Reactions of Alkenes
Lajoy Tucker
Teacher

Contents
Alkenes are unsaturated hydrocarbons with a C=C double bond.
The π bond is electron-rich and easily attacked by electrophiles.
Typical reaction type: electrophilic addition.
Key Definitions
Electrophile – an electron pair acceptor.
Addition reaction – two molecules combine to form one product.
Carbocation – an intermediate with a positively charged carbon atom.
Reaction with Bromine
Change in functional group:
Alkene → Dihalogenoalkane
Reagent: Bromine (Br₂)
Conditions: Room temperature (no UV light)
Mechanism: Electrophilic addition
Type of reagent: Electrophile, induced dipole on Br₂ results in
Test for alkenes: Bromine water is an orange solution. When shaken with an alkene, the orange colour disappears (solution becomes colourless).
This happens because the C=C double bond reacts with bromine in an addition reaction. For alkenes, bromine water goes orange → colourless.
For alkanes there is no reaction, bromine water stays orange.
This makes bromine water a simple test for unsaturation (double bonds). It can also show the presence of multiple C=C bonds, e.g. in vegetable oils (polyunsaturated fats).
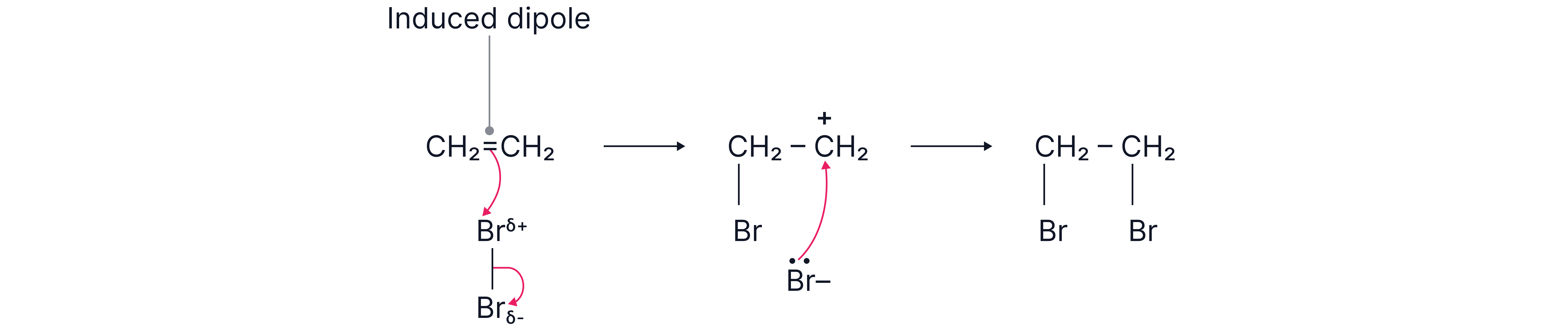
Stepwise mechanism:
1. The π bond repels electrons in Br–Br, inducing a dipole.
2. attacks the double bond, forming a carbocation.
3. Br⁻ bonds to the carbocation → dihalogenoalkane.
Reaction with Hydrogen Halides (HBr, HCl)
Change in functional group:
Alkene → Halogenoalkane
Reagent: HBr or HCl
Conditions: Room temperature
Mechanism: Electrophilic addition
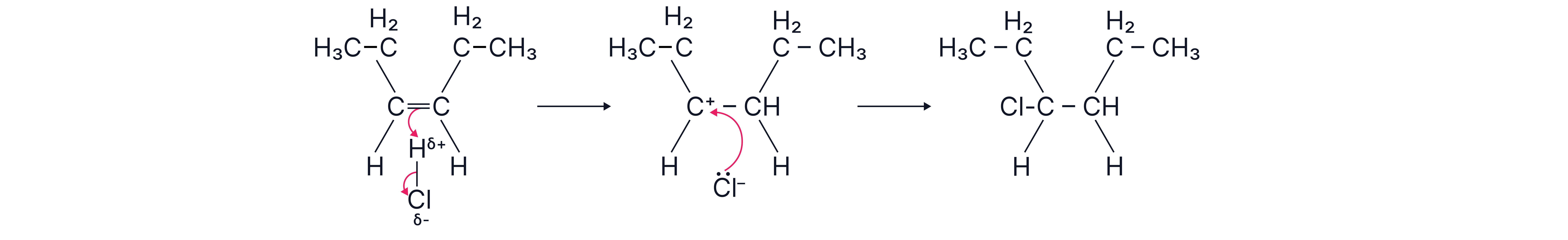
Hex-3-ene + HCl → 3-chlorohexane (only one product).
