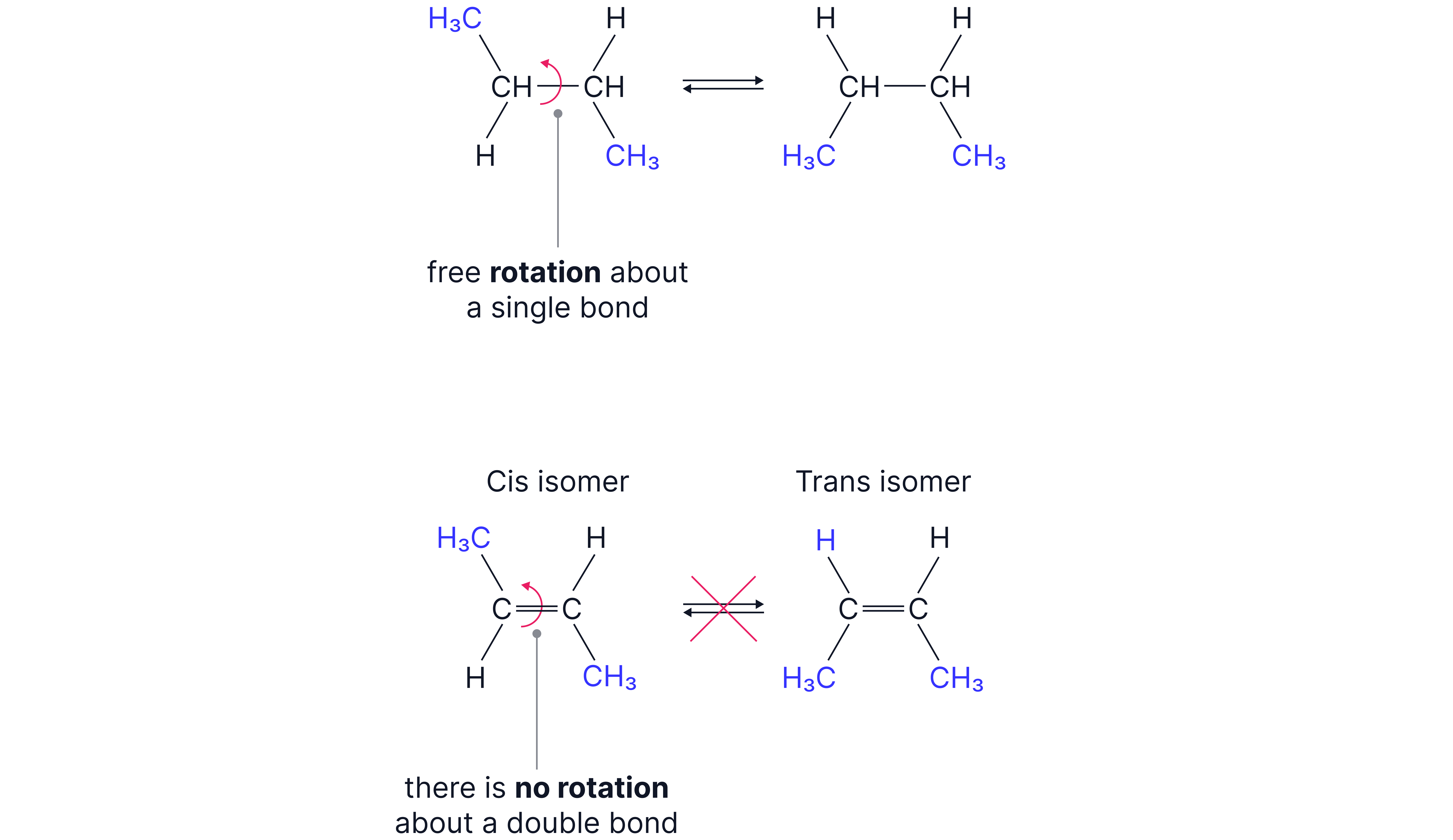
E/Z isomerism is a form of stereoisomerism that arises due to restricted rotation around the C=C double bond in alkenes.
Conditions for E/Z Isomerism
A molecule shows E/Z isomerism if:
1. It contains a C=C double bond which restricts rotation
2. Each carbon in the double bond is attached to two different groups.
Unlike with single bonds, there is no free rotation around double bonds due to the pi-bonds. This means that the substituents on each carbon atom are fixed in place.
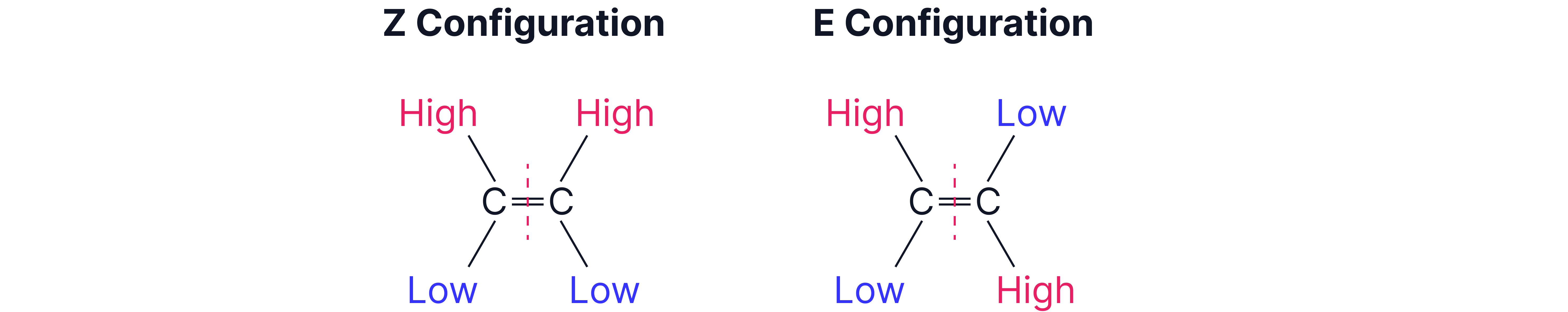
Using Cahn-Ingold-Prelog (CIP) Priority Rules
To assign E or Z:
1. Look at the two atoms directly bonded to each carbon of the C=C.
2. Assign higher priority to the atom with higher atomic number.
3. If both high priority groups are on the same side, it's Z; if on opposite sides, it's E.
Type | Arrangement | Meaning |
Z-isomer | Higher priority groups on same side | Z = zusammen (German: together) |
E-isomer | Higher priority groups on opposite sides | E = entgegen (German: opposite) |


Note – is the same atom is attached for both groups, move along the group until there is a difference in atomic number.
Practice Questions
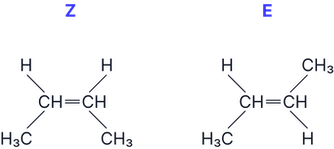
Question 1
Draw and identify the E and Z isomers of but-2-ene
Answer:
Question 2
Explain why alkenes can form E/Z isomers.
Answers:
There is restricted rotation around the C=C bond, and each carbon is bonded to two different groups.
Question 3
Which of the following compounds shows E/Z isomerism?
A.
B.
C.
D.
Answer:
B and D
Key Tips
Always check both carbons in the double bond — both must have two different groups.
Assign priority based on atomic number, not group size or weight.
Don't confuse E/Z with cis/trans — E/Z is more precise and used for non-identical groups.