Unsymmetrical alkene example (Markovnikov’s rule):
When the alkene is unsymmetrical (e.g. propene), two possible products can form depending on which carbon of the C=C the hydrogen attaches to.
Markovnikov’s Rule states that the hydrogen atom bonds to the carbon already bearing the greater number of hydrogens, because this route produces the more stable carbocation intermediate.
Carbocation stability increases in the order tertiary > secondary > primary, due to the electron-releasing inductive effect of alkyl groups.
Propene + HBr → mixture of 1-bromopropane and 2-bromopropane.
Explanation:
1. The π bond attracts from HBr.
2. Two possible carbocations can form:
Primary carbocation (less stable) → leads to 1-bromopropane (minor product).
Secondary carbocation (more stable) → leads to 2-bromopropane (major product).
3. Br⁻ bonds to the carbocation, forming the halogenoalkane.
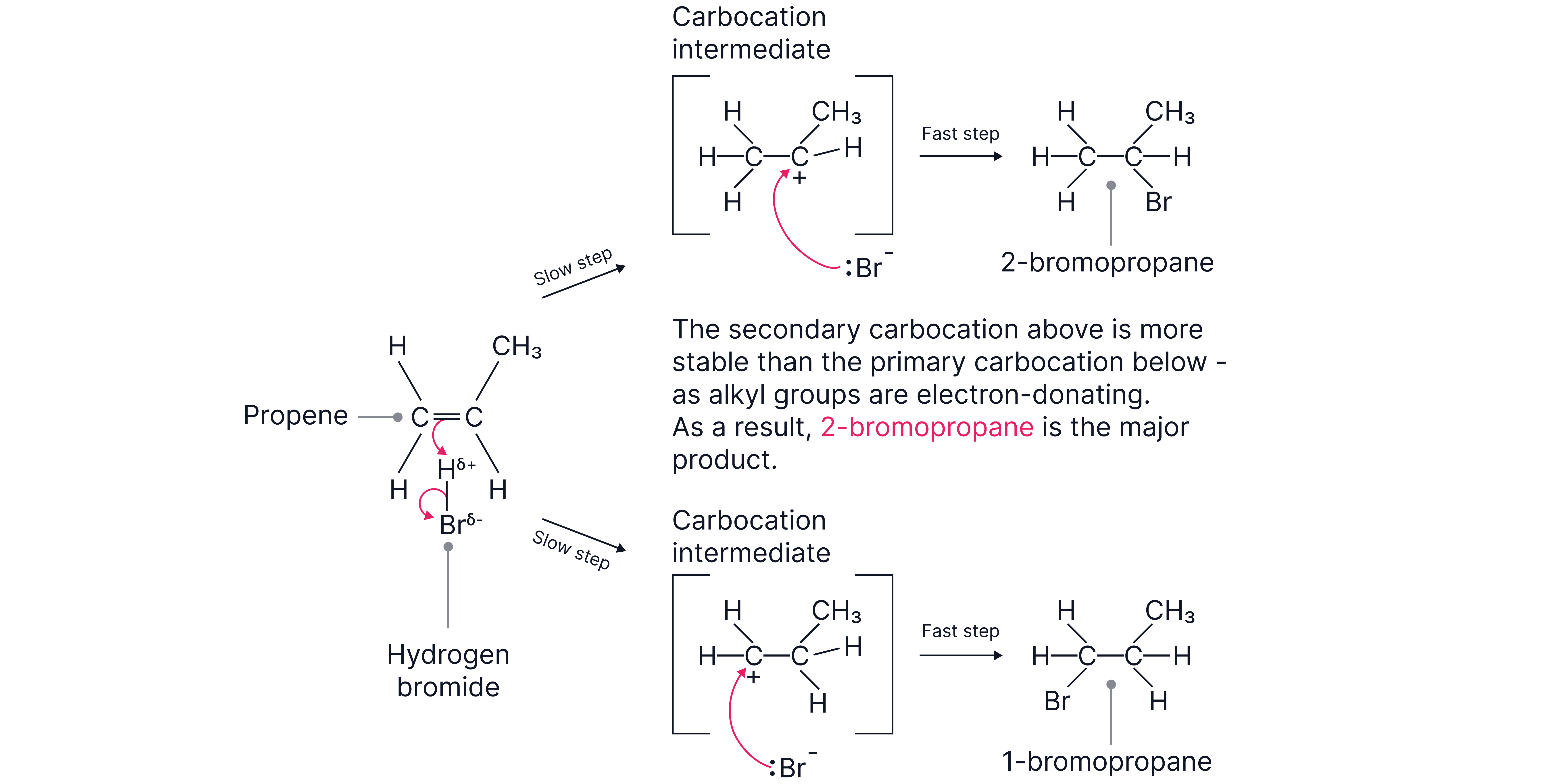
Why one product dominates:
Carbocation stability order: tertiary > secondary > primary.
Alkyl groups are electron-releasing (inductive effect). They push electron density towards the positively charged carbon, reducing its charge and stabilising the carbocation.
In propene, the secondary carbocation is stabilised by two alkyl groups, while the primary carbocation is only stabilised by one.
Therefore, 2-bromopropane is the major product.
Exam tip: Always draw both carbocations and explain stability using the inductive effect. Don’t just quote “Markovnikov’s rule” – explain why.
Reaction of Propene with Sulfuric Acid
Alkenes react with concentrated H₂SO₄ by electrophilic addition, followed by hydrolysis to give an alcohol.
Stage 1: Electrophilic Addition
Change in functional group:
Alkene → Alkyl hydrogensulfate
Reagent: Conc. H₂SO₄
Conditions: Room temperature
Mechanism: Electrophilic addition
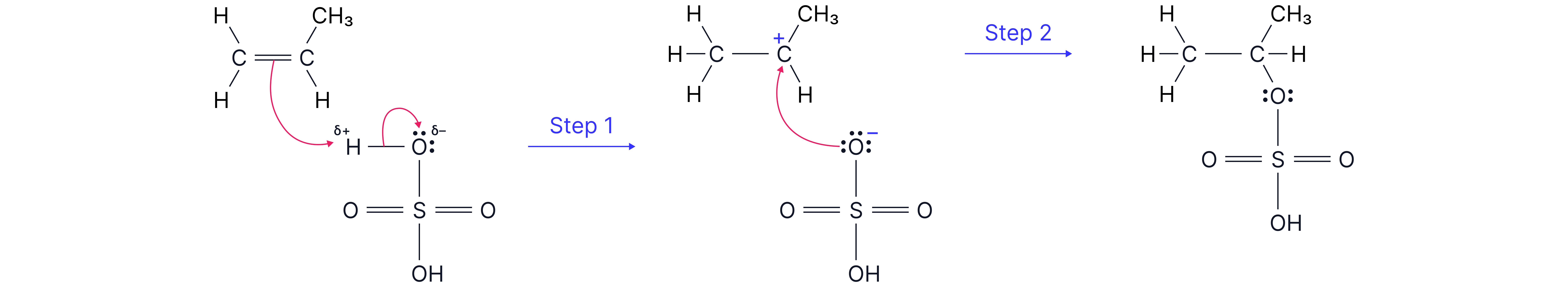
Electrophile: H–OSO₂OH (acts like H⁺)
Example: Propene + H₂SO₄ → isopropyl hydrogensulfate (major product)
Hydrogen adds to the end carbon of the double bond.
This forms a secondary carbocation, which is more stable than a primary carbocation due to the electron-releasing inductive effect of the CH₃ group.
The OSO₂OH⁻ then bonds to give isopropyl hydrogensulfate.
Stage 2: Hydrolysis
Change in functional group:
Alkyl hydrogensulfate → Alcohol
Reagent: Water
Conditions: Warm mixture
Reaction type: Hydrolysis. Hydrolysis means “splitting with water”.
Equation:
CH₃–CH(OSO₂OH)–CH₃ + H₂O → CH₃–CH(OH)–CH₃ + H₂SO₄
So the hydrogensulfate formed from propene is converted into propan-2-ol, with sulfuric acid regenerated.
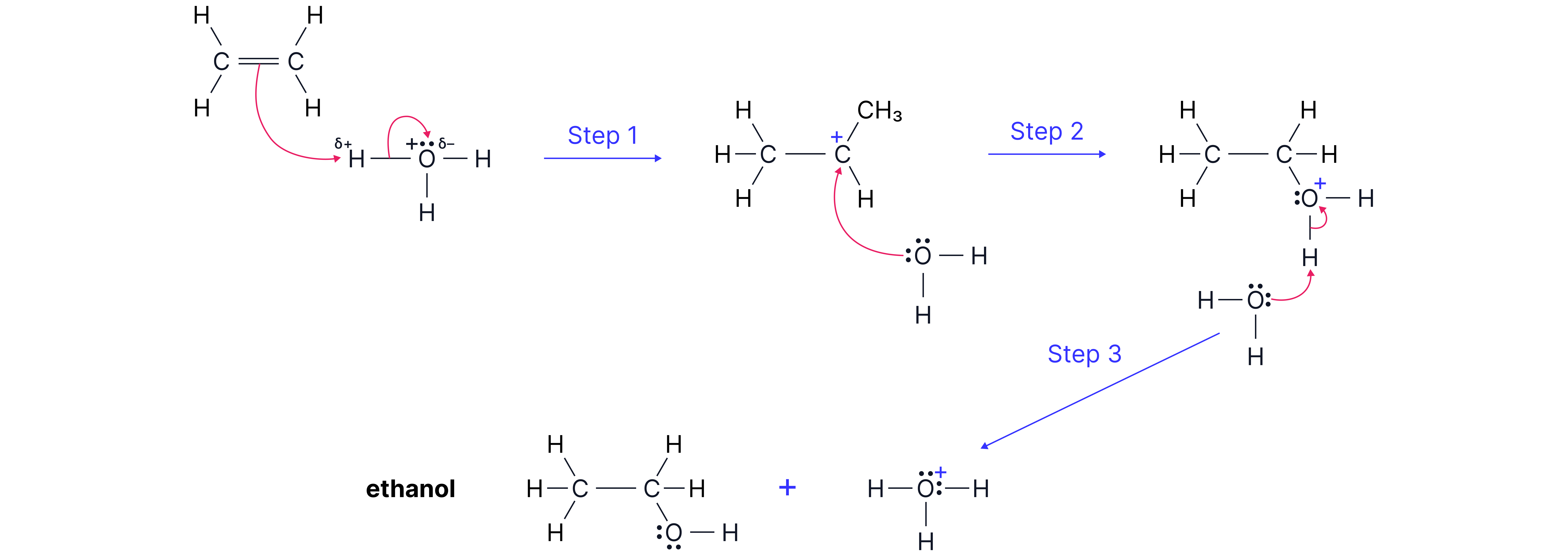
Direct Hydration of Alkenes to Form Alcohols
In industry, alkenes can be converted into alcohols in one step by reacting directly with steam in the presence of an acid catalyst.
Example:
CH₂=CH₂ + H₂O → CH₃CH₂OH
This reaction is called hydration because water is added across the double bond.
Essential Conditions
High temperature: 300–600 °C
High pressure: ≈70 atm
Catalyst: concentrated H₃PO₄ (phosphoric acid)
Why Industry Uses This Process
No waste products → high atom economy.
Products are easier and cheaper to separate than in the two-step sulfuric acid process.
However, conditions are extreme (high T and P), so it cannot be done in a school laboratory.